Illustration by Michelle Thompson; Photos: Getty, Shutterstock
When Liangfang Zhang thinks of blood, he doesn’t think of a horror movie or a crime — or even, in any direct sense, of blood’s physiological job of delivering oxygen and glucose to the body’s cells and ushering away waste products. Blood, in Zhang’s eyes, is the most available source of human cell membrane there is. “Red blood cells are the most abundant cells in the body. A cubic centimetre of blood contains about five billion of them,” says Zhang, a nanotechnologist at the University of California, San Diego in La Jolla. “That means we don’t have a supply issue.”
During a stopover in Hong Kong in late August, Zhang explains his need for red blood cells. He is en route to ChinaNANO 2017, a huge nanoscience conference in Beijing, where he plans to tell the audience about a technique he uses to rip up red-blood-cell membranes, such that the fragments form small spheres — vesicles — that each go on to encapsulate a nanoparticle of interest. Because the membrane shrouds the nanoparticle, the foreign material can travel through the body unchallenged by the immune system.
Research into cellular and subcellular membranes has been quietly taking off over the past five years, thanks largely to the development and deployment of a host of new methods. These techniques are revealing in unprecedented detail the molecules that make up membranes, how they work and, in many cases, how they move between different parts of the cell.
The advancements have opened up the field of membrane science to researchers in other areas. No longer are membranes the preserve of cell biologists and biochemists; Zhang started his career as a chemical engineer — a discipline that, in his words, “used to be geared towards the oil business”. But training in an area that emphasized chemical purification, mass and heat transfer — not to mention an engineer’s instinct to deconstruct and design — has served him well as a membrane investigator. Today, his lab houses every flavour of bench researcher, from immunologists to material scientists. And as the range of specialities that are finding a place in membrane research has broadened, so, too, has the geographical spread of leading labs, most notably into China.
Zhang turns the conversation to poison. “If you look at the toxins that attack red blood cells, currently we know of about 80 toxin families, and they each do it in different, specific ways,” he says. Some attack by binding to protein receptors on the surface of red blood cells, gaining a foothold that makes them almost impossible to shift, he explains. Others interact with the phospholipids that constitute the membrane, a process that tears the cell apart. This variance has hindered the development of any kind of ‘cure-all’ solution. “Basically, creating a synthetic material that could simultaneously target 80-odd families of toxin would be an impossible task, because many of these toxins work completely differently,” says Zhang.
Zhang’s lab has developed something they call a nanosponge, which mops up all kinds of haemolytic — red-blood-cell-rupturing — poisons, from animal venom to the fluids produced by infectious agents such as methicillin-resistant Staphylococcus aureus (MRSA). The nanosponge is made of a polymer core that serves as structural support within a red-blood-cell-membrane wrapping. Each sponge is more than 3,000 times smaller in surface area than a red blood cell, but can soak up tens to hundreds of haemolytic poison molecules. It operates as a decoy, drawing toxins towards itself and away from living cells (C.-M. J. Hu et al. Nature Nanotechnol. 8, 336–340; 2013). “The only way to produce a complete solution is to use natural cell membrane,” says Zhang. “It has all of the components the toxins can target.”
A molecule of poison is just one of hundreds of thousands of chemicals that a cell might encounter over its lifetime. The membrane holds the cell together, and works as a barrier to the outside world. If anything on the outside wants to manipulate cell behaviour — perhaps by preventing cell reproduction or changing cell metabolism — it has to get the message through the membrane first. This is true for both natural and synthetic processes; roughly 60% of prescription drugs target membrane proteins.
Methodological outreach
In the early 1980s, Johann Deisenhofer, Robert Huber and Hartmut Michel determined the structure of the photosynthetic-reaction centre of a bacterium, for which they shared the Nobel Prize in Chemistry in 1988. By bouncing X-rays off a crystallized sample — a technique at the time already well established in the orderly world of inorganic chemistry — and doing the mathematics to interpret the diffraction patterns, they built a map of the proteins involved (J. Deisenhofer et al. J. Mol. Biol. 180, 385–398; 1984).
Crystallization was a major step forward for protein characterization, but it’s not well suited to membrane proteins. When layered as a membrane, lipids apply lateral pressure that can hold an embedded protein in a particular conformation. Rip a protein from its spot in a plane of fat molecules — something typically needed for X-ray scientists to get the crystalline samples they crave — and you might change its shape, which could mask what the protein does and how it works.
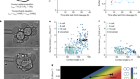
One method exciting the field is cryo-electron microscopy (cryo-EM). The technique helps scientists to elucidate molecular structures by bouncing beams of electrons over a sample, after freezing the structures to stop them from jiggling around. “Cryo-EM has become very popular as a means to determine membrane-protein structure,” says Ronald Clarke, a chemist at the University of Sydney, Australia, who studies ion pumps — proteins that transport ions across membranes.
Once the technology behind cryo-EM was fine-tuned, flash-freezing proved much more structurally respectful of the delicate associations between protein subunits than turning them into crystals and zapping them with X-rays. “Now, membrane-protein structures are being solved quite regularly,” says Clarke. “That means lots of theoreticians have been entering the field, because it’s now possible to carry out simulations of molecular dynamics. From structural clues, they model how membrane proteins function.” In other words, membrane science is slowly being permeated by computer science.
Being able to determine the detailed architecture of membrane proteins is a big deal. Today, the Protein Data Bank, an online repository for known 3D structures of biological molecules, holds just one membrane protein for about every 300 non-membrane ones, despite the fact that membrane proteins make up about one-quarter of the total roster of different proteins in the body.
Biophysicist Tao Xu has personally experienced how developing a technique can spur a career in membrane science. In 2004, Xu, at the Chinese Academy of Science’s Institute of Biophysics in Beijing, came up with a way to track the body’s response to insulin. He used a method known as total internal reflection fluorescence microscopy (or TIRF) to watch vesicles that are rich in a protein called GLUT4 as they flow from intracellular compartments to the cell-surface membrane of a muscle or fat cell, with which they fuse. Once the cell membrane is glutted with GLUT4 proteins, it can help to regulate blood-sugar levels by allowing more glucose to enter the cell.
Many people with type II diabetes have impaired GLUT4 translocation, so a method for tracking the process can isolate exactly what tends to go awry. “After we developed the vesicle-visualization method, many people wanted to collaborate with us,” says Xu.
Chinese growth
Xu has about 20 people in his lab, many of whom work on another kind of vesicle that shuffles proteins around the interior of pancreatic beta cells, also important in diabetes. “There are many people working in the field of membrane science in China — and many of them trained in the US and Europe,” he says. “There’s a feeling that it’s growing very rapidly here. I think it’s the right time to start a big programme.”
Xu is referring to a new offering from the National Natural Science Foundation of China. The organization has launched a call for research into organelle interactions, initially for projects starting in January 2018 and running for two or three years. A sum of about 30 million yuan (US$4.5 million) has been put aside this year towards this purpose, and ongoing discussions suggest that government support might continue for four years and could be extended further.
Among the applicants is Song Baoliang, a biochemist at Wuhan University in China, who researches the role of cholesterol in cell membranes. “I think this will really help membrane biology and cell biology in China,” he says. He thinks that some of the money will go towards research into membrane contact sites — an up-and-coming topic. These sites are stretches of organelle membranes that do not fuse with each other, but are near enough to interact chemically in some way. Scientists are hoping to work out what that interaction looks like.
Baoliang has watched membrane science develop in China for the past dozen years, following his return home from a postdoc at the University of Texas Southwestern Medical Center in Dallas, where he worked under 1985 Nobel prizewinners Michael Brown and Joseph Goldstein, who shared the award in physiology or medicine. The job market shifts slowly, but as grants in China have gradually increased, the competition for them is becoming stiffer. This is largely because of the quality and quantity of new work being done, says Baoliang. “A lot of the graduate students who train in my lab still apply for postdocs in the US, but in recent years, some have chosen to stay in China.” He adds, “there are even a few cases of Americans and Europeans coming to China for a postdoc”.
The Chinese government is encouraging principal investigators such as Baoliang to recruit foreigners as part of a wider drive to become competitive in the life sciences. Many applications to Baoliang’s lab come from South Asian and African research students. For researchers interested in temporarily making a move overseas, Arunima Chaudhuri, a postdoc from India at Yale University in New Haven, Connecticut (see ‘Q&A: Arunima Chaudhuri’), recommends aiming to specialize in an area in which your home country lacks expertise — that is, if your intention is to return home for most of your career. “You should work out of your comfort zone,” she says.
Wanjin Hong is in a good position to observe the shifting flow of membrane researchers around the globe. A specialist in intracellular transport at the National University of Singapore, Hong says that graduate students come to his lab from all over Asia and Europe, especially from China, Malaysia and Eastern Europe. A high percentage also come from the University of Manchester, UK, with which the Singaporean university has an institutional agreement. “We see a mixture of ability among the students,” he says. In particular, he claims that applications from Chinese students are no longer as competitive. “But that’s because there are more opportunities for them at home, with many excellent principal investigators in China now working in the area.”
Back in Hong Kong, Zhang reflects on young scientists’ prospects in the United States. “I wouldn’t say the job market is saturated in this area — I mean, membrane science is so broad, and getting broader,” he says. “It’s tough. But I think the major challenge is to have an idea and communicate it well. That’s what you need for cross-disciplinary collaboration — and that’s where innovation really comes out.”

 A day in the life of a cell biologist
A day in the life of a cell biologist
 The revolution will not be crystallized: a new method sweeps through structural biology
The revolution will not be crystallized: a new method sweeps through structural biology
 Science stars of China
Science stars of China
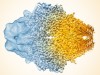 Cryo-electron microscopy wins chemistry Nobel
Cryo-electron microscopy wins chemistry Nobel