Abstract
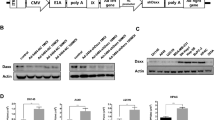
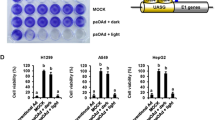
Gene-attenuated replication-competent adenoviruses are emerging as a promising new modality for the treatment of cancer. For the aim of improving adenoviral vectors for cancer gene therapy, we have constructed genetically attenuated adenoviral vectors with different combinations of E1B genes and investigated the possibility of enhanced oncolytic and replication effects of these engineered replication-competent adenoviruses. We show here that the cytolytic potency of each gene-attenuated replicating adenovirus differed significantly depending on the presence or deletion of E1B 55 kDa and E1B 19 kDa function. More specifically, among the constructed vectors (Ad-ΔE1B19, Ad-ΔE1B55, Ad-ΔE1B19/55, and Ad-wt), E1B 19 kDa–inactivated adenovirus (Ad-ΔE1B19) was the most potent against all tumor cells tested, inducing the largest-sized plaques and marked CPE. Further, cells infected with either Ad-ΔE1B19 or E1B19/55 kDa–deleted adenovirus (Ad-ΔE1B19/55) showed complete cell lysis with disintegrated cellular structure, whereas cells infected with Ad-wt maintained intact cellular and nuclear membrane with properly structured organelles. TUNEL and DNA fragmentation assay also revealed that the Ad-ΔE1B19 or Ad-ΔE1B19/55 adenovirus-infected cells showed more profound induction of apoptosis in comparison to wild-type adenovirus-infected cells. The presence of E1B 55 kDa gene was required for efficient viral replication and deletion of E1B 19 kDa function in replicating adenovirus-induced apoptosis, leading to increased cytopathic effects. Moreover, Ad-ΔE1B19 adenovirus showed a better antitumor effect than other E1B-attenuated adenoviruses. Taken together, the replicating adenoviruses deleted in E1B 19 kDa function may serve as an improved vector for anticancer gene therapy in combination with apoptosis-inducing modalities such as chemotherapeutic agents and radiation therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout











Similar content being viewed by others
References
Kirn D . Replication selective oncolytic adenoviruses: virotherapy aimed at genetic targets in cancer Oncogene 2000 19: 6660–6669
Kirn D . Clinical research results with dl1520 (Onyx-015), a replication-selective adenovirus for the treatment of cancer: what have we learned? Gene Ther 2001 8: 89–98
Kirn D, Martuza RL, Zwiebel J . Replication-selective virotherapy for cancer: biological principles, risk management and future directions Nat Med 2001 7: 781–787
Mulvihill S, Warren R, Venook A et al. Safety and feasibility of injection with an E1B-55 kDa gene–deleted, replication-selective adenovirus (ONYX-015) into primary carcinomas of the pancreas: a phase I trial Gene Ther 2001 8: 308–315
Ganly I, Eckhardt SG, Rodriguez GI et al. A phase I study of Onyx-015, an E1B attenuated adenovirus, administered intratumorally to patients with recurrent head and neck cancer Clin Cancer Res 2000 6: 798–806
Nemunaitis J, Ganly I, Khuri F et al. Selective replication and oncolysis in p53 mutant tumors with ONYX-015, an E1B-55 kD gene–deleted adenovirus, in patients with advanced head and neck cancer: a phase II trial Cancer Res 2000 60: 6359–6366
Khuri FR, Nemunaitis J, Ganly I et al. A controlled trial of intratumoral ONYX-015, a selectively-replicating adenovirus, in combination with cisplatin and 5-fluorouracil in patients with recurrent head and neck cancer Nat Med 2000 6: 879–885
Lamont JP, Nemunaitis J, Kuhn JA et al. A prospective phase II trial of ONYX-015 adenovirus and chemotherapy in recurrent squamous cell carcinoma of the head and neck (the Baylor experience) Ann Surg Oncol 2000 7: 588–592
Boyd JM, Malstrom S, Subramanian T et al. Adenovirus E1B 19 kDa and Bcl-2 proteins interact with a common set of cellular proteins Cell 1994 79: 1121
Chiou SK, Tseng CC, Rao L, White E . Functional complementation of the adenovirus E1B 19-kilodalton protein with Bcl-2 in the inhibition of apoptosis in infected cells J Virol 1994 68: 6553–6566
Boulakia CA, Chen G, Ng FW et al. Bcl-2 and adenovirus E1B 19 kDA protein prevent E1A-induced processing of CPP32 and cleavage of poly (ADP-ribose) polymerase Oncogene 1996 12: 529–535
Han J, Modha D, White E . Interaction of E1B 19 K with Bax is required to block Bax-induced loss of mitochondrial membrane potential and apoptosis Oncogene 1998 17: 2993–3005
Rao L, Debbas M, Sabbatini P et al. The adenovirus E1A proteins induce apoptosis, which is inhibited by the E1B 19-kDa and Bcl-2 proteins Proc Natl Acad Sci USA 1992 89: 7742–7746
Debbas M, White E . Wild-type p53 mediates apoptosis by E1A, which is inhibited by E1B Genes Dev 1993 7: 546–554
Shen Y, Shenk T . Relief of p53-mediated transcriptional repression by the adenovirus E1B 19-kDa protein or the cellular Bcl-2 protein Proc Natl Acad Sci USA 1994 91: 8940–8944
Huang DC, Cory S, Strasser A . Bcl-2, Bcl-XL and adenovirus protein E1B19 kD are functionally equivalent in their ability to inhibit cell death Oncogene 1997 14: 405–414
Xiang J, Gomez-Navarro J, Arafat W et al. Pro-apoptotic treatment with an adenovirus encoding Bax enhances the effect of chemotherapy in ovarian cancer J Gene Med 2000 2: 97–106
Tsuruta Y, Mandai M, Konishi I et al. Combination effect of adenovirus-mediated pro-apoptotic bax gene transfer with cisplatin or paclitaxel treatment in ovarian cancer cell lines Eur J Cancer 2001 37: 531–541
Babiss LE, Ginsberg HS, Darnell JE . Adenovirus E1B proteins are required for accumulation of late viral mRNA and for effects on cellular mRNA translation and transport Mol Cell Biol 1985 5: 2552–2558
Bridge E, Ketner G . Interaction of adenoviral E4 and E1b products in late gene expression Virology 1990 174: 345–353
Dobbelstein M, Roth J, Kimberly WT et al. Nuclear export of the E1B 55-kDa and E4 34-kDa adenoviral oncoproteins mediated by a rev-like signal sequence EMBO J 1997 16: 4276–4284
Lee H, Kim J, Lee B et al. Oncolytic potential of E1B 55 kDa–deleted YKL-1 recombinant adenovirus: correlation with p53 functional status Int J Cancer 2000 88: 454–463
Sauthoff H, Heitner S, Rom WN, Hay JG . Deletion of the adenoviral E1b-19 kD gene enhances tumor cell killing of a replicating adenoviral vector Hum Gene Ther 2000 11: 379–388
Lam KM . Apoptosis in chicken embryo fibroblasts caused by Newcastle disease virus Vet Microbiol 1995 47: 357–363
Secchiero P, Flamand L, Gibellini D et al. Human herpesvirus 7 induces CD4(+) T-cell death by two distinct mechanisms: necrotic lysis in productively infected cells and apoptosis in uninfected or nonproductively infected cells Blood 1997 90: 4502–4512
Hong JR, Lin TL, Hsu YL, Wu JL . Apoptosis precedes necrosis of fish cell line with infectious pancreatic necrosis virus infection Virology 1998 250: 76–84
Compton MM . A biochemical hallmark of apoptosis: internucleosomal degradation of the genome Cancer Metastasis Rev 1992 11: 105–119
Dobner T, Horikoshi N, Rubenwolf S, Shenk T . Blockage by adenovirus E4orf6 of transcriptional activation by the p53 tumor suppressor Science 1996 272: 1470–1473
Nevels M, Rubenwolf S, Spruss T, Wolf H, Dobner T . The adenovirus E4orf6 protein can promote E1A/E1B–induced focus formation by interfering with p53 tumor suppressor function Proc Natl Acad Sci USA 1997 94: 1206–1211
Grand RJ, Grant ML, Gallimore PH . Enhanced expression of p53 in human cells infected with mutant adenoviruses Virology 1994 203: 229–240
Querido E, Marcellus RC, Lai A et al. Regulation of p53 levels by the E1B 55-kilodalton protein and E4orf6 in adenovirus-infected cells J Virol 1997 71: 3788–3798
Steegenga WT, Riteco N, Jochemsen AG, Fallaux FJ, Bos JL . The large E1B protein together with the E4orf6 protein target p53 for active degradation in adenovirus infected cells Oncogene 1998 16: 349–357
Halbert DN, Cutt JR, Shenk T . Adenovirus early region 4 encodes functions required for efficient DNA replication, late gene expression, and host cell shutoff J Virol 1985 56: 250–257
Pilder S, Moore M, Logan J, Shenk T . The adenovirus E1B-55K transforming polypeptide modulates transport or cytoplasmic stabilization of viral and host cell mRNAs Mol Cell Biol 1986 6: 470–476
Leppard KN, Shenk T . The adenovirus E1B 55 kd protein influences mRNA transport via an intranuclear effect on RNA metabolism Embo J 1989 8: 2329–2336
Pilder S, Logan J, Shenk T . Deletion of the gene encoding the adenovirus 5 early region 1b 21,000-molecular-weight polypeptide leads to degradation of viral and host cell DNA J Virol 1984 52: 664–671
Subramanian T, Kuppuswamy M, Gysbers J, Mak S, Chinnadurai G . 19-kDa tumor antigen coded by early region E1b of adenovirus 2 is required for efficient synthesis and for protection of viral DNA J Biol Chem 1984 259: 11777–11783
Subramanian T, Kuppuswamy M, Mak S, Chinnadurai G . Adenovirus cyt+ locus, which controls cell transformation and tumorigenicity, is an allele of lp+ locus, which codes for a 19-kilodalton tumor antigen J Virol 1984 52: 336–343
Takemori N, Cladaras C, Bhat B, Conley AJ, Wold WS . cyt gene of adenoviruses 2 and 5 is an oncogene for transforming function in early region E1B and encodes the E1B 19,000-molecular-weight polypeptide J Virol 1984 52: 793–805
White E, Cipriani R, Sabbatini P, Denton A . Adenovirus E1B 19-kilodalton protein overcomes the cytotoxicity of E1A proteins J Virol 1991 65: 2968–2978
White E, Faha B, Stillman B . Regulation of adenovirus gene expression in human WI38 cells by an E1B-encoded tumor antigen Mol Cell Biol 1986 6: 3763–3773
Laster SM, Wood JG, Gooding LR . Tumor necrosis factor can induce both apoptotic and necrotic forms of cell lysis J Immunol 1988 141: 2629–2634
Gooding LR, Aquino L, Duerksen-Hughes PJ et al. The E1B 19,000-molecular-weight protein of group C adenoviruses prevents tumor necrosis factor cytolysis of human cells but not of mouse cells J Virol 1991 65: 3083–3094
Kurihara T, Brough DE, Kovesdi I, Kufe DW . Selectivity of a replication-competent adenovirus for human breast carcinoma cells expressing the MUC1 antigen J Clin Invest 2000 106: 763–771
Doronin K, Toth K, Kuppuswamy K et al. Tumor-specific, replication-competent adenovirus vectors overexpressing the adenovirus death protein J Virol 2000 74: 6147–6155
Subramanian TG, Tarodi B, Govindarajan R et al. Mutational analysis of the transforming and apoptosis suppression activities of the adenovirus E1B 175R protein Gene 1993 124: 173–181
Chartier C, Degryse E, Gantzer M et al. Efficient generation of recombinant adenovirus vectors by homologous recombination in Escherichia coli J Virol 1996 70: 4805–4810
Tollefson AE, Scaria A, Hermiston TW et al. The adenovirus death protein (E3-11.6 K) is required at very late stages of infection for efficient cell lysis and release of adenovirus from infected cells J Virol 1996 70: 2296–2306
Acknowledgements
We would like to thank all of our colleagues at Yosei Cancer Center, Seoul, Korea who have contributed to these studies. We thank J-H Son for his valueable contribution. This work was supported by grants from Ministry of Health & Welfare, Republic of Korea (HMP-01-PJI-PG3-20800-0115, C-O Yun) and Ministry of Commerce Industry and Energy, Republic of Korea (N03-990-5411-01-1-3, J-H Kim). Jaesung Kim is a graduate student sponsored by Brain Korea 21 Project for Medical Science, Yonsei University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, J., Cho, J., Kim, JH. et al. Evaluation of E1B gene-attenuated replicating adenoviruses for cancer gene therapy. Cancer Gene Ther 9, 725–736 (2002). https://doi.org/10.1038/sj.cgt.7700494
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7700494
Keywords
This article is cited by
-
Hepatocellular carcinoma-targeting oncolytic adenovirus overcomes hypoxic tumor microenvironment and effectively disperses through both central and peripheral tumor regions
Scientific Reports (2018)
-
Efficient lung orthotopic tumor-growth suppression of oncolytic adenovirus complexed with RGD-targeted bioreducible polymer
Gene Therapy (2014)
-
Local sustained delivery of oncolytic adenovirus with injectable alginate gel for cancer virotherapy
Gene Therapy (2013)
-
S100A2 promoter-driven conditionally replicative adenovirus targets non-small-cell lung carcinoma
Gene Therapy (2012)
-
Complete eradication of hepatomas using an oncolytic adenovirus containing AFP promoter controlling E1A and an E1B deletion to drive IL-24 expression
Cancer Gene Therapy (2012)