Abstract
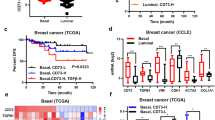
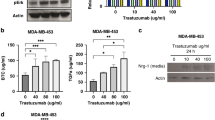
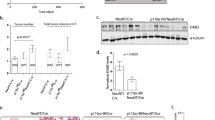
EphA2, a receptor tyrosine kinase, is elevated in many invasive human breast cancers, and the majority of EphA2 remains unphosphorylated. The successful attachment of ligand EphrinA1 present on the surface of adjacent cells to EphA2 initiates EphA2 phosphorylation leading to its turnover. In vivo efficacy of various approaches targeting EphA2 for breast cancer therapy is usually evaluated in nude mice bearing human breast cancer xenografts. In order to establish an immunocompetent mouse model of breast cancer for EphA2-targeted therapies, we evaluated a mouse breast cancer cell line (MT1A2) for EphA2 expression and phosphorylation. Overexpression of EphA2 was observed in MT1A2 cells and the majority of it remained unphosphorylated signifying that EphA2 in MT1A2 cells behaved similar to that of human breast cancer cells. Human adenovirus subtype 5 (HAd5) vectors expressing secretory forms of EphrinA1 were used for in vitro and in vivo targeting of MT1A2-derived EphA2. MT1A2 cells infected with HAd-EphrinA1-Fc (HAd expressing extracellular domain of human EphrinA1 attached to Fc portion of human IgG1 heavy chain) induced EphA2 activation and its turnover. This led to inhibition in MT1A2 cell colony formation in soft agar and cell viability in monolayer culture. In addition, MT1A2 cells-infected with HAd-EphrinA1-Fc failed to form tumors in syngeneic FVB/n mice at least 32 days postinoculation. Moreover, intratumoral inoculation of FVB/n mice-bearing MT1A2-induced tumors with HAd-EphrinA1-Fc slowed the tumor growth and also resulted in the development of vector-specific immune response. These results indicate that FVB/n mice-bearing MT1A2-induced tumors could serve as an immunocompetent model of breast cancer for EphA2-targeted therapeutic strategies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Zelinski DP, Zantek ND, Stewart JC, et al. EphA2 overexpression causes tumorigenesis of mammary epithelial cells. Cancer Res. 2001; 61: 2301–2306.
Nakamoto M, Bergemann AD . Diverse roles for the Eph family of receptor tyrosine kinases in carcinogenesis. Microsc Res Techn. 2002; 59: 58–67.
Bartley TD, Hunt RW, Welcher AA, et al. B61 is a ligand for the ECK receptor protein-tyrosine kinase. Nature. 1994; 368: 558–560.
Binns KL, Taylor PP, Sicheri F, et al. Phosphorylation of tyrosine residues in the kinase domain and juxtamembrane region regulates the biological and catalytic activities of Eph receptors. Mol Cell Biol. 2000; 20: 4791–4805.
Zisch AH, Pazzagli C, Freeman AL, et al. Replacing two conserved tyrosines of the EphB2 receptor with glutamic acid prevents binding of SH2 domains without abrogating kinase activity and biological responses. Oncogene. 2000; 19: 177–187.
Kullander K, Mather NK, Diella F, et al. Kinase-dependent and kinase-independent functions of EphA4 receptors in major axon tract formation in vivo. Neuron. 2001; 29: 73–84.
Miao H, Burnett E, Kinch M, et al. Activation of EphA2 kinase suppresses integrin function and causes focal-adhesion-kinase dephosphorylation. Nat Cell Biol. 2000; 2: 62–69.
Pratt RL, Kinch MS . Activation of the EphA2 tyrosine kinase stimulates the MAP/ERK kinase signaling cascade. Oncogene. 2002; 21: 7690–7699.
Carles-Kinch K, Kilpatrick KE, Stewart JC, et al. Antibody targeting of the EphA2 tyrosine kinase inhibits malignant cell behavior. Cancer Res. 2002; 62: 2840–2847.
Noblitt L, Bangari DS, Shukla S, et al. Inhibition in tumorigesis of breast cancer cells following EphA2 activation by adenoviral vectors expressing ligand ephrinA1. Cancer Gene Ther. 2004, in press.
Wang HT, Norris KM, Mansky LM . Analysis of bovine leukemia virus Gag membrane targeting and late domain function. J Virol. 2002; 76: 8485–8493.
Myoui A, Nishimura R, Williams PJ, et al. C-SRC tyrosine kinase activity is associated with tumor colonization in bone and lung in an animal model of human breast cancer metastasis. Cancer Res. 2003; 63: 5028–5033.
Addison CL, Braciak T, Ralston R, et al. Intratumoral injection of an adenovirus expressing interleukin 2 induces regression and immunity in a murine breast cancer model. Proc Natl Acad Sci USA. 1995; 92: 8522–8526.
Putzer BM, Hitt M, Muller WJ, et al. Interleukin 12 and B7-1 costimulatory molecule expressed by an adenovirus vector act synergistically to facilitate tumor regression. Proc Natl Acad Sci USA. 1997; 94: 10889–10894.
Graham FL, Smiley J, Russell WC, et al. Characteristics of a human cell line transformed by DNA from human adenovirus type 5. J Gen Virol. 1977; 36: 59–74.
Graham FL, Prevec L . Manipulation of adenovirus vectors. In: Murray EJ, ed. Methods of Molecular Biology: Gene Transfer and Expression Protocols. Totowa: Humana Press; 1991: 109–128.
Mittal SK, Middleton DM, Tikoo SK, et al. Pathogenesis and immunogenicity of bovine adenovirus type 3 in cotton rats (Sigmodon hispidus). Virology. 1995; 213: 131–139.
Fagotto F, Funayama N, Gluck U, et al. Binding to cadherins antagonizes the signaling activity of beta-catenin during axis formation in Xenopus. J Cell Biol. 1996; 132: 1105–1114.
Zantek ND, Azimi M, Fedor-Chaiken M, et al. E-cadherin regulates the function of the EphA2 receptor tyrosine kinase. Cell Growth Differ. 1999; 10: 629–638.
Kinch MS, Clark GJ, Der CJ, et al. Tyrosine phosphorylation regulates the adhesions of ras-transformed breast epithelia. J Cell Biol. 1995; 130: 461–471.
Stewart AK, Lassam NJ, Quirt IC, et al. Adenovector-mediated gene delivery of interleukin-2 in metastatic breast cancer and melanoma: results of a phase 1 clinical trial. Gene Therapy. 1999; 6: 350–363.
Palmer K, Hitt M, Emtage PC, et al. Combined CXC chemokine and interleukin-12 gene transfer enhances antitumor immunity. Gene Therapy. 2001; 8: 282–290.
Coffman KT, Hu M, Carles-Kinch K, et al. Differential EphA2 epitope display on normal versus malignant cells. Cancer Res. 2003; 63: 7907–7912.
Brantley DM, Cheng N, Thompson EJ, et al. Soluble Eph A receptors inhibit tumor angiogenesis and progression in vivo. Oncogene. 2002; 21: 7011–7026.
Klonjkowski B, Gilardi-Hebenstreit P, Hadchouel J, et al. A recombinant E1-deleted canine adenoviral vector capable of transduction and expression of a transgene in human-derived cells and in vivo. Hum Gene Ther. 1997; 8: 2103–2115.
Hofmann C, Loser P, Cichon G, et al. Ovine adenovirus vectors overcome preexisting humoral immunity against human adenoviruses in vivo. J Virol. 1999; 73: 6930–6936.
Moffatt S, Hays J, HogenEsch H, et al. Circumvention of vector-specific neutralizing antibody response by alternating use of human and non-human adenoviruses: implications in gene therapy. Virology. 2000; 272: 159–167.
Farina SF, Gao GP, Xiang ZQ, et al. Replication-defective vector based on a chimpanzee adenovirus. J Virol. 2001; 75: 11603–11613.
Xiang Z, Gao G, Reyes-Sandoval A, et al. Novel, chimpanzee serotype 68-based adenoviral vaccine carrier for induction of antibodies to a transgene product. J Virol. 2002; 76: 2667–2675.
Hemminki A, Kanerva A, Kremer EJ, et al. A canine conditionally replicating adenovirus for evaluating oncolytic virotherapy in a syngeneic animal model. Mol Ther. 2003; 7: 163–173.
Acknowledgements
We thank Jane Stewart, Shaji Abraham, Rebecca Pratt, and Keith Kikawa for their technical advice and Jane Kovach for excellent secretarial assistance. We are also thankful to Dr Mike Kinch, MedImmune, Inc., Gaithersburg, MD for discussions and for providing EphA2-specific reagents, and Dr William Muller, Department of Biology, McMaster University, Hamilton, Ontario, Canada for providing MT1A2 cell line. This work was supported by a Purdue Research Foundation Grant.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Noblitt, L., Bangari, D., Shukla, S. et al. Immunocompetent mouse model of breast cancer for preclinical testing of EphA2-targeted therapy. Cancer Gene Ther 12, 46–53 (2005). https://doi.org/10.1038/sj.cgt.7700763
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7700763
Keywords
This article is cited by
-
Dysregulated erythropoietin-producing hepatocellular receptor A2 (EphA2) is involved in tubal pregnancy via regulating cell adhesion of the Fallopian tube epithelial cells
Reproductive Biology and Endocrinology (2018)
-
Therapeutic targeting of EPH receptors and their ligands
Nature Reviews Drug Discovery (2014)
-
Activation of erythropoietin-producing hepatocellular receptor A2 attenuates cell adhesion of human fallopian tube epithelial cells via focal adhesion kinase dephosphorylation
Molecular and Cellular Biochemistry (2012)
-
EphrinA1 inhibits malignant mesothelioma tumor growth via let-7 microRNA-mediated repression of the RAS oncogene
Cancer Gene Therapy (2011)