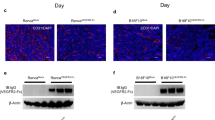
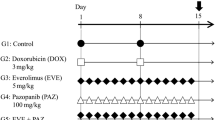
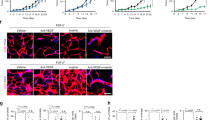
Abstract
Soluble Flk-1, a soluble vascular endothelial growth factor (VEGF) receptor, is a potent inhibitor of angiogenesis, which could restrain growth and metastasis of some experimental tumors. However, antiangiogenic agents alone cannot eradicate tumor completely, and should be combined with other therapy to enhance their effects. In this study, we evaluated the antitumor activity of the combination therapy in the immunocompetent BALB/c mice bearing H22 hepatoma and Meth A fibrosarcoma, respectively. Mice were treated with either msFlk-1 i.m. at 100 μg/mouse once every 3 days for four times from day 3 after the tumor cell injection, cisplatin cycled twice (2 mg/kg i.p. on days 4 and 11 after the tumor cell inoculation), or both agents together. Tumor growth and survival time were continually observed. Antiangiogenesis in vivo was determined by CD31 immunohistochemistry. Assessment of apoptotic cells and histological analysis was also conducted in tumor tissues. Our results showed that the combination therapy could evidently improve antitumor efficacy, including tumor growth suppression, mice survival prolongation, tumor cell apoptosis augmentation as well as neovascularization inhibition as compared with controls, without serious adverse effects. Our data suggest that the combination of DDP with msFlk-1 is more effective to suppress tumor growth in mice than either agent alone, and this combination regimen showed its potential for future clinical application.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Saris CP, van de Vaart PJ M, Rietbroek RC, Bloramaert FA . In vitro formation of DNA adducts by cisplatin, lobaplatin and oxaliplatin in calf thymus DNA in solution and in cultured human cells. Carcinogenesis 1996; 17: 2763–2769.
Faivre S, Chan D, Salinas R, Woynarowska B, Woynarowski JM . DNA strand breaks and apoptosis induced by oxaliplatin in cancer cells. Biochem Pharmacol 2003; 66: 225–237.
Yancopoulos GD, Klagsbrun M, Folkman J . Vasculogenesis, angiogenesis, and growth factors: ephrins enter the fray at the border. Cell 1998; 93: 661–664.
Folkman J . Tumor angiogenesis: therapeutic implications. N Engl J Med 1971; 285: 1182–1186.
Risau W . Mechanisms of angiogenesis. Nature 1997; 386: 671–674.
Gasparini G . The rationale and future potential of angiogenesis inhibitors in neoplasia. Drugs 1999; 58: 17–38.
Folkman J . Anti-angiogenesis: new concept for therapy of solid tumors. Ann Surg 1972; 175: 409–416.
Fidler IJ, Ellis LM . The implications of angiogenesis for the biology and therapy of cancer metastasis. Cell 1994; 79: 185–188.
Denekamp J . Angiogenesis, neovascular proliferation and vascular pathophysiology as targets for cancer therapy. Br J Radiol 1993; 66: 181–196.
Ferrara N, Alitalo K . Clinical applications of angiogenic growth factors and their inhibitors. Nat Med 1999; 5: 1359–1364.
Liotta LA, Steeg PS, Stetler-Stevenson WG . Cancer metastasis and angiogenesis: an imbalance of positive and negative regulation. Cell 1991; 64: 327–336.
Folkman J, Ingber D . Inhibition of angiogenesis. Semin Cancer Biol 1992; 3: 89–96.
Senger DR, Galli SJ, Dvorak AM, Perruzzi CA, Harvey VS, Dvorak HF . Tumor cells secrete a vascular permeability factor that promotes accumulation of ascites fluid. Science 1983; 219: 983–985.
Thomas KA . Vascular endothelial growth factor, a potent and selective angiogenic agent. J Biol Chem 1996; 271: 603–606.
Ferrara N, Davis-Smyth T . The biology of vascular endothelial growth factor. Endocr Rev 1997; 18: 4–25.
Ferrara N . Vascular endothelial growth factor. Eur J Cancer 1996; 32A: 2413–2422.
Neufeld G, Cohen T, Gengrinovitch S, Poltorak Z . Vascular endothelial growth factor (VEGF) and its receptors. FASEB J 1999; 13: 9–22.
Brekken RA, Thorpe PE . Vascular endothelial growth factor and vascular targeting of solid tumors. Anticancer Res 2001; 21: 4221–4229.
Takahashi Y, Kitadai Y, Bucana CD, Cleary KR, Ellis LM . Expression of vascular endothelial growth factor and its receptor, KDR, correlates with vascularity, metastasis, and proliferation of human colon cancer. Cancer Res 1995; 55: 3964–3968.
Asano M, Yukita A, Suruki H . Wide spectrum of antitumor activity of a neutralizing monoclonal antibody to human vascular endothelial growth factor. Jap J Cancer Res 1999; 90: 93–100.
Fong TAT, Shawver LK, Sun L, Tang C, App H, Powell TJ et al. SU5416 is a potent and selective inhibitor of the vascular endothelial growth factor receptor (Flk-1/KDR) that inhibits tyrosine kinase catalysis, tumor vascularization, and growth of multiple tumor types. Cancer Res 1999; 59: 99–106.
Marchand GS, Noiseux N, Tanguay JF, Sirois MG . Blockade of in vivo VEGF-mediated angiogenesis by antisense gene therapy: role of Flk-1 and Flt-1 receptors. Am J Physiol Heart Circ Physiol 2002; 282: 194–204.
Brekken RA, Overholser JP, Stastny VA, Waltenberger J, Minna JD, Thorpe PE . Selective inhibition of vascular endothelial growth factor (VEGF) receptor 2 (KDR/Flk-1) activity by a monoclonal anti-VEGF antibody blocks tumor growth in mice. Cancer Res 2000; 60: 5117–5124.
Prewett M, Huber J, Li Y, Santiago A, O'Connor W, King K et al. Antivascular endothelial growth factor receptor (Fetal Liver Kinase 1) monoclonal antibody inhibits tumor angiogenesis and growth of several mouse and human tumors. Cancer Res 1999; 59: 5209–5218.
Kim KJ, Li B, Winer J, Armanini M, Gillett N, Phillips HS et al. Inhibition of vascular endothelial growth factor-induced angiogenesis suppresses tumor growth in vivo. Nature 1993; 362: 841–844.
Takayama K, Ueno H, Nakanishi Y, Sakamoto T, Inoue K, Shimizu K et al. Suppression of angiogenesis and growth by gene transfer of a soluble form of vascular endothelial growth factor receptor into a remote organ. Cancer Res 2000; 60: 2169–2177.
Mahasreshti PJ, Navarro JG, Kataram M, Wang MH, Carey D, Siegal GP et al. Adenovirus-mediated soluble FLT-1 gene therapy for ovarian carcinoma. Clin Cancer Res 2001; 7: 2057–2066.
Lin P, Sankar S, Shan S, Dewhirst MW, Polverini PJ, Quinn TQ et al. Inhibition of tumor growth by targeting tumor endothelium using a soluble vascular endothelial growth factor receptor. Cell Growth Differ 1998; 9: 49–58.
Kendell RL, Thomas KA . Inhibition of vascular endothelial growth factor activity by endogenously encoded soluble receptor. Proc Natl Acad Sci 1993; 90: 10705–10709.
Klement G, Huang P, Mayer B, Green SK, Man S, Bohlen P et al. Differences in therapeutic indexes of combination metronomic chemotherapy and an Anti-VEGFR-2 antibody in multidrug-resistant human breast cancer xenografts. Clin Canc Res 2002; 8: 221–232.
Klement G, Baruchel S, Rak J, Man S, Clark K, Hicklin DJ et al. Continuous low-dose therapy with vinblastine and VEGF receptor-2 antibody induces sustained tumor regression without overt toxicity. J Clin Investig 2000; 105: 15–24.
Kakeji Y, Teicher BA . Preclinical studies of the combination of angiogenic inhibitors with cytotoxic agents. Invest New Drugs 1997; 15: 39–48.
Suzuki K, Hayashi N, Miyamoto Y, Yamamoto M, Ohkawa K, Ito Y et al. Expression of vascular permeability factor/vascular endothelial growth factor in human hepatocellular carcinoma. Cancer Res 1996; 56: 3004–3009.
Heidenreich R, Machein M, Nicolaus A, Hilbig A, Wild C, Clauss M et al. Inhibition of solid tumor growth by gene transfer of VEGF receptor-1 mutants. Int J Cancer 2004; 111: 348–357.
Xiao F, Wei Y, Yang L, Zhao X, Tian L, Ding Z et al. A gene therapy for cancer based on the angiogenesis inhibitor, vasostatin. Gene Therapy 2002; 9: 1207–1213.
Vermeulen PB, Gasparini G, Fox SB, Toi M, Martin L, McCulloch P et al. Quantification of angiogenesis in solid human tumours: an international consensus on the methodology and criteria of evaluation. Eur J Cancer 1996; 32: 2474–2784.
Su JM, Wei YQ, Tian L, Zhao X, Yang L, He QM et al. Active immunogene therapy of cancer with vaccine on the basis of chicken homologous matrix metalloproteinase-2. Cancer Res 2003; 63: 600–607.
Cai Q, Dmitrieva NI, Ferraris JD, Brooks HL, van Balkom BWM, Burg M . Pax2 expression occurs in renal medullary epithelial cells in vivo and in cell culture, is osmoregulated, and promotes osmotic tolerance. Proc Natl Acad Sci 2005; 102: 503–508.
Tseng JF, Farnebo FA, Kisker O, Becker CM, Kuo CJ, Folkman J et al. Adenovirus-mediated delivery of a soluble form of the VEGF receptor Flk1 delays the growth of murine and human pancreatic adenocarcinoma in mice. Surgery 2002; 132: 857–865.
Davidoff AM, Leary MA, Ng CY, Vanin EF . Gene therapy-mediated expression by tumor cells of the angiogenesis inhibitor flk-1 results in inhibition of neuroblastoma growth in vivo. J Pediatr Surg 2001; 36: 30–36.
Acknowledgements
This work was supported by National Basic Research Program of China (2001CB510001, 2004CB518800), the projects of National Natural Science Foundation of China, and National 863 Program.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, R., Zhang, XW., Wang, GQ. et al. Systemic inhibition of tumor growth by soluble Flk-1 gene therapy combined with cisplatin. Cancer Gene Ther 13, 940–947 (2006). https://doi.org/10.1038/sj.cgt.7700958
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7700958
Keywords
This article is cited by
-
Inhibitory effect of Bifidobacterium infantis-mediated sKDR prokaryotic expression system on angiogenesis and growth of Lewis lung cancer in mice
BMC Cancer (2012)
-
Antitumor effect of sFlt-1 gene therapy system mediated by Bifidobacterium Infantis on Lewis lung cancer in mice
Cancer Gene Therapy (2011)