Abstract
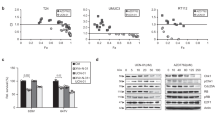
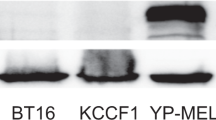
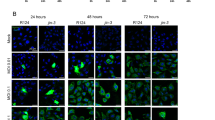
Anaplastic thyroid carcinoma (ATC) is the most aggressive thyroid cancer variant, accounting for 1–2% of all cases, but 33% of deaths, and exhibiting an average life expectancy of 5 months. ATC is largely unresponsive to radioactive iodine, chemotherapy, external beam radiation or surgery, underscoring the need for new and effective therapies. We evaluated the therapeutic potential of an oncolytic adenovirus, ONYX-411, that replicates selectively in and kills cells with dysfunction of the retinoblastoma (RB) pathway. In the present study, we report that ONYX-411 is able to induce cell death in eight human anaplastic carcinoma cell lines in vitro. The cytopathic effect of the virus is specific to cells with RB dysfunction, which appears to be frequent in ATC. We confirmed the expression of the coxsackie adenovirus receptor, CAR, in all ATC cell lines, demonstrating the potentially universal application of this oncolytic viral therapy to ATC. In addition, the growth of xenograft tumors induced in athymic mice with the ARO and DRO cell lines was significantly reduced by ONYX-411 treatment. These results indicate that ONYX-411 can be a potential therapeutic agent for the treatment of ATC, rendering this class of conditionally replicating adenoviruses an attractive candidate for clinical trials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Are C, Shaha AR . Anaplastic thyroid carcinoma: biology, pathogenesis, prognostic factors, and treatment approaches. Ann Surg Oncol 2006; 13: 453–464.
Haigh PI . Anaplastic thyroid carcinoma. Curr Treat Options Oncol 2000; 1: 353–357.
Copland JA, Marlow LA, Kurakata S, Fujiwara K, Wong AK, Kreinest PA et al. Novel high-affinity PPARgamma agonist alone and in combination with paclitaxel inhibits human anaplastic thyroid carcinoma tumor growth via p21WAF1/CIP1. Oncogene 2006; 25: 2304–2317.
Elisei R, Vivaldi A, Ciampi R, Faviana P, Basolo F, Santini F et al. Treatment with drugs able to reduce iodine efflux significantly increases the intracellular retention time in thyroid cancer cells stably transfected with sodium iodide symporter complementary deoxyribonucleic acid. J Clin Endocrinol Metab 2006; 91: 2389–2395.
Santarpia L, El-Naggar AK, Cote GJ, Myers JN, Sherman SI . Phoshatidylinositol 3-kinase/Akt and Ras/Raf-mitogen-activated kinase pathway mutations in anaplastic thyroid cancer. J Clin Endocrinol Metab 2008; 93: 278–284.
Saltman B, Singh B, Hedvat CV, Wreesmann VB, Ghossein R . Patterns of expression of cell cycle/apoptosis genes along the spectrum of thyroid carcinoma progression. Surgery 2006; 140: 899–905.
Hanahan D, Weinberg RA . The hallmarks of cancer. Cell 2000; 100: 57–70.
Bartek J, Bartkova J, Lukas J . The retinoblastoma protein pathway in cell cycle control and cancer. Exp Cell Res 1997; 237: 1–6.
Anwar F, Emond MJ, Schmidt RA, Hwang HC, Bronner MP . Retinoblastoma expression in thyroid neoplasms. Mod Pathol 2000; 13: 562–569.
Volante M, Croce S, Pecchioni C, Papotti M . E2F-1 transcription factor is overexpressed in oxyphilic thyroid tumors. Mod Pathol 2002; 15: 1038–1043.
McNeish IA, Bell SJ, Lemoine NR . Gene therapy progress and prospects: cancer gene therapy using tumour suppressor genes. Gene Therapy 2004; 11: 497–503.
Vaha-Koskela MJ, Heikkila JE, Hinkkanen AE . Oncolytic viruses in cancer therapy. Cancer Lett 2007; 254: 178–216.
Yu Z, Eisenberg DP, Singh B, Shah JP, Fong Y, Wong RJ . Treatment of aggressive thyroid cancer with an oncolytic herpes virus. Int J Cancer 2004; 112: 525–532.
Portella G, Scala S, Vitagliano D, Vecchio G, Fusco A . ONYX-015, an E1B gene-defective adenovirus, induces cell death in human anaplastic thyroid carcinoma cell lines. J Clin Endocrinol Metab 2002; 87: 2525–2531.
Abbosh PH, Li X, Li L, Gardner TA, Kao C, Nephew KP . A conditionally replicative, Wnt/beta-catenin pathway-based adenovirus therapy for anaplastic thyroid cancer. Cancer Gene Ther 2007; 14: 399–408.
Cohen EE, Rudin CM . ONYX-015. Onyx pharmaceuticals. Curr Opin Investig Drugs 2001; 2: 1770–1775.
Liu TC, Galanis E, Kirn D . Clinical trial results with oncolytic virotherapy: a century of promise, a decade of progress. Nat Clin Pract Oncol 2007; 4: 101–117.
Lam KY, Lo CY, Chan KW, Wan KY . Insular and anaplastic carcinoma of the thyroid: a 45-year comparative study at a single institution and a review of the significance of p53 and p21. Ann Surg 2000; 231: 329–338.
Johnson L, Shen A, Boyle L, Kunich J, Pandey K, Lemmon M et al. Selectively replicating adenoviruses targeting deregulated E2F activity are potent, systemic antitumor agents. Cancer Cell 2002; 1: 325–337.
Dyson N, Guida P, McCall C, Harlow E . Adenovirus E1A makes two distinct contacts with the retinoblastoma protein. J Virol 1992; 66: 4606–4611.
Denizot F, Lang R . Rapid colorimetric assay for cell growth and survival. Modifications to the tetrazolium dye procedure giving improved sensitivity and reliability. J Immunol Methods 1986; 89: 271–277.
Meier O, Greber UF . Adenovirus endocytosis. J Gene Med 2004; 6 (Suppl 1): S152–S163.
Fueyo J, Alemany R, Gomez-Manzano C, Fuller GN, Khan A, Conrad CA et al. Preclinical characterization of the antiglioma activity of a tropism-enhanced adenovirus targeted to the retinoblastoma pathway. J Natl Cancer Inst 2003; 95: 652–660.
Saito Y, Sunamura M, Motoi F, Abe H, Egawa S, Duda DG et al. Oncolytic replication-competent adenovirus suppresses tumor angiogenesis through preserved E1A region. Cancer Gene Ther 2006; 13: 242–252.
Mizukami Y, Kohgo Y, Chung DC . Hypoxia inducible factor-1 independent pathways in tumor angiogenesis. Clin Cancer Res 2007; 13: 5670–5674.
Mukhopadhyay D, Datta K . Multiple regulatory pathways of vascular permeability factor/vascular endothelial growth factor (VPF/VEGF) expression in tumors. Semin Cancer Biol 2004; 14: 123–130.
Acknowledgements
This work was supported by NIH Grant CA80117 (NLE), a generous donation from the Price Foundation, Florida Department of Health Bankhead-Coley grant (JAC) and a grant for rare cancers from Dr Ellis and Dona Brunton (JAC). We wish to extend their appreciation to Dr Lori Erickson for help with the interpretation of the hematoxylin and eosin-stained slides of ATC-xenografted tumors in nude mice. We are also indebted to Dr Henry J Hiddinga for assistance with the animal studies.
Author information
Authors and Affiliations
Corresponding author
Additional information
Disclosure
The authors have nothing to disclose.
Rights and permissions
About this article
Cite this article
Reddi, H., Madde, P., Reichert-Eberhardt, A. et al. ONYX-411, a conditionally replicative oncolytic adenovirus, induces cell death in anaplastic thyroid carcinoma cell lines and suppresses the growth of xenograft tumors in nude mice. Cancer Gene Ther 15, 750–757 (2008). https://doi.org/10.1038/cgt.2008.44
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2008.44
Keywords
This article is cited by
-
Recombinant oncolytic Newcastle disease virus displays antitumor activities in anaplastic thyroid cancer cells
BMC Cancer (2018)
-
Novel permissive murine immunocompetent orthotopic colon carcinoma model for comparison of the antitumoral and safety profiles of three Adv-TKs
Gene Therapy (2014)
-
Preclinical efficacy of the oncolytic measles virus expressing the sodium iodide symporter in iodine non-avid anaplastic thyroid cancer: a novel therapeutic agent allowing noninvasive imaging and radioiodine therapy
Cancer Gene Therapy (2012)