Host: Shamini Bundell
Welcome back to the Nature Podcast. This week, growing medicine with yeast…
Host: Nick Howe
And the mystery mechanism of general anaesthetics. I’m Nick Howe.
Host: Shamini Bundell
And I’m Shamini Bundell.
[Jingle]
Host: Shamini Bundell
First up, reporter Adam Levy is here to tell us about the use of baker’s yeast to make an important class of medicines.
Interviewer: Adam Levy
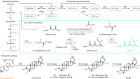
You may never have heard of tropane alkaloids, but they are classed as essential medicines by the World Health Organization, for good reason since they are used in the treatment of a wide range of conditions, as synthetic biologist Christina Smolke explains.
Interviewee: Christina Smolke
Currently, there’s a number of tropane alkaloids that are used to treat neuromuscular disorders. So, these are things when you have muscle spasms, so they’re used to treat vomiting and nausea. They’re also used to treat Parkinson’s disease and the symptoms associated with that. So, really, a wide range of different types of medicines are derived from these compounds.
Interviewer: Adam Levy
Christina is interested in new ways of creating old compounds. She previously found methods to produce another category of essential drugs – opioids – using yeast. And this week in Nature, she’s shown a similar technique for these tropane alkaloids. I called her up and started by asking her how these compounds are produced today.
Interviewee: Christina Smolke
So, because these were discovered in plants and they are of a type of compound where their structure is sufficiently complex that we cannot efficiently make them with synthetic chemistry, what we do in this case is that we continue to rely on the plant to make these compounds, and so what that means in this case is that we make these compounds by farming.
Interviewer: Adam Levy
Now, using farming as the approach to get hold of these drugs can be a fragile process at the best of times, but that’s really been highlighted in 2020, right?
Interviewee: Christina Smolke
That’s right. One set of categories is environmental events, right? So, over the past year or two, we’ve seen wildfires devastate entire regions. That can wipe out entire crops and so change the yield and the quality that we’re able to get from those crops. The second category then, of course, is what we’re seeing with COVID and these types of global crises, and when you have these relatively sudden and large spikes in demand, again, agriculture-based supply chain, it’s very difficult if not impossible to be able to respond to those because essentially you would have to basically plant more of the crop, and so it’s really a supply chain that doesn’t have the ability to rapidly respond.
Interviewer: Adam Levy
Now, given that we can’t synthesise molecules like this in the lab yet, what did you set out to do differently to growing them in a field?
Interviewee: Christina Smolke
What we’ve been interested in doing is basically developing different ways of being able to synthesise them in the lab, but ones that aren’t founded in chemistry but are founded in biology. And so, the idea here, though, is to shift from utilising plants to platform organisms, and so, for us, that platform organism is brewer’s yeast. So, brewer’s yeast is an organism that we as humans have centuries working with basically and utilising for our purposes, and so the idea then is that what we want to be able to do is basically change that yeast so that instead of making the typical products of fermentation that we like to use, which are ethanol and carbon dioxide, that we’re instead changing it so the products are these high-value active ingredients that typically would only be made in plants.
Interviewer: Adam Levy
You carried out a similar process with opioids, producing certain opioid compounds with yeast. How different is the approach that you’re employing here?
Interviewee: Christina Smolke
Certainly, this work here leverages many of the approaches and techniques that we’ve been developing in my laboratory over the past decade, but it takes it a step further. So, plants are nature’s best chemist, basically, and the types of molecules they make are incredibly interesting and also incredibly valuable. It will specialise cells and tissues for certain chemistries and biosyntheses to occur, and so in this paper, what we did is we actually wound up utilising all the organelles in the cell where we’re distributing basically different chemistries across the cell. We sort of draw an analogy to a factory, right, where you have different processes taking place across the factory floor. That’s how we’re beginning to re-envision the yeast cell.
Interviewer: Adam Levy
What stage are you currently at? I mean, presumably, we’re not going to be able to buy tropane alkaloid-based drugs which are produced by yeast in the shops anytime soon.
Interviewee: Christina Smolke
Not immediately, right, but as the first sort of proof of concept demonstration, what the challenge is that ultimately, how much of these molecules the yeast are making are low. But what we’ve seen is that while it’s not ready immediately, right, to be making drugs at scale, with a dedicated team and with the right investment, it can be ready to scale within one to two years.
Interviewer: Adam Levy
Just how big an advantage would being able to do this in yeast commercially be providing compared to the conventional way of extracting from plants?
Interviewee: Christina Smolke
My own belief is that it’s a huge advantage. This type of approach really gives us a way to make these compounds on demand when and where needed. There’s huge implications there. But the other side of this, beyond just supply chain and manufacturing, is drug discovery, right, which is to say that once you have these pathways reconstructed in yeast, we can begin to manipulate the pathways, right, and begin to make changes and modifications, and that really, I think, will have huge impacts for drug discovery.
Host: Shamini Bundell
That was Christine Smolke. For more on those microbial factories, make sure you check out her paper. We’ll put a link in the show notes.
Host: Nick Howe
Next up, it’s time for our weekly update on coronavirus with Coronapod.
Noah Baker
Hello, yes, and welcome to the Coronapod section of the show. A few things to say before we get going. Firstly, Ben and Amy are not here this week, so it’s just me, Noah Baker. I will try to do all I can. In other news, you might hear a slight difference this week in the recording. The reason is that I’m no longer in my sort of home-built duvet fort comforter studio. I’m actually on my narrowboat in London, and I’m moored next to a skate park, so I’m really sorry if there’s horrible noises. There’s’ not much I can do about it at this moment in time. And there’s a new voice, and that’s Heidi Ledford. Heidi, who are you? Let people know what it is that you do at Nature.
Heidi Ledford
I think the first thing to note is that I am nowhere near as glamorous as Noah, living on a boat in the canals in London. I am a reporter for Nature in London. Once upon a time, I used to cover things like cancer biology and drug development and sort of biomedical topics, but for the past few months, it has been mostly coronavirus and COVID.
Noah Baker
This week we’re going to talk about immunity. So, this is a really big topic and it’s a really central topic to the pandemic in general, and it sounds like it might be a simple thing to talk about but it’s actually not simple at all. I thought maybe the first thing to ask you to do, Heidi, is to give us a bit of an overview. So, immunity isn’t one thing. What do we mean when we say immunity?
Heidi Ledford
Yeah, it’s true. We can mean a lot of different things. So, the immune system is very complex, and there was a time when I would have said it’s beautiful, I love how beautifully complex it is and it’s amazing and versatile and can respond to so many different challenges. Nowadays, when we want really simple answers to important questions and we’re kind of all counting on our immune system to get us out of this pandemic, right, so now, it’s sort of miserably complex, and it’s hard for us to really get a clear read on how things are going to shake out long-term. But yes, so, the immune system has a number of different ways of responding to viruses, and we’ve got an amazing amount of data already, particularly about antibodies, which are both relatively easy to study, but also very important in immune responses. But then you also have specialised cells like T cells that are able to, in some cases, recognise the cells in your body that are infected by a virus and then destroy those cells, thereby interrupting the viral replication process. We also have memory B cells which kind of linger around, they’re a bit sleepy. They wait until you’re infected by a virus that you’ve seen before, and they jump into action and kick of the process that starts producing antibodies against that virus again so that you can respond to that virus more rapidly than you did the first time around that you encountered it. So, there are lots of different levels to the immune system. The key problem that I think we’re facing right now, when all of us really want clear answers, is that we don’t have a good marker or even a set of markers that we can look at and say, ‘Oh, okay, antibodies are doing this, T cells are doing that, therefore our long-term immunity to this virus is going to look like this.’ We don’t really have a way of doing that.
Noah Baker
Yeah, absolutely, and I guess these are all really fundamental questions to other things that we’ve talked a lot about on Coronapod, so if you think about a vaccine and you have something that might give you some kind of protection, if you don’t really have a sense of what protection looks like or how to measure it yet, it throws the whole sort of concept of a vaccine up in the air.
Heidi Ledford
Yeah, I mean there are a lot of worries about how effective is a vaccine going to be. We’re all sort of hitching our hopes to the vaccine, right, and when the vaccine comes out, we can all re-emerge from our houses and protect our loved ones and so on, but we don’t know yet how effective that vaccine is going to be. Will one shot work at all? Will we need booster shots, which is not the end of the world, right? We have booster shots for lots of our vaccines. But when you’re thinking about vaccinating a globe full of 7 billion people and then now you’ve got to add in a booster shot or two or three or whatever, then that does complicate things. We also don’t know will a vaccine be able to make us completely immune so that the virus won’t even be able to replicate in our bodies anymore, right, that we won’t be infected at all, and that’s something called sterilising immunity, or maybe the best we can hope for is some situation where we would still get infected a second time, perhaps, but maybe our symptoms won’t be as severe or even show up at all.
Noah Baker
Yeah, I think when a lot of people hear ‘immunity’, what they’ll imagine is that sterilising immunity, that first thing that you talked about, that kind of barrier that says you cannot get this disease, you cannot be infected anymore, but actually, in reality, often what people are talking about when they say immunity is just your immune system is strengthened enough that you can cope with that infection in a way that it’s not going to be too damaging to your health.
Heidi Ledford
And the situation we’re in now, I mean, better that than nothing, right, in a way? But it is true that long-term, that could still pose a problem, particularly for the elderly and the vulnerable populations. If we are walking around not realising that we’re infected and not with that presence of mind maybe still to wear a mask and have the hand sanitiser and so on and so forth, that could still pose a risk for those populations.
Noah Baker
So, if scientists want to find out more about immunity to COVID, what’s in the toolbox of a researcher to better understand immunity? How do you study this?
Heidi Ledford
Yeah, they’ve done a lot of really good work and amazingly fast work so far, looking at things like antibodies, T cells and B cells. A lot of what we really need, I guess, is just data from what happens in the real world, right. I mean, we will get some really good data, potentially, from vaccines and vaccine trials because they’ll be looking at whether or not these vaccines can stimulate an immune response, and they may also give us some idea. If a vaccine doesn’t perform particularly well, for example, we may be able to use that data to figure out why did some patients do well with this vaccine but it didn’t work for other participants in this study, right, so that could give us some good clues going forward. The other thing which has not been great news, I guess, over the past week or so is that we’ve had several pretty well documented cases now of reinfection. I mean, we’ve have anecdotes all along, but now we’re getting some cases where people have actually looked and been able to sequence the virus both during the initial infection and during the second infection to show that they’re distinct variants and distinct viruses and you actually had two infection events, and people do think that we’re going to see more of those cases in the future. As we see more of those cases, we may begin to learn a bit more about why some people are becoming re-infected. Are they showing symptoms that are more severe or less severe than they did the first time around? Some of the key questions that we want to have answered may actually kind of come from this what we would consider to be bad news, I guess, from these stories of re-infection.
Noah Baker
Right, and I guess these re-infection stories, you can sort of spin them different ways depending on what you want to know. It’s information that’s helpful for the future but also, it’s maybe worrying that people don’t have long-term immunity after being infected, but there’s also even more things that confuse it, right? So, if someone had maybe a mild infection, they might not have had as large an immune response and so they may be less likely to develop a longer-term immunity. That’s the same reason that people argue that people don’t really become immune to colds, because they don’t trigger a big enough infection. Should be worried at this point? In your judgement, is there enough positive evidence that things are going to be alright?
Heidi Ledford
Well, I think we’re in the middle of a really terrible situation, so I think we can’t help but be worried, and I think sometimes, some of the bad news carries a lot of weight maybe in our minds because it feels apocryphal sometimes. But I think the main thing to keep in mind is that all of this data is preliminary, right? I mean, these re-infection cases, there were reports of maybe four last week. Two of them I’ve seen preprints for. In one of those cases, the person, on second infection, had fewer symptoms, and then the other case, the other person had more severe symptoms, and just what can you do with that information because it’s n=1 in each category. You just don’t know. There’s so many things that could be confounding that. I think we may also have lost sight of some of the good news that came out early on and we’ve just kind of gotten used to it. The fact that we can mount what look to be effective antibody responses against SARS-CoV-2 at all is great news because we can’t do that against every virus, right? HIV, we don’t do a good job with that. I think if anything, well, there are many lessons we should have learnt from the pandemic, but I think one of them really has to be that we have to be careful about reading too much into anecdotes. If anecdotes were the gospel truth then hydroxychloroquine would have saved us all months ago, so sometimes we do just have to sit and wait. But I can say, like on a personal level, my daughter was quite sick back in April with something that seemed like COVID. You couldn’t get tested back then, right, it was very difficult to get tested in London at the time, so we don’t know for sure. But I catch myself, now, on those beautiful occasions when we leave the house, when I think, ‘She’s fine, she’s had it already,’ and then I have to honestly take that thought and step back and think, ‘No, no, no, wait. I mean, a) we don’t know for certain she had it but, b) we don’t know for certain she would be immune even if she did, and then c) even if she seemed to be immune, maybe she would still become mildly infected and shed it and carry the virus around to other members of the community.’ So, I do, I have this conversation with myself constantly. She wears the mask and she puts on the hand sanitiser and does everything just like we all do because we don’t know.
Noah Baker
Okay, well it seems like there’s going to be an awful lot to keep an eye on. Heidi, thank you so much for joining us and telling us all there is to know about immunity.
Heidi Ledford
I have to laugh at the idea of telling you all there is to know about immunity because it is so complicated, but thank you so much for having me. It’s always good fun.
Host: Shamini Bundell
Noah and Heidi there. And we’ll have more on the ongoing pandemic next week. Coming up, we’ll be learning about research uncovering how anaesthetics work. Before that, though, Dan Fox is here with this week’s Research Highlights.
[Jingle]
Dan Fox
If you’re anything like me, then there have been times when a plan to have a small scoop of ice cream for dessert has ended with you clearing out a whole tub. Now, scientists have found the circuit in the brain that could explain why it’s so easy to overindulge, even after a hearty meal. Working in mice, the researchers identified a region of the brainstem that is home to a set of neurons whose activity is influenced by food and water intake. They found that activating these neurons inhibited eating, but blocking them caused mice to eat or drink more and for longer, even when the animals were well fed and hydrated. The team served the mice a variety of liquids while monitoring the neurons’ activity, and found that the activity was reduced when the mice drank water or bitter-tasting compounds. However, this production in neuronal activity was even more noticeable when the mice consumed tasty drinks like a vanilla-flavoured beverage. This suggests a feedback loop that allows consumption of tasty food and drink to trick the brain into wanting more. Indulge in the full buffet of that research over at Cell.
[Jingle]
Dan Fox
Certain bacteria have been found to have a special skill – a hidden talent for electrical wiring. A team of researchers investigating the electrically conductive bacteria Geobacter sulfurreducens found that the microbes flourished when grown on a copper electrode, forming tough layers known as biofilms. What’s more, these biofilms produced double the electrical current of those grown on a graphite electrode. Chemical analysis revealed copper sulphite solids deposited throughout the films. The team concluded that the bacteria promote chemical reactions between the copper electrode and sulphate ions in their food source to form sulphide wires, which enhance the flow of electricity within the biofilm. The researchers hope that their finding can help to improve the design of fuel cells that could take advantage of such electrically conductive bacteria. Plug into that research at Energy & Environmental Science.
[Jingle]
Interviewer: Nick Howe
Next up on the show, I’ve been looking into general anaesthetics. Now, if you’re about to go into surgery or something, you might want to skip this section because it might not comfort you to know that despite being used for over 100 years, there’s a lot we don’t know about anaesthetics.
Interviewee: Ryan Hibbs
I think it’s fair to say we do not have a rock-solid understanding of how general anaesthetics work.
Interviewer: Nick Howe
This is Ryan Hibbs, a biophysicist who’s interested in neurological questions, like that of how anaesthetics do indeed function. This week in Nature, he’s published a paper looking at the detailed mechanisms of how anaesthetics bind to a certain receptor in the brain. I called him up to find out more, and asked, ‘What do we know about how anaesthetics work?’
Interviewee: Ryan Hibbs
It helps to step back just a little bit to understand that the central nervous system – let’s focus on the brain in particular – works as a balance of excitatory and inhibitory or positive and negative signalling. If you have too much positive signalling, too much excitation, you get convulsive disorders like epilepsy. If you have too much inhibitory signalling, too much inhibition, too much negative signalling, then you go to sleep, eventually stop breathing, you go into a coma and you die. So, much like when you’re running the water for a bath, you need this balance of hot and cold signalling, excitation and inhibition, for things to work well. So, general anaesthetics calm down signalling in the brain. They dampen the signalling in the brain, and they do that by inhibiting the excitation and stimulating the inhibition signalling. So, some of the molecular targets for general anaesthetics include this receptor, this GABAA receptor we’re working on. Several general anaesthetics act through making it easier for that receptor to work.
Interviewer: Nick Howe
And so, what were you trying to understand with this paper? What were your ideas going in to it?
Interviewee: Ryan Hibbs
Other groups over the past several decades have identified specific GABAA receptors and components of them to be targets for some general anaesthetics – many of them – and we wanted to go to the next level of detail and look at exactly how some general anaesthetics bind to these GABAA receptors to visualise exactly where they are nestling into their binding sites in these receptors, hoping that by doing that we would gain some additional understanding into how they do what they do.
Interviewer: Nick Howe
And so, what you did is you looked at this receptor and how general anaesthetics were binding using electron microscopy, but my understanding is that you didn’t just look at these anaesthetics, you also looked at a few other different compounds and how they were binding to the GABAA receptor too. Why were you interested in those ones as well?
Interviewee: Ryan Hibbs
That’s right. So, this GABAA receptor is the target of several different important drug classes, some that are very useful clinically as well as some abused drugs, and those include benzodiazepines, those include Valium and Xanax. They act through potentiating this GABAA receptor, similar to what general anaesthetics do to it, and we wanted to compare how general anaesthetics work on this receptor with benzodiazepine drugs. And so, we also obtained structural information for the receptor bound to a classical benzodiazepine, diazepam, and an inhibitor of this benzodiazepine site called flumazenil. So, we collected structural information for this receptor with these different drug classes bound to understand what mechanisms they might share and what mechanisms might be distinctive to better understand how they work and how the receptor works.
Interviewer: Nick Howe
And so, you had these different pictures, essentially, of how these different compounds were binding to the receptor. What were the differences? What were the similarities into how these things were binding?
Interviewee: Ryan Hibbs
So, it got quite complicated, more complicated than we expected. So, it turns out that the benzodiazepine, diazepam, the one that’s extremely popular for anxiety, Valium, binds to four different places on this receptor. One of them is the classical benzodiazepine site, and that sites overlaps perfectly with where flumazenil binds. So, flumazenil, it’s easier to understand how it would directly compete with the interaction of diazepam with the receptor at this one site. That site has been well characterised and is called the benzodiazepine site. We were interested to see that the classical benzodiazepine, diazepam, also bound in three different locations in another region of the protein, the part of this protein that is buried in the cell membrane, and these are sites that overlap with where general anaesthetics bind and, in fact, would compete with some of the general anaesthetics we looked at. So, it is known that, at low concentrations, benzodiazepines like the one we looked at help with anxiety, but at high concentrations or higher concentrations, they can be used to directly induce anaesthesia, so it may be through binding to these additional sites in the transmembrane domain of the receptor that diazepam is able to directly induce anaesthesia.
Interviewer: Nick Howe
What might that mean then? What might be the implications of these results if you see how these different things can bind?
Interviewee: Ryan Hibbs
General anaesthetics do have side effects, and propofol, while it’s extremely popular, for example, cannot be given in cases in elderly patients, especially where cardiovascular issues are a problem, and so there are side effects of these drugs that we don’t really understand, and one of the reasons we get these side effects is because general anaesthetics like propofol or volatile general anaesthetics like isoflurane or sevoflurane that are extremely useful in the clinic, they bind to all different kinds of proteins. If we can make these drugs more selective then we could target them specifically to have tailored effects in the nervous system and maybe get the desirable components of anaesthesia without the undesirable respiratory depression, something like that. And so, our hope is that by understanding in this exquisite level of detail how these drugs interact with their receptor targets, we can add a methyl group here or put an oxygen there or a nitrogen there and tailor them to bind to these receptors more specifically and have fewer off-target effects. That’s the dream.
Interviewer: Nick Howe
So, now, if I was to ask you after this research, how do general anaesthetics work, do you think you would have an answer for me now?
Interviewee: Ryan Hibbs
I’d say we know how some of them work through this receptor at a very fine level of detail with a lot of confidence, but there are definitely more mysteries to resolve related to the mechanisms of general anaesthetics.
Host: Nick Howe
That was Ryan Hibbs from the University of Texas Southwestern Medical Center. For more details about his research, you can find a link to his paper in the show notes.
Host: Shamini Bundell
Finally on the show, it’s time for the weekly Briefing chat. Now, the Nature Briefing is in fact having a brief summer break but, Nick, I know you’ve been digging around for some fun science stories we can chat about. What have you found?
Host: Nick Howe
Well, this is a story that I don’t think you could have missed if you’ve been on any kind of science or science adjacent social media, and this is about the company Neuralink has put a chip in a pig’s brain.
Host: Shamini Bundell
I really have missed this. I’m familiar with the headline but I don’t know the story at all. Robot-controlled pigs – what’s happening here?
Host: Nick Howe
Let’s go back a couple of steps. So, Neuralink is a company that was founded a few years ago by Elon Musk, who you may know as the founder of SpaceX and Tesla, and basically, the idea is to make a sort of human-computer interface, so like a chip that can be implanted into your brain and then it can do all sorts of things, augment you or interface with computers and stuff like that. These are the things they’re sort of claiming. And so, there was an update this past week where they rolled out a pig that actually had one of these chips implanted into its brain.
Host: Shamini Bundell
Like a live pig with a pig-computer interface?
Host: Nick Howe
Yeah, basically. It was a little coin-shaped object with really thin, floppy wires coming out of it that was implanted into the pig’s brain and you could see, during this live broadcast, you could see basically the neurons firing as the pig snuffled about. The pig was called Gertrude. That’s not relevant but it’s just cute.
Host: Shamini Bundell
But Gertrude wasn’t there sort of controlling any computers or having super X-ray vision?
Host: Nick Howe
No, it wasn’t doing anything like that. This is quite an early prototype from what can be gathered about it, and so some newer scientists have looked into this. There was a white paper that also came out at the same time and they said that it’s really interesting technology and this is quite interesting and quite impressive, as well, to see sort of these live broadcasts of different neurons firing as the pig sort of snuffled about, but it’s a long way away from what some of the things that the company are claiming that it will be able to do in the future, such as improve people’s vision or store memories and, well, interface with computers and things like that. That seems to be a long way off. But the actual chip itself seems to be a quite intriguing bit of technology.
Host: Shamini Bundell
I guess I can see how a chip in your brain could detect electrical impulses going on in your head, but all this other stuff about whether we’re sort of controlling computers with our mind or whether the computer is somehow augmenting you, like, that’s really far off from just a chip reading electrical signals. How would that even work?
Host: Nick Howe
Well, it’s not outright impossible but it may just be a long way off, and they sort of haven’t really given a clear timeline on when different things like this are going to be available. But the idea is that each of the different little wires will be able to interface with neurons and they’ll be able to cause neurons to fire to do certain things and they’ll also be able to receive impulses from neurons so you can control things with your brain, essentially. So, it is those sorts of things that the company are claiming, that one day maybe you’ll be able to limit pain or maybe even telepathy, but one thing that does seem like it is a possibility is they talked about helping people with spinal injuries and people who have nerve damage and stuff like that, and that is something that may well be possible by stimulating certain parts of the brain by using a chip like this.
Host: Shamini Bundell
So, yes, this isn’t just super sci-fi video game super-tech. Useful medical applications – that’s good to know. One day.
Host: Nick Howe
Yeah, one day. And the other thing will be that people have to see the sort of long-term effects of this and how this will work in humans too. There could be all sorts of things going on with implanting something into someone’s brain. But can you implant something into my brain, Shamini? What is your story this week?
Host: Shamini Bundell
That was so bad. Nick, that was a terrible segue. So, yeah, I’m cheating this week, actually. I haven’t gone up and dug out an exciting news story. I’m going to tell you something that I’ve been working on for video a for our YouTube channel. Maybe it’s got to be seen to be believed – I don’t know – but let me describe it to you and see if you believe it, I guess. So, here is the image that I need you to get into your head. Right, you have a glass container a little bit taller and wider than your hand, say. It’s filled with a sort of blue-ish liquid, but in this container, a layer of this liquid, about an inch-deep, is levitating. So, it fills the container to the side, above it is some air, below it is some more air, and it seems to be floating of its own accord. That’s weird thing number one. Weird thing number two – my favourite part – is the tiny, cute plastic boat that the researchers have decided to use to demonstrate this phenomenon that they’ve been working on, and this tiny, cute plastic boat is floating upside down at the bottom of this levitating liquid layer. So, the hull of the boat is in the liquid but the sail is poking down into the air underneath the layer of liquid. So, it looks like it’s defying gravity and just sailing along, floating there upside down.
Host: Nick Howe
Right, okay, so there’s a boat floating upside down and liquid levitating. Can you… I mean, what is going on here?
Host: Shamini Bundell
It’s science, funnily enough. I love this story because you just look at it and you’re like, ‘There is no possible, sensible explanation for this.’ It’s just ridiculous and counter-intuitive, but I’ll start off with the levitating liquid, which is not a particularly new discovery, so this this was known to happen, and the way that you do it is with vertical vibrations. So, basically, this glass container that I talked about is placed on a shaker, so it’s just shaking up and down quite high. You can hear it buzzing away in the video, just shake, shake, shake, shake. And it’s that shaking which stops the liquid from falling. There’s a particular effect which means that liquid tends to fall in drips, right? Now, the shaking prevents the dripping, and if the bottom surface can’t form these little drips, it can’t fall and therefore the whole thing ends up with this weird sort of suspension effect.
Host: Nick Howe
Right, okay, so that’s something that we did know that happens. I’m still struggling to get my head around it but physics says that happens – sure. Where does the boat come in?
Host: Shamini Bundell
So, the boat is the sort of new part of the science where these researchers in Paris were looking at this shaking levitation effect, and they discovered that if you put not necessarily a boat but sort of like light plastic object that would usually float, it would also float on the bottom surface of this levitating liquid. This was quite hard from them to set up and get in position. The little plastic boats had a tendency to either fall to the bottom or bob up to the top, but if you get it in the kind of right area, it will just float there in the kind of right area, it will just float there in a kind of… they described it as ‘reverse buoyancy’.
Host: Nick Howe
Yeah, I mean, I guess that’s what it is. It’s still buoyant, it’s still floating, but it’s just upside down. Now, is this just my human brain resisting this? Does this make a lot of sense mathematically or with the laws of physics? I’m just trying to work out how it is that the boat is floating there upside down.
Host: Shamini Bundell
Well, my human brain looking at it has decided it definitely doesn’t make sense, but I have strong reassurances from world experts that if you go through the maths, the energy sort of inputted by this shaking effect changes the forces that are acting on this boat and causes this phenomenon. But I would recommend going and checking out this video which has just come out – there’ll be a link in the show notes – and seeing for yourself that it really does work. It does, if you read the paper, make some kind of sense.
Host: Nick Howe
Well, I think I’m going to have to see that video to quite fully believe this, but thanks for chatting to me, Shamini. And listeners, for more on those stories, we’ll put some link in the show notes, and make sure you check out the Nature Briefing as well which will resume shortly. We’ll put a link to that too.
Host: Shamini Bundell
And if the Nature Podcast floats your boat, why not tell us about it? We love hearing your feedback. You can find us on Twitter – we’re @NaturePodcast – or drop us an email – we’re podcast@nature.com. I’m Shamini Bundell.
Host: Nick Howe
And I’m Nick Howe. Thanks for listening.

 Read the paper: Biosynthesis of medicinal tropane alkaloids in yeast
Read the paper: Biosynthesis of medicinal tropane alkaloids in yeast
 What the immune response to the coronavirus says about the prospects for a vaccine
What the immune response to the coronavirus says about the prospects for a vaccine