Abstract
Non-invasive prenatal testing (NIPT) using cell-free DNA for aneuploidy is a highly accurate screening test; however, concerns exist around the potential for routinisation of testing. The multidimensional measure of informed choice (MMIC) is a quantitative instrument developed to assess informed choice for Down syndrome screening (DSS). We have validated a modified MMIC for NIPT and measured informed choice among women offered NIPT in a public health service. The measure was distributed to women recruited across eight maternity units in the United Kingdom who had accepted DSS. Construct validity was assessed by simultaneously conducting qualitative interviews. Five hundred and eighty-five questionnaires were completed and 45 interviews conducted after blood-draw (or equivalent for those that declined NIPT). The measure demonstrated good internal consistency and internal validity. Results indicate the vast majority of women (89%) made an informed choice; 95% were judged to have good knowledge, 88% had a positive attitude and 92% had deliberated. Of the 11% judged to have made an uninformed choice, 55% had not deliberated, 41% had insufficient knowledge, and 19% had a negative attitude. Ethnicity (OR=2.78, P=0.003) and accepting NIPT (OR=16.05, P=0.021) were found to be significant predictors of informed choice. The high rate of informed choice is likely to reflect the importance placed on the provision of pre-test counselling in this study. It will be vital to ensure that this is maintained once NIPT is offered in routine clinical practice.
Similar content being viewed by others
Introduction
In most developed countries it is now routine practice to offer pregnant women Down syndrome screening (DSS) followed by invasive testing for definitive diagnosis for those women at increased risk.1 The limitations of this approach are that DSS currently has a detection rate (DR) of ~80–90% and a false-positive rate (FPR) of 3–5%,2 and invasive testing through chromosome analysis of amniocytes or chorionic villus samples carries a procedure-related miscarriage risk of 0.5–1%.3 Following the identification of cell-free DNA in the maternal plasma in the late 1990’s4 together with the technological advances delivered by next-generation sequencing, it has become possible to offer non-invasive prenatal testing (NIPT) for aneuploidy. It has been clinically available in the private sector since 2011, and is currently being evaluated for use within several publicly funded health-care systems.5, 6, 7 It has high DRs and low FPRs for Down syndrome (99.0% DR; 0.08% FPR) as well as trisomies 18 (92.1% DR; 0.20% FPR) and 13 (88.6%DT; 0.12F PR),8 but as NIPT requires confirmation through invasive testing it is considered to be an advanced screening test.9 The clinical advantages of NIPT are the reduced need for invasive testing, and the opportunity for earlier reassurance as NIPT can be conducted from 10 weeks gestation. Concerns around routinisation of testing because of the ease with which this near-diagnostic test can be conducted have also been raised.10, 11 Consequently, much discussion has centred around the importance of non-directive counselling and ensuring informed choice when offering NIPT.1, 9 As such, any evaluation of implementing NIPT into clinical practice should include an assessment of whether women are making informed choices.
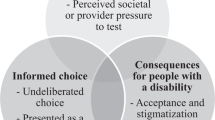
There is a consensus in clinical guidelines published in the United Kingdom, United States of America and elsewhere that prenatal testing services should facilitate women to make informed choices.12, 13, 14 Although numerous definitions of informed choice exist,15, 16, 17 how to assess informed choice has been widely debated and several validated measures have been developed.17, 18, 19 One of the most accepted quantitative measures is the Multidimensional Measure of Informed Choice (MMIC),17, 20 which is based on a definition of informed choice being one made with sufficient knowledge, in line with the persons values and attitudes to testing, and behaviourally implemented. The MMIC was originally developed for women undergoing DSS,20 but has been adapted for other conditions including screening for thalassaemia and sickle cell,21 fragile X syndrome22 and cancer.23 Our aim was to validate a modified MMIC instrument for NIPT and measure informed choice among women offered NIPT following DSS in a public health service.
Materials and methods
We describe the development of a modified MMIC as a measure of informed choice for women offered NIPT for aneuploidy following DSS. This work forms part of a larger study (The NIPT Evaluation Study) evaluating the implementation of NIPT within the National Health Service (NHS) (www.rapid.nhs.uk/).5 NHS Research Ethics Committee approval was obtained (London – Camden and Islington 13/LO/0082) in February 2013.
Adaptation of MMIC
The original MMIC comprised a knowledge scale, scored as good or poor knowledge, and an attitude scale, scored as positive or negative attitude, which was then combined with test behaviour to determine whether an informed choice had been made.17 To refine the MMIC for use with NIPT a multidisciplinary expert panel was established to assist in this process. They comprised a specialist in fetal medicine and NIPT, a genetic counsellor with expertise in developing competences for health professionals, the Associate Director of the NHS Genetics and Genomics Education Centre, a research genetic counsellor, two research midwives and representatives from the patient organisations Antenatal Results and Choices and Genetic Alliance UK.
Knowledge scale
Adaption of the MMIC requires the development of a knowledge scale specifically for the condition and screening test being used. An initial set of 11 knowledge items for NIPT were generated by CL. These were informed by previous qualitative interviews exploring patient’s informational needs regarding NIPT,11 a patient information leaflet developed by the expert panel for use in the Evaluation Study, clinical guidelines for offering NIPT1, 24 and questions used in the original MMIC knowledge scale.17 The knowledge items were designed to cover following four domains: (1) characteristics of NIPT, (2) how NIPT compares with standard DSS, (3) implications of testing and (4) knowledge about condition(s) for which testing is being offered. These were presented in a multiple choice format with a range of three to six potential choices. To refine the knowledge items, a series of consultations were conducted with the members of the expert panel. During the first round of consultation, the expert panel was asked to rate each knowledge item on a scale of 1–5 (1=not at all, 5=extremely) in terms of both the importance and difficulty. The panel was also asked whether any important questions were missing. Following consultation, some questions were reworded and one additional question was included to address participants’ understanding of a ‘highly unlikely’ NIPT result.
During the second round of consultation the panel was again asked to rate the importance and difficulty of each question. No further amendments to the questions were made. As there is no external criteria for what constitutes ‘good’ or ‘poor’ knowledge, the panel agreed a cutoff of ≥9/12 would constitute good knowledge. In addition, it was agreed that the responder had to answer the question about what condition(s) NIPT was testing for correctly as this was considered crucial to making an informed choice.
Attitude scale
The items for inclusion into the attitude scale were also subject to a round of consultation with the expert panel. It was agreed to include three of the four items used in the original MMIC scale17 (beneficial/harmful; important/unimportant; good thing/bad thing), which had been validated in the prenatal setting.20 However, there was consensus that the fourth item (pleasant/unpleasant) did not represent an important aspect of women’s attitude towards NIPT because women have many blood tests during pregnancy. The panel agreed to include two other items, desirable/undesirable and reassuring/not reassuring, which had been validated in other modified MMIC scales.25, 26 The expert panel was asked to agree a cutoff for the attitude scale. The options included the midpoint of the scale (≥11, range 0–20) in line with the original application of the MMIC attitude scale20 or an alternative cutoff used by Van den Berg and colleagues27 in which attitude is scored into three equal categories (positive, neutral and negative) and people with a neutral attitude are removed from the informed choice calculation. The expert panel unanimously agreed on the Van den Berg approach.
Deliberation scale
Deliberation is considered to be a key component of the decision-making process in many health behaviour theories,28, 29 and a deliberation scale has been included alongside the original MMIC in a previous study measuring informed choice in prenatal screening.30 The panel agreed to include the deliberation scale in the questionnaire and the results of informed choice are presented both with and without this scale.
Piloting the questionnaire
In the final stage of development the MMIC was piloted with 20 women who had been offered NIPT as part of the NIPT Evaluation Study. A researcher (CL) then conducted a short face to face interview with each responder to assess the clarity of question and answer options. Some minor modifications to wording were made. The questionnaire including the final MMIC scale is included in the Supplementary Information.
Study procedure
A protocol detailing recruitment into the Evaluation Study has been published.5 In brief, women were recruited through one of eight antenatal clinics located across the United Kingdom, which varied in terms of the demography of their catchment areas. Women were eligible to participate if they had accepted DSS as part of routine care, were older than 16 years, had a singleton pregnancy and able to read and understand English (owing to resource constraints we were unable to translate the information materials and questionnaire) and provide informed consent. Those with an intermediate (1:1000–1:151) or high-risk result (1:150–1:2) following DSS were invited into the Evaluation Study and offered NIPT free of charge. Women with an intermediate risk DSS result were offered NIPT or no further testing; women with a high-risk DSS result were offered NIPT, invasive testing, NIPT and invasive testing or no further testing. All women received individualised pre-test counselling with a dedicated NIPT research midwife as well as written information about NIPT at two time points (booking-in and pre-test counselling) prior to decision-making. Information given to women during pre-test counselling is summarised in Figure 1. A subset of women eligible for the Evaluation Study was invited to take part in the Informed Choice study. Women were recruited as a consecutive sample between December 2013 and February 2015. This was a longitudinal cohort study involving questionnaires and interviews at two time points. Only questionnaire and interview responses from the first time-point (T1) are presented here as these relate directly to validation of the amended MMIC.
Questionnaires
The first questionnaire (Q1) was given to women after blood had been taken (or within a similar timeframe for women who declined testing). Q1 contained the modified MMIC as well as: The Decisional Conflict Scale;31 the short form of the State Trait Anxiety Inventory (STAI-6);32 questions to explore motivations for testing and preference for test attributes; questions on parity and socio-demographic questions (Table 1). The results from these additional questions are presented in a forthcoming paper (‘Women’s experience of being offered NIPT for aneuploidy in a national health service setting’ manuscript in preparation). Participants were requested to leave their contact details so that a follow-up questionnaire could be sent and indicate their willingness to take part in an interview. We aimed to collect at least 50 completed questionnaires from women seen consecutively at each of eight participating maternity units. On the basis of the results of the informed choice study by Michie et al.20 a sample of 350 completed questionnaires would detect a difference of 10% in rates of informed choice with 97% power at the 5% level of significance.
Interviews
Semi-structured interviews were conducted with a subset of women. The interviews included questions drawn from the concepts of the MMIC (knowledge, attitude and deliberation). Construct validity of the measure was assessed by comparing women’s scores on the questionnaire with their responses during interview. Interviews were also used to validate the cutoff for good knowledge. In asking women to recount what information they remembered being given about NIPT, the aim was not only to assess recall of information but also ensure understanding of the implications of the test. Women’s attitudes towards NIPT were assessed by exploring their reasons for accepting or declining testing, and the values underpinning those attitudes. Deliberation was assessed by exploring whether they had considered the possible consequences of NIPT or evaluated alternative options. Participants were purposively sampled to represent a range of demographic groups (educational level, ethnicity, religion and religiosity), testing choices, MMIC scores and test outcomes. Interviews were conducted until saturation was reached.
Data analysis
Analysis of the MMIC
To assess informed choice, knowledge scores (which were dichotomized as good or poor), attitude scores (which were classified as positive, negative or neutral with neutral responders being omitted) and deliberation scores (which were dichotomized as deliberated or non-deliberated) were combined with test behaviour. When participants had good knowledge, had deliberated, and if attitude was consistent with test uptake, an informed choice had been made. If knowledge was poor, the choice was not deliberated or if attitude was not consistent with test uptake, an uninformed choice had been made.
Reliability of the knowledge, attitude and deliberation scales was assessed using Cronbach’s alpha, which measures internal consistency. Alpha values of 0.7–0.9 are assumed to indicate good internal consistency.33 Construct validity of the adapted attitude scale was also measured using factor analysis to determine whether the items spanned one or more dimensions.34 To validate the cutoff for good knowledge women’s mean knowledge scores were compared using the t-test. To look at the impact of socio-demographic variables on informed choice, descriptive analysis was conducted on single items; relationships between groups were tested using χ2-tests and Fisher’s exact test for categorical variables and the Kruskal–Wallis test for continuous variables. Owing to the dominance of one category in the variables education, ethnicity and religion, the categories were collapsed into binary categories to strengthen the analysis, ie, White ethnicity vs other, degree education or higher vs other, Christian vs other. Logistic regression analysis was conducted to determine, which independent variables were significant predictors of informed choice. Missing data on the knowledge scale were treated as incorrect answers. Questionnaires with missing data on the attitude and knowledge scale were removed from analysis as they prohibited calculation of informed choice. Data were analysed using SPSS 22 (IBM, Chicago, IL, USA).
Analysis of qualitative interviews
Construct validity of the MMIC was also assessed by comparing results of the component scales with qualitative responses from interviews. This validation process was undertaken by two researchers independently (CL and MH). Qualitative data were independently coded by CL and MH to score knowledge (good or poor), attitude (positive, negative or neutral) and deliberation (deliberated or non-deliberated). The results were compared following independent analysis and any discrepancies discussed until consensus was reached. For each participant the assessment of whether an informed choice had been made was then compared back to the assessment made via the MMIC.
Results
Participant characteristics
In total, 731 women were invited to take part in the Informed Choice study and 593 agreed to participate and completed Q1 (81% response rate). During the first phase of data analysis, eight questionnaires were removed due to missing data (N=585). A summary of maternal characteristics are presented in Table 1. Regarding DSS risk, 72% of women had an intermediate risk and 28% were high risk. Regarding NIPT uptake, in the intermediate risk group (n=421), 97% chose NIPT and 3% chose no further testing. In the high-risk group (n=163), 86% chose NIPT and 14% chose NIPT and invasive testing. We do not have demographic or testing decision data for participants who declined to participate in the Informed Choice study.
Internal reliability
The alpha coefficients for the knowledge, attitude and deliberation scales indicated internal reliability (Table 2). The screening test for the factor analysis showed an elbow between factors 1 and 2. The factor, with an eigenvalue of 4.12, accounted for 82% of the variability across the five items of the attitude scale, indicating construct validity.
Construct validity
Forty-five interviews were conducted with women at T1 to assess and explore knowledge, attitude and deliberation. Forty-three women were judged to have good knowledge (96%), 41 were judged to have a positive attitude (91%) and 44 were judged to have deliberated (98%). Example quotes illustrating knowledge, attitude and deliberation are presented in Table 3. The interview assessment of informed choice was then compared with the corresponding questionnaire. Overall, five (11%) discrepancies were identified; in two cases these related to attitude, in two cases deliberation, and in one case a discrepancy was found for both deliberation and knowledge. In each case the participant had failed to reach the required cutoff on the questionnaire but had displayed adequate knowledge, a positive attitude and deliberation during the interview. The knowledge scores were significantly higher in women judged at interview to have good knowledge (M=11.12, SD=1.31) compared with those judged to have poor knowledge (M=6; SD=1.41), suggesting the cutoff for what constitutes good knowledge was appropriate (t(43)=5.38, P<0.001).
Informed choice
According to the MMIC, 89% (n=467) of women were judged to have made an informed choice about NIPT. When we excluded the deliberation scale this increased slightly to 94% (n=492).
Of the total sample, 95% (n=553) were judged to have good knowledge, 88% (n=513) had a positive attitude (2% had a negative attitude, and 10% had a neutral attitude and were excluded from the informed choice calculation) and 92% (n=539) had deliberated. In all cases apart from one, the informed choices made (when deliberation was included in the measure) were those in which women had good knowledge, a positive attitude, had deliberated on their decision and had undergone testing. In one case a woman had made an informed choice based on good knowledge, deliberation, a negative attitude and had declined invasive testing. Of the 11% (n=58) who were judged to have made an uninformed choice, 55% (n=32) had not deliberated, 41% (n=24) had insufficient knowledge, and 19% (n=11) had a negative attitude (eight of whom accepted NIPT and three of whom declined).
Bivariate analysis showed that women found to have made an informed choice were significantly more likely to be: NIPT accepters (OR=5.38, P<0.001), White (X2(1)=12.99, OR=1.15, P<0.001), university educated (X2(1)=4.10, OR=1.07, P=0.043) and high risk (X2(1)=4.082, OR=1.07, P=0.043). There was no significant difference in rates of informed choice across antenatal clinics (P=0.101). Multivariable regression analysis demonstrated that ethnicity and NIPT uptake were significant predictors of informed choice when controlling for education, age, having a religious faith, parity and screening risk. White participants had almost three times higher odds of making an informed choice than other ethnic groups who were more likely to agree to testing despite their beliefs (OR=2.78, 95% CI: 1.42–5.46, P=0.003); NIPT accepters had 16 times higher odds of making an informed choice than NIPT decliners (OR=16.05, 95% CI: 1.53–168.81, P=0.021) although the large confidence interval indicates a low level of precision due to the small number of NIPT decliners in the study (Table 4).
Knowledge, attitude and deliberation scores
A Kruskal–Wallis test revealed significant differences in knowledge and attitude scores and a number of socio-demographic variables. Knowledge scores were significantly affected by education (H(4)=41.99, P<0.001) and ethnicity (H(4)=20.21, P=<0.001). Pairwise comparisons with a Bonferroni correction for multiple comparisons revealed participants with no qualification (U=191.08, P=0.027, r=−0.16), GCSE/O level qualifications (U=119.03, P<0.001, r=−0.15) and vocational qualifications (U=63.86, P=0.001, r=−0.18) had significantly lower knowledge scores compared with those with a university degree; and Asian participants had significantly lower knowledge scores compared with White participants (U=65.64, P=0.012, r=−0.15). Attitude scores were also significantly affected by education (H(4)=9.99, P=0.041) and ethnicity (H(4)=24.43, P<0.001). Pairwise comparisons revealed participants with vocational qualifications had significantly more negative attitudes towards NIPT compared with those with a university degree (U=46.55, P=0.034, r=−0.14); and Black participants had significantly more negative attitudes compared with White participants (U=112.39, P<0.001, r=−0.21). Screening risk was also found to be significantly associated with attitude and deliberation scores; women with a high-risk DSS result had more positive attitudes towards NIPT than those with a low risk result (U=30846, z=−2.15, P=0.032, r=−0.09) and had deliberated more (U=26504, z=−4.33, P<0.001, r=−0.18).
Discussion
This is, to our knowledge, the first adaptation of the MMIC scale for use with women considering NIPT for aneuploidy. The vast majority of participants in this sample were found to have made an informed choice, although we recognise that these results represent data collected in a research project and may reflect the high importance placed on the provision of pre-test counselling as part of the project. These findings do however emphasise the importance of providing pre-test counselling to provide women with sufficient information and support should NIPT be implemented into routine maternity-care pathways. This finding is in stark contrast to previous research on informed choice in DSS,35 which has generally found rates of informed choice to be low,17, 26, 36, 37 commonly due to poor knowledge.19, 36, 37 A further reason for the high rates of informed choice in our sample may be because women with an increased risk result may have found more relevance in their decision to accept or decline NIPT, leading them to becoming more informed. Introducing NIPT for aneuploidy outside of a research setting is likely to result in less time for pre-test counselling specifically around this test, which has the potential to impact on patient knowledge and consequently informed choice. This concern is supported by recent research in the USA with genetic counsellors offering NIPT for aneuploidy, which suggests that a large number of patients do not understand that NIPT is a screening test.38 If and when the test is adopted into standard maternity-care pathways, evaluation will be needed to confirm whether women are making informed choices.
In this study, non-White participants and participants with a lower educational level were significantly less likely to make an informed choice. This is in line with previous research assessing informed choice among women offered prenatal screening in the United Kingdom, where South Asian and Black African Caribbean women and those from low-socioeconomic backgrounds were less likely to make an informed choice than other women.39 Similarly, in a study conducted in the Netherlands, women of Turkish and Surinamese origin were less likely to make an informed choice than those of Dutch origin.40 These observations support the theory that health-care systems are less good at facilitating informed choice in the prenatal setting for women from minority ethnic groups and from lower educational backgrounds. This may be for a number of reasons including inequity of access for ethnic minority groups,41 or that pre-test counselling and/or informed choice measures are too complex for those with low-literacy levels.42 Our findings suggest that poor knowledge and negative attitudes had a significant role. This emphasises the importance of finding ways to enhance the informed choice process among different cultural and economic groups in order not to exacerbate the health divide, for example through different media formats to enhance understanding or decision aids to help clarify women’s attitudes. Further research in this area would therefore be valuable.
Interestingly, the greatest number of uninformed choices were made as a result of decisions not being deliberated on. This may be because reporting whether one has deliberated on something is more difficult than reporting on one’s knowledge or attitudes, or because women did not necessarily deliberate on the decision to have NIPT because they had already considered DSS and wanted a more accurate assessment of risk. NIPT was essentially a continuation of this pathway. This was a finding that emerged during qualitative interviews. Future use of the Deliberation scale in this context could include rewording so that deliberation encompasses both screening and NIPT. A further reason may be because women do not give full consideration to NIPT because it is a blood test of which there are a number during pregnancy. This is a concern that has been raised previously by both patients and health-care professionals.11, 43 This emphasises the importance of ensuring that as well as focussing on knowledge, health professionals must encourage patients to consider the implications of the test results, what information would be of benefit to them and what options might be available. We also found that women with a high-risk screening result were more likely to deliberate on NIPT than those with an intermediate risk. This is not unexpected given that that those at a higher risk in this study may be weighing up the option of NIPT against invasive testing and may consider their decision in more depth because of the potential implications; however, this finding does highlight the need for health professionals to ensure women with a DSS risk <1:150 give sufficient thought to the advantages and disadvantages of NIPT, particularly given that there is still a chance, albeit small, that the fetus may be affected.
The methodology used to modify the MMIC has a number of notable strengths including the use of qualitative methods to explore the decision-making process, which has provided an additional dimension to the evaluation and validation of the modified MMIC.44 There was a high concordance between the modified MMIC and the interviews in determining whether an informed choice had been made. However, although overall the modified MMIC appears to be a robust measure of informed choice, like other measures it is not possible to pick up subtle nuances in decision-making that may be evident from qualitative interviews or clinical history. For example, in one case there was a discordant result whereby the questionnaire indicated a non-deliberated decision. However, the interview revealed that the woman had in fact deliberated but that this had occurred much earlier on in the process when considering DSS. For this woman, the decision to have NIPT was an extension of the decision to have screening and she therefore made the decision to accept NIPT relatively quickly and without much consideration. In another case a participant’s neutral attitude score was found to most likely stem from a dislike of needles rather than a neutral attitude to NIPT, which she viewed positively as an opportunity for reassurance. A further limitation of the MMIC is that it is unable to account for situations whereby a positive attitude towards NIPT is not behaviourally implemented for practical reasons, eg, being unwilling to return to the clinic for a blood test due to time or cost implications. These findings highlight a potential limitation of the questionnaire in that it is unable to capture the reasons underpinning women’s attitudes and highlights the value of a mixed methods approach supported by interviews as in this study.
The measure was validated among women considering NIPT as a second screening test following a high- or medium-risk DSS result and not women using NIPT as a first screening test. Further validation would need to take place to use the measure among these different population groups. We were only able to recruit a small number of NIPT decliners (n=13) into the study and of these the vast majority were judged to have made an uniformed choice. However, given the small number, this finding should be interpreted with caution, particularly as others have reported higher rates of informed choice in screening decliners.19, 27 Our sample predominantly comprised older, well-educated women, however this probably reflects the fact that older women are more likely to be at increased risk and be highly educated as they have delayed child bearing for educational or vocational reasons. Currently, it is not clear whether NIPT will be offered as a contingent screening test or a first-line test offered to all women. Offering NIPT to a wider group of women may impact rates of informed choice and will thus require further assessment. Lastly, the high rate of informed choice may reflect the fact that women were given up to 30 min pre-test counselling with a dedicated NIPT midwife and written information at a number of different points prior to decision-making. This degree of input may be challenging in routine clinical settings, especially if there is not the same emphasis on pre-test counselling, highlighting the need for evaluation following implementation.
Conclusion
We have developed and validated a measure of informed choice for women offered NIPT for aneuploidy, which can be used in other maternity settings. Previous concerns have been raised relating to routinisation of NIPT and erosion of informed choice given the relative ease with which NIPT can be conducted. Whilst recognising that our sample comprised a high proportion of well-educated women, our results indicate that given adequate information, time and expert pre-test counselling a high rate of informed choice can be achieved. Further research as and when NIPT is implemented into routine clinical settings is required.
References
Benn P, Borrell A, Crossley J et al: Aneuploidy screening: a position statement from a committee on behalf of the Board of the International Society for Prenatal Diagnosis, January 2011. Prenat Diagn 2011; 31: 519–522.
Nicolaides KH : Screening for fetal aneuploidies at 11 to 13 weeks. Prenat Diagn 2011; 31: 7–15.
Tabor A, Alfirevic Z : Update on procedure-related risks for prenatal diagnosis techniques. Fetal Diagn Ther 2010; 27: 1–7.
Lo YM, Corbetta N, Chamberlain PF et al: Presence of fetal DNA in maternal plasma and serum. Lancet 1997; 350: 485–487.
Hill M, Wright D, Daley R et al: Evaluation of non-invasive prenatal testing (NIPT) for aneuploidy in an NHS setting: a reliable accurate prenatal non-invasive diagnosis (RAPID) protocol. BMC Pregnancy Childbirth 2014; 14: 229–239.
Chan YM, Leung WC, Chan WP, Leung TY, Cheng YK, Sahota DS : Women's uptake of non-invasive DNA testing following a high risk screening test for trisomy 21 within a publicly funded Healthcare System: findings from a retrospective review. Prenat Diagn 2014; 35: 342–347.
Gil MM, Akolekar R, Quezada MS, Bregant B, Nicolaides KH : Analysis of cell-free DNA in maternal blood in screening for aneuploidies: meta-analysis. Fetal Diagn Ther 2014; 35: 156–173.
The American College of Obstetricians and Gynecologists Committee on Genetics: Noninvasive prenatal testing for aneuploidy - Committee opinion. Obstet Gynecol 2012; 120: 1532–1534.
de Jong A, Dondorp WJ, de Die-Smulders CE, Frints SG, de Wert GM : Non-invasive prenatal testing: ethical issues explored. Eur J Hum Genet 2010; 18: 272–277.
Lewis C, Silcock C, Chitty LS : Non-invasive prenatal testing for Down’s syndrome – pregnant women’s views and likely uptake. Public Health Genomics 2013; 16: 223–232.
American College of Obstetricians and Gynecologists: ACOG Practice Bulletin No. 77: screening for fetal chromosomal abnormalities. Obstet Gynecol 2007; 109: 217–227.
National Screening Committee. Annual Report: Screening in England. London, 2011-12.
World Health Organisation Proposed International Guidelines on Ethical Issues in Medical Genetics and Genetic Services. Geneva, Switzerland, 1998.
Beauchamp T, Faden R: : Meaning and Elements of Informed Consentin; in Reich W (ed): Encyclopedia of Bioethics. New York: MacMillan,, 1995, pp 1238–1241.
Green JM, Hewison J, Bekker HL, Bryant LD, Cuckle HS : Psychosocial aspects of genetic screening of pregnant women and newborns: a systematic review. Health Technol Assess 2004; 8: iii, ix-x 1–109.
Marteau TM, Dormandy E, Michie S : A measure of informed choice. Health Expect 2001; 4: 99–108.
Irwig L, McCaffery K, Salkeld G, Bossuyt P : Informed choice for screening: implications for evaluation. BMJ 2006; 332: 1148–1150.
Constantine ML, Allyse M, Wall M, De Vries R, Rockwood TH : Imperfect informed consent for prenatal screening: lessons from the Quad screen. Clin Ethics 2014; 9: 17–27.
Michie S, Dormandy E, Marteau TM : The multi-dimensional measure of informed choice: a validation study. Patient Educ Couns 2002; 48: 87–91.
Brown K, Dormandy E, Reid E, Gulliford M, Marteau T : Impact on informed choice of offering antenatal sickle cell and thalassaemia screening in primary care: a randomized trial. J Med Screen 2011; 18: 65–75.
Ames AG, Jaques A, Ukoumunne OC et al: Development of a fragile X syndrome (FXS) knowledge scale: towards a modified multidimensional measure of informed choice for FXS population carrier screening. Health Expect 2012; 18: 69–80.
Steckelberg A, Hulfenhaus C, Haastert B, Muhlhauser I : Effect of evidence based risk information on ‘informed choice’ in colorectal cancer screening: randomised controlled trial. BMJ 2011; 342: d3193.
Skirton H, Goldsmith L, Jackson L, Lewis C, Chitty L : Offering prenatal diagnostic tests – European guidelines for clinical practice. Eur J Hum Genet 2014; 22: 580–586.
van den Bergh KA, Essink-Bot ML, van Klaveren RJ, de Koning HJ : Informed participation in a randomised controlled trial of computed tomography screening for lung cancer. Eur Res J 2009; 34: 711–720.
van den Berg M, Timmermans DR, Ten Kate LP, van Vugt JM, van der Wal G : Are pregnant women making informed choices about prenatal screening? Genet Med 2005; 7: 332–338.
van den Berg M, Timmermans DR, Kleinveld JH, Garcia E, van Vugt JM, van der Wal G : Accepting or declining the offer of prenatal screening for congenital defects: test uptake and women's reasons. Prenat Diagn 2005; 25: 84–90.
Lazarus RS, Folkman S : Stress, appraisal and coping. New York: Springer,, 1984.
Elwyn G, Miron-Shatz T : Deliberation before determination: the definition and evaluation of good decision making. Health Expect 2010; 13: 139–147.
van den Berg M, Timmermans DR, ten Kate LP, van Vugt JM, van der Wal G : Informed decision making in the context of prenatal screening. Patient Educ Couns 2006; 63: 110–117.
O'Connor A User Manual – Decisional Conflict Scale (16 item statement format) Ottawa: Ottawa Hospital Research Institute 1993 (updated 2010). http://decisionaid.ohri.ca/docs/develop/User_Manuals/UM_Decisional_Conflict.pdf.
Marteau TM, Bekker H : The development of a six-item short-form of the state scale of the Spielberger State-Trait Anxiety Inventory (STAI). Br J Clin Psychol 1992; 31: 301–306.
Kline P : The handbook of psychological testing, 2nd edn. London: Routledge, 2000.
Pett MA, Lackey NR, Sullivan JJ : Making Sense of Factor Analysis: The Use of Factor Analysis for Instrument Development in Health Care Research. Thousand Oaks, CA: Sage Publications Inc.,, 2003.
Ames AG, Metcalfe SA, Dalton Archibald A, Duncan RE, Emery J : Measuring informed choice in population-based reproductive genetic screening: a systematic review. Eur J Hum Genet 2015; 23: 8–21.
Dormandy E, Michie S, Hooper R, Marteau TM : Informed choice in antenatal Down syndrome screening: a cluster-randomised trial of combined versus separate visit testing. Patient Educ Couns 2006; 61: 56–64.
Gourounti K, Sandall J : Do pregnant women in Greece make informed choices about antenatal screening for Down's syndrome? A questionnaire survey. Midwifery 2008; 24: 153–162.
Horsting JM, Dlouhy SR, Hanson K, Quaid K, Bai S, Hines KA : Genetic counselors' experience with cell-free fetal DNA testing as a prenatal screening option for aneuploidy. J Genet Couns 2014; 23: 377–400.
Dormandy E, Michie S, Hooper R, Marteau TM : Low uptake of prenatal screening for Down syndrome in minority ethnic groups and socially deprived groups: a reflection of women's attitudes or a failure to facilitate informed choices? Int J Epidemiol 2005; 34: 346–352.
Fransen MP, Essink-Bot ML, Vogel I, Mackenbach JP, Steegers EA, Wildschut HI : Ethnic differences in informed decision-making about prenatal screening for Down's syndrome. J Epidemiol Commun Health 2010; 64: 262–268.
Allford A, Qureshi N, Barwell J, Lewis C, Kai J : What hinders minority ethnic access to cancer genetics services and what may help? Eur J Hum Genet 2014; 22: 866–874.
Dormandy E, Tsui EY, Marteau TM : Development of a measure of informed choice suitable for use in low literacy populations. Patient Educ Couns 2007; 66: 278–295.
Alexander E, Kelly S, Kerzin-Storrar L : Non-invasive prenatal testing: UK genetic counselors' experiences and perspectives. J Genet Couns 2014; 24: 300–311.
Potter BK, O'Reilly N, Etchegary H et al: Exploring informed choice in the context of prenatal testing: findings from a qualitative study. Health Expect 2008; 11: 355–365.
Acknowledgements
We are grateful to the women who participated in the study and the staff who helped to recruit them. This manuscript presents independent research. Work is funded by The National Institute for Health Research (NIHR) under its Programme Grants for Applied Research scheme (RP-PG-0707-10107) (the 'RAPID' Project) and the NIHR Clinical Research Network. LSC is partially funded by the Great Ormond Street Hospital Children’s Charity and the NIHR Biomedical Research centre at Great Ormond Street Hospital NHS Foundation Trust. The funder had no role in the design of the study, collection and analysis of data and decision to publish. The views expressed in the paper are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on European Journal of Human Genetics website
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Lewis, C., Hill, M., Skirton, H. et al. Development and validation of a measure of informed choice for women undergoing non-invasive prenatal testing for aneuploidy. Eur J Hum Genet 24, 809–816 (2016). https://doi.org/10.1038/ejhg.2015.207
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejhg.2015.207
This article is cited by
-
Exploring attitudes and experiences with reproductive genetic carrier screening among couples seeking medically assisted reproduction: a longitudinal survey study
Journal of Assisted Reproduction and Genetics (2024)
-
Couples’ experiences with expanded carrier screening: evaluation of a university hospital screening offer
European Journal of Human Genetics (2021)
-
Awareness of paternal age effect disorders among Japanese pregnant women: implications for prenatal genetic counseling for advanced paternal age
Journal of Community Genetics (2021)
-
Female and male perspectives on male partner roles in expanded carrier screening
Journal of Assisted Reproduction and Genetics (2021)
-
Facilitating autonomous, confident and satisfying choices: a mixed-method study of women’s choice-making in prenatal screening for common aneuploidies
BMC Pregnancy and Childbirth (2018)