Abstract
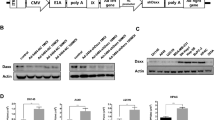
Survivin (SVV), an inhibitor of apoptosis protein, is found to be upregulated in many cancers. We previously demonstrated that a dominant-negative mutant SVV-D53A was able to induce apoptosis in a p53-independent manner. Here, we report the construction and characterization of a recombinant replication-deficient adenoviral vector encoding a human SVV-D53A gene for its effectiveness against tumor growth both in vitro and in vivo. Transfection of liver tumor cells QGY-7703 with Ad-SVV-D53A results in significant apoptosis as measured by an increase in sub-G1 DNA content, procaspase-9 activation and further downstream PARP-1 cleavage. Furthermore, animal studies using QGY-7703 liver carcinoma xenografts in nude mice revealed that treatment of QGY-7703 cells with dominant-negative SVV-D53A, but not with wild-type SVV-adenovirus, prevents tumor outgrowth, inhibits growth of established tumors and results in a notably improved survival advantages in xenograft studies. Both the transferase-mediated dUTP nick-end labeling assay and immunostaining experiment demonstrated that tumor growth inhibition is associated with apoptosis induced by SVV-D53A expression. Taken together, these data suggest that recombinant adenovirus Ad-SVV-D53A carrying a Survivin dominant-negative gene SVV-D53A promotes apoptosis-mediated tumor suppression and could potentially be a promising candidate for cancer therapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ameisen JC . On the origin, evolution, and nature of programmed cell death: a timeline of four billion years. Cell Death Differ 2002; 9: 367–393.
Raff MC, Barres BA, Burne JF, Coles HS, Ishizaki Y, Jacobson MD . Programmed cell death and the control of cell survival: lessons from the nervous system. Science 1993; 262: 695–700.
Meier P, Finch A, Evan G . Apoptosis in development. Nature 2000; 407: 796–801.
Borner C . The Bcl-2 protein family: sensors and checkpoints for life-or-death decisions. Mol Immunol 2003; 39: 615–647.
Reed JC . The Survivin saga goes in vivo. J Clin Invest 2001; 108: 965–969.
Tamm I, Wang Y, Sausville E, Scudiero DA, Vigna N, Oltersdorf T et al. IAP-family protein survivin inhibits caspase activity and apoptosis induced by Fas (CD95), Bax, caspases, and anticancer drugs. Cancer Res 1998; 58: 5315–5320.
Verdecia MA, Huang H, Dutil E, Kaiser DA, Hunter T, Noel JP . Structure of the human anti-apoptotic protein survivin reveals a dimeric arrangement. Nat Struct Biol 2000; 7: 602–608.
Pisarev V, Yu B, Salup R, Sherman S, Altieri DC, Gabrilovich DI . Full-length dominant-negative survivin for cancer immunotherapy. Clin Cancer Res 2003; 9: 6523–6533.
Mesri M, Wall NR, Li J, Kim RW, Altieri DC . Cancer gene therapy using a survivin mutant adenovirus. J Clin Invest 2001; 108: 981–990.
Song Z, Liu S, He H, Hoti N, Wang Y, Feng S et al. A single amino acid change (Asp53 → Ala53) converts Survivin from anti-apoptotic to pro-apoptotic. Mol Biol Cell 2004; 15: 1287–1296.
Wu Q, Moyana T, Xiang J . Cancer gene therapy by adenovirus-mediated gene transfer. Curr Gene Ther 2001; 1: 101–122.
He TC, Zhou S, da Costa LT, Yu J, Kinzler KW, Vogelstein B . A simplified system for generating recombinant adenoviruses. Proc Natl Acad Sci USA 1998; 95: 2509–2514.
Dion LD, Fang J, Garver Jr RI . Supernatant rescue assay vs polymerase chain reaction for detection of wild type adenovirus-contaminating recombinant adenovirus stocks. J Virol Methods 1996; 56: 99–107.
Hehir KM, Armentano D, Cardoza LM, Choquette TL, Berthelette PB, White GA et al. Molecular characterization of replication-competent variants of adenovirus vectors and genome modifications to prevent their occurrence. J Virol 1996; 70: 8459–8467.
Song Z, Yao X, Wu M . Direct interaction between survivin and Smac/DIABLO is essential for the anti-apoptotic activity of survivin during taxol-induced apoptosis. J Biol Chem 2003; 278: 23130–23140.
Anderson RD, Haskell RE, Xia H, Roessler BJ, Davidson BL . A simple method for the rapid generation of recombinant adenovirus vectors. Gene Therapy 2000; 7: 1034–1038.
Li J, Werner E, Hergenhahn M, Poirey R, Luo Z, Rommelaere J et al. Expression profiling of human hepatoma cells reveals global repression of genes involved in cell proliferation, growth, and apoptosis upon infection with parvovirus H-1. J Virol 2005; 79: 2274–2286.
Freedman DA, Levine AJ . Nuclear export is required for degradation of endogenous p53 by MDM2 and human papillomavirus E6. Mol Cell Biol 1998; 18: 7288–7293.
Fasbender A, Zabner J, Chillon M, Moninger TO, Puga AP, Davidson BL et al. Complexes of adenovirus with polycationic polymers and cationic lipids increase the efficiency of gene transfer in vitro and in vivo. J Biol Chem 1997; 272: 6479–6489.
Byk T, Haddada H, Vainchenker W, Louache F . Lipofectamine and related cationic lipids strongly improve adenoviral transfection efficiency of primitive human hematopoietic cells. Hum Gene Ther 1998; 9: 2493–2502.
Sharma S, Stolina M, Lin Y, Gardner B, Miller PW, Kronenberg M et al. T cell-derived IL-10 promotes lung cancer growth by suppressing both T cell and APC function. J Immunol 1999; 163: 5020–5028.
Modesitt SC, Ramirez P, Zu Z, Bodurka-Bevers D, Gershenson D, Wolf JK . In vitro and in vivo adenovirus-mediated p53 and p16 tumor suppressor therapy in ovarian cancer. Clin Cancer Res 2001; 7: 1765–1772.
Tu SP, Cui JT, Liston P, Huajiang X, Xu R, Lin MC et al. Gene therapy for colon cancer by adeno-associated viral vector-mediated transfer of survivin Cys84Ala mutant. Gastroenterology 2005; 128: 361–375.
Fangusaro JR, Jiang Y, Holloway MP, Caldas H, Singh V, Boue DR et al. Survivin, survivin-2B, and survivin-deItaEx3 expression in medulloblastoma: biologic markers of tumour morphology and clinical outcome. Br J Cancer 2005; 92: 359–365.
Kawasaki H, Altieri DC, Lu CD, Toyoda M, Tenjo T, Tanigawa N . Inhibition of apoptosis by survivin predicts shorter survival rates in colorectal cancer. Cancer Res 1998; 58: 5071–5074.
Andersen MH, Pedersen LO, Capeller B, Brocker EB, Becker JC, thor Straten P . Spontaneous cytotoxic T-cell responses against survivin-derived MHC class I-restricted T-cell epitopes in situ as well as ex vivo in cancer patients. Cancer Res 2001; 61: 5964–5968.
Salvesen GS, Duckett CS . IAP proteins: blocking the road to death's door. Nat Rev Mol Cell Biol 2002; 3: 401–410.
Altieri DC . Survivin, versatile modulation of cell division and apoptosis in cancer. Oncogene 2003; 22: 8581–8589.
Ambrosini G, Adida C, Altieri DC . A novel anti-apoptosis gene, survivin, expressed in cancer and lymphoma. Nat Med 1997; 3: 917–921.
Andersen MH, thor SP . Survivin – a universal tumor antigen. Histol Histopathol 2002; 17: 669–675.
Kato J, Kuwabara Y, Mitani M, Shinoda N, Sato A, Toyama T et al. Expression of survivin in esophageal cancer: correlation with the prognosis and response to chemotherapy. Int J Cancer 2001; 95: 92–95.
Li F, Ambrosini G, Chu EY, Plescia J, Tognin S, Marchisio PC et al. Control of apoptosis and mitotic spindle checkpoint by survivin. Nature 1998; 396: 580–584.
Li F, Ackermann EJ, Bennett CF, Rothermel AL, Plescia J, Tognin S et al. Pleiotropic cell-division defects and apoptosis induced by interference with survivin function. Nat Cell Biol 1999; 1: 461–466.
Kappler M, Bache M, Bartel F, Kotzsch M, Panian M, Wurl P et al. Knockdown of survivin expression by small interfering RNA reduces the clonogenic survival of human sarcoma cell lines independently of p53. Cancer Gene Therapy 2004; 11: 186–193.
Lens SM, Wolthuis RM, Klompmaker R, Kauw J, Agami R, Brummelkamp T et al. Survivin is required for a sustained spindle checkpoint arrest in response to lack of tension. EMBO J 2003; 22: 2934–2947.
Uchida H, Tanaka T, Sasaki K, Kato K, Dehari H, Ito Y et al. Adenovirus-mediated transfer of siRNA against survivin induced apoptosis and attenuated tumor cell growth in vitro and in vivo. Mol Ther 2004; 10: 162–171.
Acknowledgements
We are grateful to Dr Ronald Rodriguez (Johns Hopkins University) for LNCAP and DU145 cell lines. This research was supported by a 973 grant (2002CB713702) from Ministry of Science and Technology of China; grants from the National Natural Science Foundation of China (30530200, 90208027, 30370308 and 30121001) and an ARC grant (to Wu Mian) from Ministry of Education, Singapore.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhu, DE., Höti, N., Song, Z. et al. Suppression of tumor growth using a recombinant adenoviral vector carrying the dominant-negative mutant gene Survivin-D53A in a nude mice model. Cancer Gene Ther 13, 762–770 (2006). https://doi.org/10.1038/sj.cgt.7700952
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7700952
Keywords
This article is cited by
-
The roles of tumor-derived exosomes in non-small cell lung cancer and their clinical implications
Journal of Experimental & Clinical Cancer Research (2018)
-
The novel gene HA117 promotes in vitro and in vivo drug resistance in mouse colon tumor cells
Cancer Gene Therapy (2017)
-
Gene therapy of liver cancer: an update
Journal Africain du Cancer / African Journal of Cancer (2011)
-
Survivin knockdown by short hairpin RNA abrogates the growth of human hepatocellular carcinoma xenografts in nude mice
Cancer Gene Therapy (2010)