- NEWS AND VIEWS
Fossil ape hints at how walking on two feet evolved
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 575, 445-446 (2019)
doi: https://doi.org/10.1038/d41586-019-03347-0
References
Wood. B. & Harrison, T. Nature 470, 347–352 (2011).
Almécija, S. et al. Nature Commun. 4, 2888 (2013).
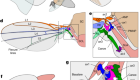
Böhme, M. et al. Nature 575, 489–493 (2019).
Darwin, C. The Descent of Man (Murray, 1871).
Richmond, B. G., Begun, D. R. & Strait, D. S. Am. J. Phys. Anthropol. 116, 70–105 (2001).
Crompton, R. H., Seller, W. I. & Thorpe, S. K. S. Phil. Trans. R. Soc. B 265, 3301–3314 (2010).
Lovejoy, C. O. Science 326, 74 (2009).
Thorpe, S. K. S., Holder, R. L. & Crompton, R. H. Science 316, 1328–1331 (2007).
Begun, D. R., Nargolwalla, M. C. & Kordos, L. Evol. Anthropol. 21, 10–23 (2012).
Ward, C. V. in Handbook of Paleoanthropology (eds Henke, W. & Tattersall, I.) 1363–1386 (Springer, 2015).
Morgan, M. E. et al. Proc. Natl Acad. Sci. USA 112, 82–87 (2015).

 Read the paper: A new Miocene ape and locomotion in the ancestor of great apes and humans
Read the paper: A new Miocene ape and locomotion in the ancestor of great apes and humans
 Walking on your sensitive sole
Walking on your sensitive sole
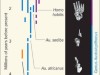 Those feet in ancient times
Those feet in ancient times