- NEWS AND VIEWS
New antibiotics target the outer membrane of bacteria
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 576, 389-390 (2019)
doi: https://doi.org/10.1038/d41586-019-03730-x
References
World Health Organization. Global Priority List of Antibiotic-Resistant Bacteria to Guide Research, Discovery, and Development of New Antibiotics (WHO, 2017).
Nikaido, H. Microbiol. Mol. Biol. Rev. 67, 593–656 (2003).
Li, X.-Z. & Nikaido, H. Drugs 64, 159–204 (2004).
Munita, J. M. & Arias, C. A. Microbiol. Spectr. 4, VMBF-0016-2015 (2016).
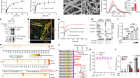
Imai, Y. et al. Nature 576, 459–464 (2019).
Luther, A. et al. Nature 576, 452–458 (2019).
Hart, E. M. et al. Proc. Natl Acad. Sci. USA 116, 21748–21757 (2019).
Konovalova, A., Kahne, D. E. & Silhavy, T. J. Annu. Rev. Microbiol. 71, 539–556 (2017).
Storek, K. M. et al. Proc. Natl Acad. Sci. USA 115, 3692–3697 (2018).
Srinivas, N. et al. Science 327, 1010–1013 (2010).
Mares, J., Kumaran, S., Gobbo, M. & Zerbe, O. J. Biol. Chem. 284, 11498–11506 (2009).
Li, J. et al. Lancet Infect. Dis. 6, 589–601 (2006).

 Read the paper: Chimeric peptidomimetic antibiotics against Gram-negative bacteria
Read the paper: Chimeric peptidomimetic antibiotics against Gram-negative bacteria
 Read the paper: A new antibiotic selectively kills Gram-negative pathogens
Read the paper: A new antibiotic selectively kills Gram-negative pathogens
 Fighting evolution with chemical synthesis
Fighting evolution with chemical synthesis
 To the rescue of old drugs
To the rescue of old drugs