- NEWS FEATURE
The lasting misery of coronavirus long-haulers
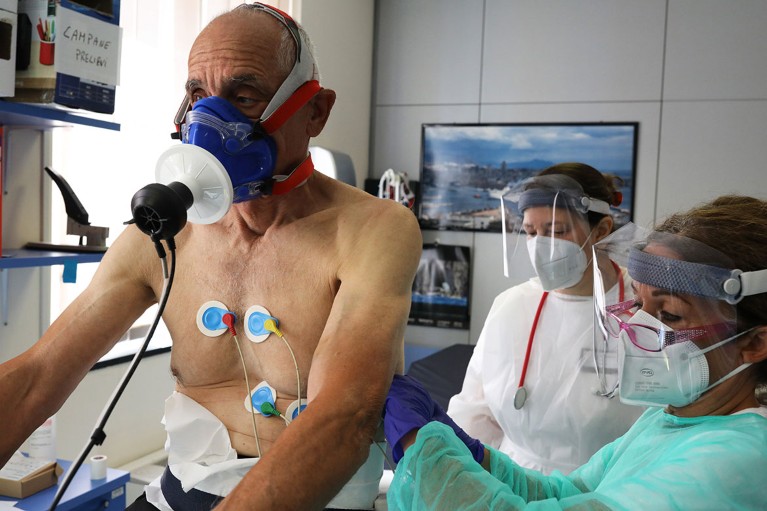
A person who has recovered from COVID-19 takes part in a rehabilitation programme in Genoa, Italy. Credit: Marco Di Lauro/Getty
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 585, 339-341 (2020)
doi: https://doi.org/10.1038/d41586-020-02598-6
References
Salehi, S., Abedi, A., Balakrishnan. S. & Gholamrezanezhad, A. Am. J. Roentgenol. 215, 87–93 (2020).
Weerahandi, H. et al. Preprint at medRxiv https://doi.org/10.1101/2020.08.11.20172742 (2020).
Zhang, P. et al. Bone Res. 8, 8 (2020).
Hu, Y. et al. J. Virol. 91, e02143-16 (2017).
Varga, Z. et al. Lancet 395, 1417–1418 (2020).
Xiong, T.-Y., Redwood, S., Prendergast, B. & Chen, M. Eur. Heart J. 41, 1798–1800 (2020).
Carfi, A. et al. J. Am. Med. Assoc. 324, 603–605 (2020).
Zhao, Y.-M. et al. EClinicalMedicine 25, 100463 (2020).
The Prevail III study group. N. Engl. J. Med. 380, 924–934 (2019).
Moldofsky, H. & Patcai, J. BMC Neurol. 11, 37 (2011).
Lam, M. H.-B. et al. Arch. Intern. Med. 169, 2142–2147 (2009).
Gerwyn, M. & Maes, M. Curr. Rheumatol. Rep. 19, 1 (2017).
Lyons, D. et al. Ir. J. Psychol. Med. https://doi.org/10.1017/ipm.2020.40 (2020).

 How COVID-19 can damage the brain
How COVID-19 can damage the brain
 Profile of a killer: the complex biology powering the coronavirus pandemic
Profile of a killer: the complex biology powering the coronavirus pandemic
 Thousands of people will help scientists to track the long-term health effects of the coronavirus crisis
Thousands of people will help scientists to track the long-term health effects of the coronavirus crisis
 A negative COVID-19 test does not mean recovery
A negative COVID-19 test does not mean recovery