- NEWS FEATURE
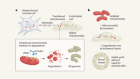
What lava lamps and vinaigrette can teach us about cell biology
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 555, 300-302 (2018)
doi: https://doi.org/10.1038/d41586-018-03070-2
References
Brangwynne, C. P. et al. Science 324, 1729–1732 (2009).
Wilson, E. B. Science 10, 33–45 (1899).
Brangwynne, C. P., Mitchison, T. J. & Hyman, A. A. Proc. Natl Acad. Sci. USA 108, 4334–4339 (2011).
Li, P. et al. Nature 483, 336–340 (2012).
Kato, M. et al. Cell 149, 753–767 (2012).
Nott, T. J. et al. Mol. Cell. 57, 936–947 (2015).
lbaum-Garfinkle, S. et al. Proc. Natl Acad. Sci. USA 112, 7189–7194 (2015).
Patel, A. et al. Cell 162, 1066–1077 (2015).
Molliex, A. et al. Cell 163, 123–133 (2015).
Wegmann, S. et al. EMBO J. 37, e98049 (2018).
Boulay, G. et al. Cell 171, 163–178.e19 (2017).
Alberti, S. & Hyman, A. A. Bioessays 38, 959–968 (2016).
Franzmann, T. M. et al. Science 359, eaao5654 (2018).
Riback, J. A. et al. Cell 168, 1028–1040.e19 (2017).
Larson, A. G. et al. Nature 547, 236–240 (2017).
Zeng, M. et al. Cell 166, 1163–1175.e12 (2016).
Shin, Y. et al. Cell 168, 159–171.e14 (2017).

 Single-cell biology
Single-cell biology
 DNA’s secret weapon against knots and tangles
DNA’s secret weapon against knots and tangles
 A sticky problem for chromosomes
A sticky problem for chromosomes
 RNA puts a freeze on cells
RNA puts a freeze on cells