- NEWS AND VIEWS
Stretch exercises for stem cells expand the skin
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 584, 196-198 (2020)
doi: https://doi.org/10.1038/d41586-020-02158-y
References
Neumann, C. G. Plast. Reconstruct. Surg. 19, 124–130 (1957).
Zöllner, A. M., Holland, M. A., Honda, K. S., Gosain, A. K. & Kuhl, E. J. Mech. Behav. Biomed. Mater. 28, 495–509 (2013).
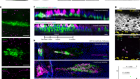
Aragona, M. et al. Nature 584, 268–273 (2020).
Ladoux, B. & Mège, R.-M. Nature Rev. Mol. Cell Biol. 18, 743–757 (2017).
Chug, P. & Paluch, E. K. J. Cell Sci. 131, jcs186254 (2018).
Panciera, T., Azzolin, L., Cordenonsi, M. & Piccolo, S. Nature Rev. Mol. Cell Biol. 18, 758–770 (2017).
Hirata, H. et al. EMBO Rep. 16, 250–257 (2015).
Rübsam, M. et al. Nature Commun. 8, 1250 (2017).
Mesa, K. R. et al. Cell Stem Cell 23, 677–686 (2018).
Nava, M. M. et al. Cell 181, 800–817 (2020).
Piedrafita, G. et al. Nature Commun. 11, 1429 (2020).

 Read the paper: Mechanisms of stretch-mediated skin expansion at single-cell resolution
Read the paper: Mechanisms of stretch-mediated skin expansion at single-cell resolution
 A gut feeling for cellular fate
A gut feeling for cellular fate
 Regenerative medicine could pave the way to treating baldness
Regenerative medicine could pave the way to treating baldness