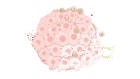
Biopharmaceutical firm CureVac’s RNA printer can rapidly supply mRNA vaccine candidates. Credit: CureVac
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 589, 630-632 (2021)
doi: https://doi.org/10.1038/d41586-021-00191-z
These interviews have been edited for length and clarity.
Updates & Corrections
-
Correction 11 March 2021: An earlier version of this feature erroneously stated that GenEPi was developed at Imperial College London.
References
Zeng, C., Zhang, C., Walker, P. G. & Dong, Y. ‘Formulation and Delivery Technologies for mRNA Vaccines’ in Current Topics in Microbiology and Immunology. https://doi.org/10.1007/82_2020_217 (Springer, 2020).
Papagiakoumou, E., Ronzitti, E. & Emiliani, V. Nature Methods 17, 571–581 (2020).
Chenoweth, A. M., Wines, B. D., Anania, J. C. & Hogarth, P. M. Immunol. Cell Biol. 98, 287–304 (2020).
Chauhan, J. et al. Antibodies 9, 55 (2020).
Collombet, S. et al. Nature 580, 142–146 (2020).
Kaya-Okur, H. S. et al. Nature Commun. 10, 1930 (2019).
Ma, S. et al. Cell 183, 1103–1116 (2020).
Yaganoglu, S. et al. Preprint at bioRxiv https://doi.org/10.1101/702423 (2019).
Nakamura, H. et al. Preprint at bioRxiv https://doi.org/10.1101/2020.03.30.016360 (2020).
Kompauer, M., Heiles, S. & Spengler, B. Nature Methods 14, 90–96 (2017).
Cassat, J. E. et al. Sci. Transl. Med. 10, eaan6361 (2018).
Geier, B. et al. Nature Microbiol. 5, 498–510 (2020).
Zhang, J. et al. Sci. Transl. Med. 9, eaan3968 (2017).
Zhang, J. et al. Preprint at medRxiv https://doi.org/10.1101/2020.12.14.20248101 (2020).
Jeong, S.-Y., Kim, J.-S. & Lee, J.-H. Adv. Mater. 32, 2002075 (2020).

 Technologies to watch in 2020
Technologies to watch in 2020
 The secret forces that squeeze and pull life into shape
The secret forces that squeeze and pull life into shape
 The lightning-fast quest for COVID vaccines — and what it means for other diseases
The lightning-fast quest for COVID vaccines — and what it means for other diseases
 NatureTech hub
NatureTech hub