- NEWS
Reversal of biological clock restores vision in old mice
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 588, 209 (2020)
doi: https://doi.org/10.1038/d41586-020-03403-0
Read the related News & Views, ‘Sight restored by turning back the epigenetic clock’.
References
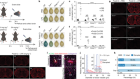
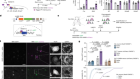
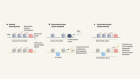
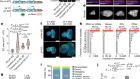
Lu, Y. et al. Nature 588, 124–129 (2020).
Ocampo, A. et al. Cell 167, 1719–1733 (2016).

 First hint that body’s ‘biological age’ can be reversed
First hint that body’s ‘biological age’ can be reversed
 How anti-ageing drugs could boost COVID vaccines in older people
How anti-ageing drugs could boost COVID vaccines in older people
 Biomarkers and ageing: The clock-watcher
Biomarkers and ageing: The clock-watcher
 Can epigenetics help verify the age claims of refugees?
Can epigenetics help verify the age claims of refugees?