Abstract
Aim:
To investigate the mechanism of silibinin-protected isoproterenol-induced apoptosis in rat cardiac myocytes.
Methods:
The viability of rat cardiac myocytes was measured by MTT method. The apoptotic ratio was measured by terminal deoxynucleotidyl transferase-mediated dUTP nick end-labeling. Protein kinase C (PKC) activity assay was carried out according to the instructions of the PepTag non-radioactive protein kinase C assay kit. Western blot analysis was used to evaluate the level of Ras, Raf-1 and mitogen-activated protein kinase (MAPK) expression.
Results:
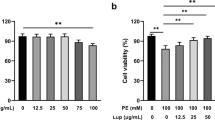
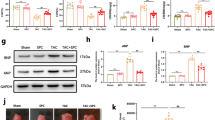
The protective effects of silibinin were significantly suppressed by inhibitors, including genistein, manumycin A and GW5074 [inhibitors for protein tyrosine kinases (PTK), Ras and Raf-1, respectively]. The exposure of rat cardiac myocytes to isoproterenol alone caused decreased PKC activity, which was prevented by pretreatment with silibinin dose-dependently. Simultaneously, the increased expression of Ras and Raf-1 activated by silibinin were blocked by the PKC inhibitor, stauroporine. In addition, the extracellularly responsive kinase (ERK) inhibitor, PD98059, suppressed silibinin-protected apoptosis, whereas the p38 MAPK inhibitor, SB203580, protected cardiac myocytes from isoproterenol-induced injury, and the c-Jun N-terminal kinase (JNK) inhibitor, SP600125 had no protective effects. Furthermore, Western blot analysis showed that the expression of phosphorylated ERK was increased by silibinin, the expression of phosphorylated p38 MAPK was decreased and total ERK, p38, JNK and phosphorylated JNK MAPK did not change after treatment with both isoproterenol and silibinin. Furthermore, pretreatment of cardiac myocyte with PKC, Ras and Raf inhibitors significantly blocked ERK phosphorylation.
Conclusion:
Silibinin is suggested to protect isoproterenol-induced rat cardiac myocyte apoptosis by activating the tyrosine kinase pathway, PKC and MAPK pathways.
Similar content being viewed by others
Article PDF
References
Berenji K, Drazner MH, Rothermel BA, Hill JA . Does load induced ventricular hypertrophy progress to systolic heart failure? Am J Physiol Heart Circ Physiol 2005; 289: H8–H16.
Frey N, Katus HA, Olson EN, Hill JA . Hypertrophy of the heart: a new therapeutic target? Circulation 2004; 109: 1580–9.
Francis GS, Chon JN, Johnson G, Rector TS, Goldman S, Simon A . Plasma norepinephrine, plasma renin activity, and congestive heart failure. Relations to survival and the effects of therapy in V-HeFT II. The V-HeFT VA Cooperative Studies Group. Circulation, 1993; 87 ( Suppl 6): VI40–8.
Communal C, Singh K, Pimentel DR, Colucci WS . Norepinephrine stimulates apoptosis in adult rat ventricular myocytes by activation of the β-adrenergic pathway. Circulation, 1998; 98: 1329–34.
Li LH, Wu LJ, Zhou B, Wu Z, Tashiro S, Onodera S, et al. Silymarin prevents UV irradiation-induced A375-S2 cell apoptosis. Biol Pharm Bull, 2004; 27: 1031–6.
Vaziri H, Dessain SK, Eaton EN, Imai SI, Frye RA, Pandita TK, et al. SIR2(SIRT1) functions as an NAD-dependent p53 deacetylase. Cell, 2001; 107: 149–59.
Luo J, Nikolaev AY, Imai S, Chen D, Su F, Shiloh A, et al. Negative control of p53 by Sir2alpha promotes cell survival under stress. Cell, 2001; 107: 137–48.
Cohen HY, Miller C, Bitterman KJ, Wall NR, Hekking B, Kessler B, et al. Calorie restriction promotes mammalian cell survival by inducing the SIRT1 deacetylase. Science, 2004; 305: 390–2.
Valenzuela A, Guerra R, Videla LA . Antioxidant properties of the flavonoids silymarin and (+)-cyanidanol–3: comparison with butylated hydroxyanisole and butylated hydroxytoluene. Planta Med, 1986; 6: 438–40.
Kang JS, Jeon YJ, Park SK, Yang KH, Kim HM . Protection against lipopolysaccharide-induced sepsis and inhibition of interleukin-1β and prostaglandin E2 synthesis by silymarin. Biochem Pharmacol, 2004; 67: 175–81.
Higmi Y, Shimokawa I . Apoptosis in the aging process. Cell Tissue Res, 2000; 301: 125–32.
Zhang YP, Herman B . Ageing and apoptosis: mechanisms of ageing and development. Mech Aging Dev, 2002; 123: 245–60.
Campisi J . Cellular senescence and apoptosis: how cellular responses might influence aging phenotypes. Exp Gerontol, 2003; 38: 5–11.
Ping P, Zhang J, Zheng YT, Li RC, Dawn B, Tang XL, et al. Demonstration of selective protein kinase C-dependent activation of Src and Lck tyrosine kinases during ischemic preconditioning in conscious rabbits. Circ Res, 1999; 85: 542–50.
Post GR, Brown JH . G protein-coupled receptors and signaling pathways regulating growth responses. FASEB J, 1996; 10: 741–9.
Lowy DR, Willumsen BM . Function and regulation of Ras. Annu Rev Biochem, 1993; 62: 851–91.
Mattingly RR, Macara IG . Phosphorylation-dependent activation of the Ras-GRF/CDC25Mm exchange factor by muscarinic receptors and G-protein βγ subunits. Nature, 1996; 382: 268–70.
Marais R, Light Y, Paterson HF, Marshall CJ . Ras recruits Raf-1 to the plasma membrane for activation by tyrosine phosphorylation. EMBO J, 1995; 14: 3136–45.
Jelinek T, Dent P, Sturgill TW, Weber MJ . Ras-induced activation of Raf-1 is dependent on tyrosine phosphorylation. Mol Cell Biol, 1996; 16: 1027–34.
Fabian JR, Daar IO, Morrison DK . Critical tyrosine residues regulate the enzymatic and biological activity of Raf-1 kinase. Mol Cell Biol, 1993; 13: 7170–9.
Wang X, Wu BW, Wu DM . Effects of AMP579 and adenosine on L-type Ca2+ current in isolated rat ventricular myocytes. Acta Pharmacol Sin, 2005; 26: 559–62.
Esposito G, Rapacciuolo A, Naga Prasad SV . Genetic alterations that inhibit in vivo pressure-overload hypertrophy prevent cardiac dysfunction despite increased wall stress. Circulation, 2002; 105: 85–92.
Kyriakis JM, App H, Zhang XF, Banerjee P, Brautigan DL, Rapp UR, et al. Raf-1 activates MAP kinase-kinase. Nature, 1992; 358: 417–21.
Tian ZJ, Cui W, Li YJ, Hao YM, Du J, Liu F . Different contributions of STAT3, ERK1/2, and PI3-K signaling to cardiomyocyte hypertrophy by cardiotrophin- 1. Acta Pharmacol Sin, 2004; 25: 1157–64.
Mark L, Hugo AK, Norbert F . Novel molecular targets in the treatment of cardiac hypertrophy. Recent Pat Cardiovas Drug Discov, 2006; 1: 1–20.
Clerk A, Sugden PH . Untangling the web: specific signaling from PKC isoforms to MAPK cascades. Circ Res, 2001; 89: 847–9.
Kaburagi S, Hasegawa K, Morimoto T . The role of endothelin converting enzyme-1 in the development of α1-adrenergic-stimulated hypertrophy in cultured neonatal rat cardiac myocytes. Circulation, 1999; 99: 292–8.
Lasek W, Wankowicz A, Kuc K, Feleszko W, Golab J, Giermasz A . Augmentation of antitumor efficacy by the combination of actinomycin D with tumor necrosis factor-alpha and interferon-gamma on amelanoma model in mice. Cancer Immunol Immunother, 1995; 40: 315–21.
Suzuki K, Hino M, Kutsuna H, Hato F, Sakamoto C, Takahashi T . Selective activation of p38 mitogen-activated protein kinase cascade in human neutrophils stimulated by IL–1β. J Immunol, 2001; 167: 5940–7.
Marais R, Light Y, Mason C, Paterson H, Olson MF, Marshall CJ . Requirement of Ras-GTP/Raf complexes for activation of Raf-1 by protein kinase C. Science, 1998; 280: 109–12.
Clerk A, Pham FH, Fuller SJ . Regulation of mitogen activated protein kinases in cardiac myocytes through the small G protein Rac1. Mol Cell Biol, 2001; 21: 1173–84.
Wang L, Proud CG . Ras/Erk signaling is essential for activation of protein synthesis by Gq protein-coupled receptor agonists in adult cardiomyocytes. Circ Res, 2002; 91: 821–9.
Clerk A, Sugden PH . Small guanine nucleotide-binding proteins and myocardial hypertrophy. Circ Res, 2000; 86: 1019–23.
Sugden PH, Clerk A . Regulation of ERK subgroup of MAP kinase cascades through G protein-coupled receptors. Cell Signal, 1997; 9: 337–51.
Buday L, Downward J . Epidermal growth factor regulates p21ras through the formation of a complex of receptor, Grb2 adapter protein, and Sos nucleotide exchange factor. Cell, 1993; 73: 611–20.
Lowenstein EJ, Daly RJ, Batzer AG, Li W, Margolis B, Lammers R, et al. The SH2 and SH3 domain-containing protein Grb2 links receptor tyrosine kinases to ras signaling. Cell, 1992; 70: 431–42.
Bolli R . The late phase of preconditioning. Circ Res, 2000; 87: 972–83.
Tong H, Chen W, Steenbergen C, Murphy E . Ischemic preconditioning activates phosphatidylinositol-3-kinase upstream of protein kinase C. Circ Res, 2000; 87: 309–15.
Ping P, Zhang J, Pierce WM Jr, Bolli R . Functional proteomic analysis of protein kinase C signaling complexes in the normal heart and during cardioprotection. Circ Res, 2001; 88: 59–62.
Goke M, Kanai M, Lynch-Devaney K, Podolsky DK . Rapid mitogen-activated protein kinase activation by transforming growth factor α in wounded rat intestinal epithelial cells. Gastroenterology, 1998; 114: 697–705.
Yamazaki T, Komuro I, Kudo S, Zuo Y, Shiojima I, Mizuno T, et al. Mechanical stress activates protein kinase cascade of phosphorylation in neonatal rat cardiac myocytes. J Clin Invest, 1995; 96: 438–46.
Takahashi M, Berk BC . Mitogen-activated protein kinase (ERK1/2) activation by shear stress and adhesion in endothelial cells. J Clin Invest, 1996; 99: 2623–31.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhou, B., Wu, Lj., Tashiro, Si. et al. Activation of extracellular signal-regulated kinase during silibinin-protected, isoproterenol-induced apoptosis in rat cardiac myocytes is tyrosine kinase pathway-mediated and protein kinase C-dependent. Acta Pharmacol Sin 28, 803–810 (2007). https://doi.org/10.1111/j.1745-7254.2007.00472.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2007.00472.x
Keywords
This article is cited by
-
Beneficial Effect of Silymarin in Pressure Overload Induced Experimental Cardiac Hypertrophy
Cardiovascular Toxicology (2019)
-
Cystatin F involvement in adenosine A2A receptor-mediated neuroinflammation in BV2 microglial cells
Scientific Reports (2018)
-
Biokinetically-Based In Vitro Cardiotoxicity of Residual Oil Fly Ash: Hazard Identification and Mechanisms of Injury
Cardiovascular Toxicology (2013)
-
Inhibition of silibinin on migration and adhesion capacity of human highly metastatic breast cancer cell line, MDA-MB-231, by evaluation of β1-integrin and downstream molecules, Cdc42, Raf-1 and D4GDI
Medical Oncology (2012)
-
Isoproterenol Cytotoxicity is Dependent on the Differentiation State of the Cardiomyoblast H9c2 Cell Line
Cardiovascular Toxicology (2011)