Abstract
Purpose
To describe the macular findings on optical coherence tomography (OCT) in patients with cat-scratch disease (CSD) neuroretinitis.
Methods
Medical records of all patients diagnosed with CSD neuroretinitis at the Tel Aviv Medical Center between April 2006 and May 2010 were retrospectively reviewed. All patients underwent Stratus OCT macular examination.
Results
Eight eyes of seven patients with confirmed CSD neuroretinitis, (mean age 33±9.9 years, range 6–48 years) were included in the study. All patients presented clinically with optic nerve swelling and macular edema or macular exudates. OCT demonstrated flattening of the foveal contour, thickening of the neurosensory retina, and accumulation of subretinal fluid (SRF) in all studied eyes. Retinal exudates appeared as multiple hyper-reflective foci in the outer plexiform layer. The average central macular thickness was 460 μm (range 170–906 μm) and the average maximal retinal thickness was 613 μm (range 387–1103 μm), at presentation. The macula appeared normal on repeated exams during follow-up.
Conclusion
Similar OCT findings were demonstrated in patients with CSD neuroretinitis. SRF was found in all eyes, although was not visible on clinical examination or fluorescein angiography. OCT may be used as an adjunct imaging tool in the diagnosis and follow-up of patients with CSD neuroretinitis.
Similar content being viewed by others
Introduction
Cat-scratch disease (CSD) was first described in three patients with chronic fever, regional lymphadenopathy, and a follicular conjunctivitis. The association between cats and the disease was first made by Debr’e in 1931.1 Only in 1992 was Bartonella henselae (previously known as Rochalimaea henselae) shown to be the main causative agent.2
Parinaud's oculoglandular syndrome is the most common ocular finding of CSD primary complex, appearing in 2–3% of the patients.3, 4, 5
Disseminated CSD may involve the eye and cause vision-threatening manifestations, including neuroretinitis, optic neuritis, retinitis, choroiditis, anterior uveitis, vitritis, branch retinal arteriolar and venular occlusions, vasculitis, and peripapillary angiomatosis.3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21
In 1970, Sweeney and Drance22 were first to find a connection between CSD and the appearance of neuroretinitis. Since then, numerous reports had been published on cases of neuroretinitis in CSD and it is the most common posterior segment complication of CSD. In the last years, with the development of specific and sensitive serologic investigations for B. henselae infection, CSD was found to be the most common infection associated with neuroretinitis.23 Neuroretinitis is characterized by optic nerve swelling, which usually resolves within 2–8 weeks, accompanied by stellate macular exudates that become prominent over the first 2 or 3 weeks. Additional manifestations that may accompany neuroretinitis include intra-retinal hemorrhages,10 focal or multifocal retinitis or choroiditis,7, 11 retinal artery and vein occlusions,11, 12, 20 and anterior or intermediate uveitis.7, 9
The diagnosis of CSD neuroretinitis is made clinically and confirmed by positive serology and fluorescein angiography (FA). The major finding in FA is late leakage from the optic nerve. Other FA findings are peripapillary angiomatosis and peripapillary serous retinal detachment.24 Macular findings are poorly demonstrated by FA in cases of neuroretinitis.
The aim of the present study was to evaluate optical coherence tomography (OCT) macular findings in eyes with CSD neuroretinitis and describe their features.
Materials and methods
A retrospective chart review of consecutive patients diagnosed with CSD neuroretinitis at the Department of Ophthalmology at the Tel-Aviv Medical Center (Tel-Aviv, Israel) between April 2006 and May 2010 was performed. The study had institutional review board approval and adhered to the Declaration of Helsinki for research involving human subjects.
Patients were included in the study if they met the criteria of a history for contact with cats, presented with clinical signs of neuroretinitis defined as optic nerve swelling and macular edema or macular exudates in a star-shaped figure, had positive serology for B. henselae, and underwent OCT examination.
The enzyme immunoassay for the detection of serum anti-B. henselae antibodies was used in all patients.
The study patients underwent a complete ophthalmologic examination including fundus biomicroscopy. OCT was carried out as soon as neuroretinitis was diagnosed. Four patients underwent FA as well, whereas three other patients refused to have the FA test (refusal of two adults and the parents of a 6-year-old patient). Digital FA pictures were taken with a retina angiograph (Heidelberg Retina Angiograph; Heidelberg Engineering, Heidelberg, Germany). Pictures of the fundus were taken with a Topcon TRC 50X fundus camera (NISCORP, Farmingdale, NY, USA) through a dilated pupil. The Stratus OCT (software ver. 4.0; Carl Zeiss Meditec, Dublin, CA, USA) was used for retinal imaging.
The macular thickness was evaluated by six consecutive macular scans, 6 mm in length, centered on the fovea, at equally spaced angular orientations. The cross-sectional images were analyzed by means of OCT3 mapping software that used an edge detection technique to locate the strongest two edges in each tomogram, presumed to be at the vitreoretinal interface and the anterior surface of the retinal pigment epithelial–choriocapillaris region. Retinal thickness was measured as the distance between these two interfaces at each measurement point along the scan's x-axis.
Results
Eight eyes of seven patients were included. There were three females and four males with a median age of 27. Table 1 summarizes the patients’ demographic data and clinical characteristics. The duration of visual symptoms before referral to our clinic ranged between 1 to 10 days, and the mean follow-up time was 3.7 months (range 1–8 months).
All seven patients (eight eyes) presented with optic nerve swelling and macular edema or macular exudates. Four patients (four eyes) had macular exudates in a star-shaped figuration at presentation, although a macular star was formed in the later course of the disease in the remaining four eyes. Six eyes had a retinal inflammatory lesion adjacent to the optic nerve (Figure 1).
Mean Snellen best-corrected visual acuity (BCVA) at presentation was 20/590 (range 20/25–20/4000). BCVA was better than 20/40 in two eyes, between 20/40 and 20/200 in five eyes, and less than 20/200 in one eye. At the end of follow-up, mean Snellen BCVA was 20/84. Seven eyes of six patients had improved vision, and one patient, who had initially presented with good VA, kept the same VA (Table 1).
All adult patients were treated with doxycycline 100 mg BID and rifampin 300 mg BID. Case 6, a 6-year-old girl, was treated with doxycycline 40 mg BID and rifampin 200 mg. In all patients treatment was given for a period of 4–6 weeks.
OCT scans performed at presentation demonstrated in all eight eyes flattening of the foveal contour, retinal thickening, and accumulation of subretinal fluid (SRF). In all eyes the SRF was present underneath the foveal center as well as in the parafoveal area, except case 4 that had only parafoveal SRF. In 7/8 eyes retinal exudates appeared as multiple hyper-reflective foci in the outer plexiform layer.
Quantitative evaluation of macular thickness revealed an average central macular thickness of 460 μm (range 170–906 μm) and an average maximal neurosensory retinal thickness of 613 μm (range 387–1103μm). Repeated OCT scans were performed in three eyes (cases 1 and 3, Table 1). Retinal thickening and SRF, which were observed during the first 2 weeks, gradually resolved to normal thickness retina with complete resolution of the SRF by 2 months. Retinal exudates were first visible after 1 week, remained visible up to 2 months, but were not detected at 4 months.
Five eyes of four patients (cases 1, 3, 4, and 7, Table 1) underwent FA. Optic nerve swelling was clearly visible on the red-free images. FA demonstrated optic nerve leakage in all five eyes. In 4/5 eyes (cases 1, 3, and 7, Table 1) the peripapillary inflammatory mass appeared as a cluster of telangiectatic vessels on the optic nerve edge with early hyper-fluorescence leaking at late stages of the angiogram. The retinal vessels had a normal appearance on FA, and there was no evidence of macular leakage or any other macular pathology.
Representative case (case 3)
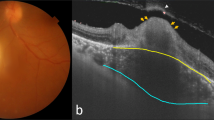
A 20-year-old generally healthy man presented with severe loss of vision in his right eye following an acute febrile illness. He declared contact with cats. At presentation BCVA was 20/160 in the right eye (RE) and 20/20 in the left eye. In the RE an afferent pupillary defect was detected. Fundus biomicroscopy showed optic nerve swelling with a retinal infiltrate adjacent to optic nerve and macular edema. The left eye was normal. FA demonstrated peripapillary telangiectasis on the optic nerve head with early hyper-fluorescence and late leakage (Figure 2a). OCT at presentation revealed central retinal thickening of 611 μm and the presence of subfoveal fluid (Figure 2b). Laboratory tests were remarkable for high anti-B. henselae IgM and IgG serum antibodies. Antibiotic treatment was initiated. Eight days later a macular star of exudates was observed and the OCT demonstrated a decrease in central retinal thickness to 330 μm, as well as hyper-reflective foci located at the outer plexiform layer representing the exudates visible on clinical examination (Figure 2c). During the follow-up of 6 months, BCVA improved to 20/20, a gradual resolution of the optic nerve swelling as well as complete resolution of the macular exudates was observed. These findings coincided with the normal macular appearance measuring 154 μm on OCT (Figure 2d and Table 1).
(a) Fluorescein angiography of patient no. 3. Peripapillary teleangiectasis temporal to the optic nerve with late leakage from the optic nerve. (b) Optical coherence tomography of patient 3. Significant thickening of neurosensory retina (white asterisk) and subretinal fluid (red asterisk). (c) Optical coherence tomography of patient 3, 8 days later. Generation of foveal contour, reduction of retinal thickening (white asterisk), and partial absorption of subretinal fluid (red asterisk). Hyper-reflective signals (red arrow) at the level of the outer plexiform layer representing retinal exudates. (d) Optical coherence tomography of patient 3 after 6 months shows normal macular OCT.
Discussion
This study describes macular OCT findings in eyes with CSD neuroretinitis. To the best of our knowledge, this is the first study to report these findings. Similar macular changes were found in all eyes. The main findings were flattening of the foveal contour, thickening of the neurosensory retina and presence of SRF. Macular exudates were clearly depicted by the OCT scans.
Optic nerve head leakage can be easily demonstrated on FA in cases of CSD neuroretinitis, however, macular changes are poorly visible,10, 11 as was also shown in our study. We showed that the OCT is able to accurately demonstrate the macular findings described above in a qualitative and a quantitative manner. The interesting finding in our study was the presence of SRF in all eyes. The optic nerve microvasculature is the target tissue of Bartonella infection and its vasculopathic effects causes leakage with formation of intra- and subretinal fluids.25 OCT provided quantitative measurements of central macular thickness, maximal retinal thickness, and SRF height, thereby allowing precise objective assessment and follow-up of the macular involvement in CSD neuroretinitis. The presence of SRF can be harmful to the photoreceptors; the height and duration of fluids can be depicted by the OCT examination and may help us to decide upon treatment duration in cases of CSD neuroretinitis.
The macular star formation is caused by leakage of lipid-rich exudates originating from the optic nerve permeable capillaries, along the outer plexiform layer. As the serum is absorbed, the lipid precipitates in a stellate pattern around the fovea.4 Ayata et al26 recently reported that macular star exudates appeared as hypo-autofluorescent star-shaped lesions because of their blockage of retinal pigment epithelial cells. In our study, exudates appeared on OCT as multiple hyper-reflective foci in the outer plexiform layer.
The major limitation of this study is its retrospective nature, which precluded a controlled follow-up. In addition, although the optic nerve is involved to a significant extent in CSD neuroretinitis, it had not been evaluated in these patients. The OCT images at our disposal were acquired by a time domain OCT. The new generation spectral-domain OCT devices work on the basis of spatially encoded frequency, provide a much higher resolution, and can be expected to deliver even better insight into retinal pathology in CSD neuroretinitis.
In summary, the macular edema and exudates that were demonstrated in the clinical examination were quantitatively and qualitatively demonstrated by the OCT. Moreover, SRF that was not detected on clinical examination and FA were clearly demonstrated in the OCT. Our data suggest that OCT is a valuable imaging tool in the diagnosis and follow-up in cases of CSD neuroretinitis.

References
Jerris RC, Regnery RL . Will the real agent of cat-scratch disease please stand up? Annu Rev Microbiol 1996; 50: 707–725.
Bergmans AM, Groothedde JW, Schellekens JF, van Embden JD, Ossewaarde JM, Schouls LM . Etiology of cat scratch disease: comparison of polymerase chain reaction detection of Bartonella (formerly Rochalimaea) and Afipia felis DNA with serology and skin tests. J Infect Dis 1995; 171: 916–923.
Ormerod LD, Dailey JP . Ocular manifestations of cat-scratch disease. Curr Opin Ophthalmol 1999; 10: 209–216.
Solley WA, Martin DF, Newman NJ, King R, Callanan DG, Zacchei T et al. Cat scratch disease: posterior segment manifestations. Ophthalmology 1999; 106: 1546–1553.
Cunningham ET, Koehler JE . Ocular bartonellosis. Am J Ophthalmol 2000; 130: 340–349.
Ulrich GG, Waecker Jr NJ, Meister SJ, Peterson TJ, Hooper DG . Cat scratch disease associated with neuroretinitis in a 6-year-old girl. Ophthalmology 1992; 99: 246–249.
Golnik KC, Marotto ME, Fanous MM, Heitter D, King LP, Halpern JI et al. Ophthalmic manifestations of Rochalimaea species. Am J Ophthalmol 1994; 118: 145–151.
Wong MT, Dolan MJ, Lattuada CP, Regner RL, Garcia ML, Mokulis EC et al. Neuroretinitis, aseptic meningitis, and lymphadenitis associated with Bartonella (Rochalimaea) henselae infection in immunocompetent patients and patients infected with human immunodeficiency virus type I. Clin Infect Dis 1995; 21: 352–360.
Ghauri RR, Lee AG . Optic disk edema with a macular star. Surv Ophthalmol 1998; 43: 270–274.
Reed JB, Scales KD, Wong MT, Lattuada CP, Dolan MJ, Schwab IR . Bartonella henselae neuroretinitis in cat scratch disease: diagnosis, management, and sequelae. Ophthalmology 1998; 105: 459–466.
Ormerod LD, Skolnick KA, Menosky MM, Pavan PR, Pon DM . Retinal and choroidal manifestations of cat-scratch disease. Ophthalmology 1998; 105: 1024–1031.
Wade NK, Po S, Wong IG, Cunningham Jr ET . Bilateral Bartonella-associated neuroretinitis. Retina 1999; 19: 355–356.
Thompson PK, Vaphiades MS, Saccente M . Cat-scratch disease presenting as neuroretinitis and peripheral facial palsy. J Neuroophthalmol 1999; 19: 240–241.
Gray AV, Michels KS, Lauer AK, Samples JR . Bartonella henselae infection associated with neuroretinitis, central retinal artery and vein occlusion, neovascular glaucoma, and severe vision loss. Am J Ophthalmol 2004; 137: 187–189.
Irshad FA, Gordon RA . Bartonella henselae neuroretinitis in a 15-year-old girl with chronic myelogenous leukaemia. J AAPOS 2009; 13: 602–604.
Brazis PW, Stokes HR, Ervin FR . Optic neuritis in cat scratch disease. J Clin Neuroophthalmol 1986; 6: 172–174.
Patel SJ, Petrarca R, Shah SM, Zimmer-Galler I, Janjua KA, Do DV . Atypical Bartonella hensalae chorioretinitis in an immunocompromised patient. Ocul Immunol Inflamm 2008; 16: 45–49.
Soheilian M, Markomichelakis N, Foster CS . Intermediate uveitis and retinal vasculitis as manifestations of cat scratch disease. Am J Ophthalmol 1996; 122: 582–584.
Cohen SM, Davis JL, Gass DM . Branch retinal arterial occlusions in multifocal retinitis with optic nerve edema. Arch Ophthalmol 1995; 113: 1271–1276.
Waisbourd M, Goldstein M, Giladi M, Shulman S, Loewenstein A . Cat-scratch disease associated with branch retinal artery occlusion. Retin Cases Brief Rep 2010; 4: 28–30.
Fish RH, Hogan RN, Nightingale SD, Anand R . Peripapillary angiomatosis associated with cat-scratch neuroretinitis. Arch Ophthalmol 1992; 110: 323.
Sweeney VP, Drance SM . Optic neuritis and compressive neuropathy associated with cat scratch disease. Can Med Assoc J 1970; 103: 1380–1381.
Suhler EB, Lauer AK, Rosenbaum JT . Prevalence of serologic evidence of cat scratch disease in patients with neuroretinitis. Ophthalmology 2000; 107: 871–876.
Cunningham ET, McDonald HR, Schatz H, Johnson RN, Ai E, Grand MG . Inflammatory mass of the optic nerve head associated with systemic Bartonella henselae infection. Arch Ophthalmol 1997; 115: 1596–1597.
Gass JDM . Diseases of the optic nerve that may simulate macular disease. Trans Am Acad Ophthalmol Otolarnygol 1977; 83: 763–770.
Ayata A, Unal M, Erşanli D, Tatlipinar S . Fundus autofluorescence imaging of macular star. Acta Ophthalmol 2009; 87: 690–691.
Acknowledgements
We would like to thank the photographer Galit Yair-Pur for assisting with the OCT, FA, and fundus pictures.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Habot-Wilner, Z., Zur, D., Goldstein, M. et al. Macular findings on optical coherence tomography in cat-scratch disease neuroretinitis. Eye 25, 1064–1068 (2011). https://doi.org/10.1038/eye.2011.125
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/eye.2011.125
Keywords
This article is cited by
-
Inner choroidal ischaemia and CNV due to handheld laser-induced maculopathy: a case report and review
Eye (2020)
-
Treatment Strategies for Neuroretinitis: Current Options and Emerging Therapies
Current Treatment Options in Neurology (2019)
-
Dynamics of intraretinal fluid accumulation evidenced by SD-OCT in a case of cat scratch neuroretinitis
Eye (2014)
-
Atypical cat scratch disease with vitritis, serous macular detachment, neuroretinitis, and retrobulbar optic neuritis
Graefe's Archive for Clinical and Experimental Ophthalmology (2013)