- NEWS FEATURE
Electric cars and batteries: how will the world produce enough?

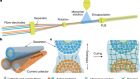
Batteries for an electric car are assembled at the Audi production plant in Brussels. Credit: Audi AG
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 596, 336-339 (2021)
doi: https://doi.org/10.1038/d41586-021-02222-1
References
International Energy Agency. The Role of Critical Minerals in Clean Energy Transitions (IEA, 2021); available at https://www.iea.org/reports/the-role-of-critical-minerals-in-clean-energy-transitions
BloombergNEF. Electric Vehicle Outlook 2021 (BNEF, 2021); available at https://about.bnef.com/electric-vehicle-outlook
Baars, J., Domenech, T., Bleischwitz, R., Melin, H. E. & Heidrich, O. Nature Sustain. 4, 71–79 (2021).
Li, W., Lee, S. & Manthiram, A. Adv. Mater. 32, 2002718 (2020).
Yang, J. H., Kim, H. & Ceder, G. Molecules 26, 3173 (2021).
Melin, H. E. et al. Science 373, 384–387 (2021).
Lei, C. et al. Green Chem. 23, 4710–4715 (2021).
International Energy Agency. Net Zero by 2050: A Roadmap for the Global Energy Sector (IEA, 2021); available at https://www.iea.org/reports/net-zero-by-2050

 Lithium-ion batteries need to be greener and more ethical
Lithium-ion batteries need to be greener and more ethical
 Recycling lithium-ion batteries from electric vehicles
Recycling lithium-ion batteries from electric vehicles