Abstract
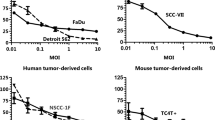
RH2 is a neurovirulent γ134.5 gene-deficient herpes simplex virus type 1 (HSV-1) with a lytic ability in human squamous cell carcinoma (SCC) cells; it is related to spontaneously occurring HSV-1 mutant HF10. The effect of RH2 on SCC was examined using a syngeneic C3H mouse model. After infection of mouse SCCVII cells with RH2, cell viability was decreased at first, but recovered by prolonged culture, indicating the limited replication of RH2. The antitumor ability of RH2 was examined using a bilateral SCCVII tumor model. The growth of the RH2-injected tumors was suppressed compared with that of phosphate-buffered saline-injected tumors. Moreover, the growth of contralateral tumor of RH2-treated mice was also suppressed significantly. The splenocytes of C3H mice treated with RH2 lysed more SCCVII cells than NFSaY83 cells and YAC-1 cells. The cytotoxicity of the splenocytes on SCCVII cells was significantly greater than that of splenocytes from tumor-bearing mice. Removal of CD8+ T cells from splenocytes decreased their cell killing activity remarkably. The antitumor effect of RH2 on SCCVII xenografts in nude mice was not demonstrated. These results indicate that RH2 exhibited a suppressive effect on mouse SCC, even if the replication of RH2 was limited. This is ascribed to the ability of RH2 to enhance existing tumor-specific cytotoxic T lymphocyte activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Martuza RL, Malick A, Maekert JM, Ruffner KL, Coen DM . Experimental therapy of human glioma by means of a genetically engineered virus mutant. Science 1991; 252: 854–856.
Mineta T, Rabkin SD, Yazaki T, Hunter WD, Martuza RL . Attenuated multi-mutated herpes simplex virus-1 for the treatment of malignant glioma. Nature Med 1995; 1: 938–943.
Liu BL, Robinson M, Han ZQ, Branston RH, English C, Reay P et al. ICP34.5 deleted herpes simplex virus with enhanced oncolytic, immune stimulating, and anti-tumour properties. Gene Ther 2003; 10: 292–303.
Senzer NN, Kaufman HL, Amatruda T, Nemunaitis M, Reid T, Daniels G et al. Phase II clinical trial of a granulocyte-macrophage colony-stimulating factor-encoding, second-generation oncolytic herpesvirus in patients with unresectable metastatic melanoma. J Clin Oncol 2009; 27: 5763–5771.
Harrington KJ, Hingorani M, Tanay MA, Hickey J, Bhide SA, Clarke PM et al. Phase I/II study of oncolytic HSVGM-CSF in combination with radiotherapy and cisplatin in untreated stage III/IV squamous cell cancer of the head and neck. Clin Cancer Res 2010; 16: 4005–4015.
Carew JF, Kooby DA, Halterman MW, Federoff HJ, Fong Y . Selective infection and cytolysis of human head and neck squamous cell carcinoma with sparing of normal mucosa by cytotoxic herpes simplex virus type 1 (G207). Hum Gene Ther 1999; 10: 1599–1606.
Chou J, Kern ER, Whitley RJ, Roizman B . Mapping of herpes simplex virus-1 neurovirulence to γ134.5, a gene nonessential for growth in culture. Science 1990; 250: 1262–1266.
He B, Gross M, Roizman B . The gamma(1) 34.5 protein of herpes simplex virus 1 complexes with protein phosphatase 1 alpha to dephosphorylate the alpha subunit of the eukaryotic translation initiation factor 2 and preclude the shutoff of protein synthesis by double-stranded RNA-activated protein kinase. Proc Natl Acad Sci USA 1997; 94: 843–848.
Takakuwa H, Goshima F, Nozawa N, Yoshikawa T, Kimata H, Nakao A et al. Oncolytic viral therapy using a spontaneously generated herpes simplex virus type 1 variant for disseminated peritoneal tumor in immunocompetent mice. Arch Virol 2003; 148: 813–825.
Nakamori M, Fu X, Rousseau R, Chen SY, Zhang X . Destruction of nonimmunogenic mammary tumor cells by a fusogenic oncolytic herpes simplex virus induces potent antitumor immunity. Mol Ther 2004; 9: 658–665.
Fu X, Tao L, Cai R, Prigge J, Zhang X . A mutant type 2 herpes simplex virus deleted for the protein kinase domain of the ICP10 gene is a potent oncolytic virus. Mol Ther 2006; 13: 882–890.
Israyelyan A, Chouljenko VN, Baghian A, David AT, Kearney MT, Kousoulas KG . Herpes simplex virus type-1(HSV-1) oncolytic and highly fusogenic mutants carrying the NV1020 genomic deletion effectively inhibit primary and metastatic tumors in mice. Virol J 2008; 5: 68.
Price DL, Lin SF, Han Z, Simpson G, Coffin RS, Wong J et al. Oncolysis using herpes simplex virus type 1 engineered to express cytosine deaminase and a fusogenic glycoprotein for head and neck squamous cell carcinoma. Arch Otolaryngol Head Neck Surg 2010; 136: 151–158.
Fujimoto Y, Mizuno T, Sugiura S, Goshima F, Kohno S, Nakashima T et al. Intratumoral injection of herpes simplex virus HF10 in recurrent head and neck squamous cell carcinoma. Acta Otolaryngol(Stockh) 2006; 126: 1115–1117.
Nakao A, Kimata H, Imai T, Kikumori T, Teshigawara O, Nagasaka T et al. Intratumoral injection of herpes simplex virus HF10 in recurrent breast cancer. Ann Oncol 2004; 15: 988–989.
Nakao A, Kasuya H, Sahin TT, Nomura N, Kanzaki A, Misawa M et al. A phase I dose-escalation clinical trial of intraoperative direct intratumoral injection of HF10 oncolytic virus in non-resectable patients with advanced pancreatic cancer. Cancer Gene Ther 2011; 18: 167–175.
Wong RJ, Patel SG, Kim S, DeMatteo RP, Malhotra S, Bennett JJ et al. Cytokine gene transfer enhances herpes oncolytic therapy in murine squamous cell carcinoma. Hum Gene Ther 2001; 12: 253–265.
Miller CG, Fraser NW . Requirement of an integrated immune response for successful neuroattenuated HSV-1 therapy in an intracranial metastatic melanoma model. Mol Ther 2003; 7: 741–747.
Varghese S, Rabkin SD, Liu R, Nielsen PG, Ipe T, Martuza RL . Enhanced therapeutic efficacy of IL-12, but not GM-CSF, expressing oncolytic herpes simplex virus for transgenic mouse derived prostate cancers. Cancer Gene Ther 2006; 13: 253–265.
Toda M, Rabkin SD, Kojima H, Martuza RL . Herpes simplex virus as an in situ cancer vaccine for the induction of specific anti-tumor immunity. Hum Gene Ther 1999; 10: 385–393.
Ghiringhelli F, Apetoh L, Housseau F, Kroemer G, Zitvogel L . Links between innate and cognate tumor immunity. Curr Opin Immunol 2007; 19: 224–231.
Prestwich RJ, Errington F, Diaz RM, Pandha HS, Harrington KJ, Melcher AA et al. The case of oncolytic viruses versus the immune system: waiting on the judgment of Solomon. Hum Gene Ther 2009; 20: 1119–1132.
Fu X, Tao L, Rivera A, Xu H, Zhang X . Virotherapy induces massive infiltration of neutrophils in a subset of tumors defined by a strong endogenous interferon response activity. Cancer Gene Ther 2011; 18: 785–794.
Andreansky S, Soroceanu L, Flotte ER, Chou J, Market JM, Gillespie GY et al. Evaluation of genetically engineered herpes simplex viruses as oncolytic agents for human malignant brain tumors. Cancer Res 1997; 57: 1502–1509.
Wheeler CE . The effect of temperature upon the production of herpes simplex virus in tissue culture. J Immunol 1958; 81: 98–106.
Yura Y, Kusaka J, Bando T, Yamamoto S, Yoshida H, Sato M . Enhancement of herpes simplex virus-induced polykaryocyte formation by 12-o-tetradecanoyl phorbol 13-acetate: Association with the reorganization of actin filaments and cell motility. Intervirology 2000; 43: 129–138.
Ogawa F, Takaoka H, Iwai S, Aota K, Yura Y . Combined oncolytic virotherapy with herpes simplex virus for oral squamous cell carcinoma. Anticancer Res 2008; 28: 3637–3646.
Takaoka H, Takahashi G, Ogawa F, Imai T, Iwai S, Yura Y . A novel fusogenic herpes simplex virus for oncolytic virotherapy of squamous cell carcinoma. Virol J 2011; 8: 294.
Takahashi G, Meshii N, Hamada M, Iwai S, Yura Y . Sequence of a fusogenic herpes simplex virus RH2 for oncolytic virotherapy. J Gen Virol 2012; 94 (Pt 4): 726–737.
Ushijima Y, Luo C, Goshima F, Yamauchi Y, Kimura H, Nishiyama Y . Determination and analysis of the DNA sequence of highly attenuated herpes simplex virus type 1 mutant HF10, a potential oncolytic virus. Microbes Infect 2007; 9: 142–149.
Sugiura S, Goshima F, Takakuwa H, Sata T, Nakashima T, Nishiyama Y . Treatment of solid sarcomas in immunocompetent mice with novel, oncolytic herpes simplex viruses. Otolaryngol Head Neck Surg 2004; 130: 470–478.
Tam MR, Emmons SL, Pollack SB . Analysis and enrichment of murine natural killer cells with the fluorescence-activated cell sorter. J Immunol 1980; 124: 650–655.
Decker T, Lohmann-Matthes ML . A quick and simple method for the quantitation of lactate dehydrogenase release in measurements of cellular cytotoxicity and tumor necrosis factor (TNF) activity. J. Immunol Methods 1988; 115: 61–69.
Sasaki T, Ohno T . Cytotoxicity tests on eye drop preparations by LDH release assay in human cultured cell lines. Toxicol In Vitro 1994; 8: 1113–1119.
Gessner A, Blum H, Röllinghoff M . Differential regulation of IL-9-expression after infection with Leishmania major in susceptible and resistant mice. Immunobiology 1993; 189: 419–435.
Martinez C, Delgado M, Gomariz RP, Ganea D . Vasoactive intestinal peptide and pituitary adenylate cyclase-activating polypeptide-38 inhibit IL-10 production in murine T lymphocytes. J Immunol 1996; 156: 4128–4136.
Watanabe D, Goshima F, Mori I, Tamada Y, Matsumoto Y, Nishiyama Y . Oncolytic virotherapy for malignant melanoma with herpes simplex virus type 1 mutant HF10. J Dermatol Sci 2008; 50: 185–196.
Esaki S, Goshima F, Kimura H, Murakami S, Nishiyama Y . Enhanced antitumoral activity of oncolytic herpes simplex virus with gemcitabine using colorectal tumor models. Int J Cancer 2012; 132: 1592–1601.
Waldhauer I, Steinle A . NK cells and cancer immunosurveillance. Oncogene 2008; 27: 5932–5943.
Tamesis RR, Foster CS . Natural killer cellular cytotoxicity against herpes simplex virus-infected cells in Igh-1-disparate mice. Invest Opthalmol Vis Sci 1990; 31: 2224–2229.
Thomas DL, Fraser NW . HSV-1 therapy of primary tumors reduces the number of metastases in an immune-competent model of metastatic breast cancer. Mol Ther 2003; 8: 543–551.
Todo T, Martuza RL, Rabkin SD, Johnson PA . Oncolytic herpes simplex virus vector with enhanced MHC class I presentation and tumor cell killing. Proc Natl Acad Sci USA 2001; 98: 6396–6401.
Li H, Dutuor A, Fu X, Zhang X . Induction of strong antitumor immunity by an HSV-2-based oncolytic virus in a murine mammary tumor model. J Gene Med 2007; 9: 161–169.
McKee TD, Grandi P, Mok W, Alexandrakis G, Insin N, Zimmer JP et al. Degradation of fibrillar collagen in a human melanoma xenograft improves the efficacy of an oncolytic herpes simplex virus vector. Cancer Res 2006; 66: 2509–2513.
Acknowledgements
This investigation was supported in part by a Grant-in-aid for Scientific Research from the Ministry of Education, Science and Culture of Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Meshii, N., Takahashi, G., Okunaga, S. et al. Enhancement of systemic tumor immunity for squamous cell carcinoma cells by an oncolytic herpes simplex virus. Cancer Gene Ther 20, 493–498 (2013). https://doi.org/10.1038/cgt.2013.45
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2013.45