- NEWS AND VIEWS
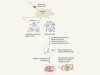
Brain–spleen connection aids antibody production
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 581, 142-143 (2020)
doi: https://doi.org/10.1038/d41586-020-01168-0
References
Mebius, R. E. & Kraal, G. Nature Rev. Immunol. 5, 606–616 (2005).
Zhang. X. et al. Nature 581, 204–208 (2020).
Nutt, S. L., Hodgkin, P. D., Tarlinton D. M. & Corcoran, L. M. Nature Rev. Immunol. 15, 160–171 (2015).
Jung, W. C., Levesque, J. P. & Ruitenberg, M. J. Semin. Cell Dev. Biol. 61, 60–70 (2017).
Ben-Shaanan, T. L. et al. Nature Med. 22, 940–944 (2016).
Rosas-Ballina, M. et al. Science 334, 98–101 (2011).
Davis, M. Annu. Rev. Neurosci. 15, 353–375 (1992).
Smith, S. M. & Vale, W. W. Dialogues Clin. Neurosci. 8, 383–395 (2006).
Peng, J. et al. Front. Neuroanat. 11, 63 (2017).
Coutinho, A. E. & Chapman, K. E. Mol. Cell. Endocrinol. 335, 2–13 (2011).
Cathomas, F., Murrough, J. W., Nestler, E. J., Han, M. & Russo, S. J. Biol. Psychiat. 86, 410–420 (2019).

 Read the paper: Brain control of humoral immune responses amenable to behavioural modulation
Read the paper: Brain control of humoral immune responses amenable to behavioural modulation
 Trispecific antibodies offer a third way forward for anticancer immunotherapy
Trispecific antibodies offer a third way forward for anticancer immunotherapy
 The advent and rise of monoclonal antibodies
The advent and rise of monoclonal antibodies