- NEWS AND VIEWS
The structure of a T-cell mechanosensor
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 573, 502-504 (2019)
doi: https://doi.org/10.1038/d41586-019-02646-w
References
Dong, D. et al. Nature 573, 546–552 (2019).
Meuer, S. C. et al. Nature. 303, 808–810 (1983).
Davis, M. M. & Bjorkman, P. J. Nature 334, 395–402 (1988).
Rudolph, M. G., Stanfield, R. L. & Wilson, I. A. Annu. Rev. Immunol. 24, 419–466 (2006).
Wang, J. H. & Reinherz, E. L. Immunol. Rev. 250, 102–119 (2012).
Feng, Y., Reinherz, E. L. & Lang, M. J. Trends Immunol. 39, 596–560 (2018).
Call, M. E., Pyrdol, J., Wiedmann, M. & Wucherpfennig, K. W. Cell 111, 967–979 (2002).
Brazin, K. N. et al. Immunity 49, 829–841 (2018).
Mallis, R. J. et al. J. Biomol. NMR 73, 319–332 (2019).
Liu, B., Chen, W., Evavold, B. D. & Zhu, C. Cell 157, 357–368 (2014).
Sibener, L. V. et al. Cell 174, 672–687 (2018).
Chakraborty A. K. & Weiss, A. Nature Immunol. 15, 798–807 (2014).
Bäckström, B. T. et al. Immunity 5, 437–447 (1996).

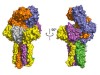 Read the paper: Structural basis of assembly of the human T cell receptor–CD3 complex
Read the paper: Structural basis of assembly of the human T cell receptor–CD3 complex
 How T cells spot tumour cells
How T cells spot tumour cells
 The patterns of T-cell target recognition
The patterns of T-cell target recognition



