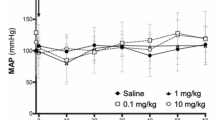
Abstract
Progressive growth and metastasis of solid tumors require angiogenesis, or the formation of new blood vessels. Endostatin is a 20-kDa carboxy-terminal fragment of collagen XVIII that has been shown to inhibit endothelial cell proliferation and tumor angiogenesis. Replication-deficient recombinant adenovirus (rAd) vectors were constructed, which encoded secreted forms of human and mouse endostatin (HECB and MECB, respectively), and, as a control, human alkaline phosphatase (APCB). Accumulation of endostatin was demonstrated in supernatants of cultured cells infected with the endostatin rAds. These supernatants disrupted tubule formation, inhibited migration and proliferation, and induced apoptosis in human dermal vascular endothelial cells or human vascular endothelial cells. Endostatin-containing supernatants had no effect on the proliferation of MidT2-1 mouse mammary tumor cells in vitro. A pharmacokinetic study of MECB in immunocompetent FVB mice demonstrated a 10-fold increase of serum endostatin concentrations 3 days after intravenous administration of 1×1010 particles of this rAd (215–257 ng/mL compared to 12–38 ng/mL in control rAd-treated mice). Intravenous administration of MECB reduced b-FGF stimulated angiogenesis into Matrigel plugs by 38%. Intratumoral MECB inhibited growth of MidT2-1 syngeneic mammary tumors in FVB mice, but had minimal impact on the growth of MDA-MB-231 human breast tumors in SCID mice. Intravenous therapy with MECB also initially inhibited growth of MidT2-1 tumors, but this activity was subsequently blocked by induced anti-rAd antibodies. In summary, endostatin gene therapy effectively suppressed angiogenic processes in vitro and in vivo in several model systems. Cancer Gene Therapy (2001) 8, 982–989
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Folkman JDAP . Blood vessel formation: What is its molecular basis? Cell 1996 87: 1153–1155
Hanahan DFJ . Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis Cell 1996 86: 353–364
O'Reilly MSBT, Shing Y, Fukai N, et al . Endostatin: An endogenous inhibitor of angiogenesis and tumor growth Cell 1997 88: 277–285
Boehm TFJ, Browder T, O'Reilly MS . Antiangiogenic therapy of experimental cancer does not induce acquired drug resistance Nature 1997 390: 404–407
Nielsen LL, M Gurnani, B Shi, et al . Derivation and initial characterization of a mouse mammary tumor cell line carrying the polyomavirus middle T antigen: Utility in the development of novel cancer therapeutics Cancer Res 2000 60: 7066–7074
Chartier CED, Gantzer M, Dieterle A, Pavirani A, Mehtali M . Efficient generation of recombinant adenovirus vectors by homologous recombination in Escherichia coli J Virol 1996 70: 4805–4810
Huyghe BLX, Sutjipto S, Sugarman B, et al . Purification of a type 5 recombinant adenovirus encoding human p53 by column chromatography Hum Gene Ther 1995 6: 1403–1416
Shabram PGD, Gouldreau A, Gregory R, et al . Analytical anion exchange HPLC of recombinant type-5 adenoviral particles Hum Gene Ther 1997 8: 453–465
Maizel JWD, Scharff M . The polypeptides of adenovirus: 1. Evidence for multiple protein components in the virion and a comparison of types 2, 7a, and 12 Virology 1968 36: 115–125
Musco MCS, Small D, Nodelman M, Sugarman B, Grace M . Comparison of flow cytometry and laser scanning cytometry for the intracellular evaluation of adenoviral infectivity and p53 protein expression in gene therapy Cytometry 1998 33: 290–296
Schuetz JDSE . Extracellular matrix regulation of multidrug resistance in primary monolayer cultures of adult rat hepatocytes Cell Growth Differ 1993 4: 31–40
Kozarsky KF, Mckinley DR, Austin LL, Raper SE, Stratford-Perricaudet LD, Wilson JM . In vivo correction of low density lipoprotein receptor deficiency in the Watanabe heritable hyperlipidemic rabbit with recombinant adenoviruses J Biol Chem 1994 269: 13695–13702
Wills KN, Maneval DC, Menzel P, et al . Development and characterization of recombinant adenoviruses encoding human p53 for gene therapy of cancer Hum Gene Ther 1994 5: 1079–1088
Mohanraj Dhanabal RR, Waterman MJF, Lu H, Knebelmann B, Segal M, Sukhatme VP . Endostatin induces endothelial cell apoptosis J Biol Chem 1999 274: 11721–11726
Gabriele Bergers KJ, Lo K-M, Folkman J, Hanahan D II . Effects of angiogenesis inhibitors on multistage carcinogenesis in mice Science 1999 284: 808–812
Erhard Hohenester TS, Olsen BR, Timp R . Crystal structure of the angiogenesis inhibitor endostatin at 1.5 Å resolution EMBO J 1998 17: 1656–1664
Dhanabal MRR, Volk R, Stillman IE, et al . Endostatin: Yeast production, mutants, and antitumor effect in renal cell carcinoma Cancer Res 1999 59: 189–197
Dhanabal MVR, Ramchandran R, Simons M, Sukhatme VP . Cloning, expression, and in vitro activity of human endostatin Biochem Biophys Res Commun 1999 258: 345–352
Noriko Yamaguchi BA-A, Lee M, Takako, et al . Endostatin inhibits VEGF-induced endothelial cell migration and tumor growth independently of zinc binding EMBO J 1999 18: 4414–4423
Boehm TOrM, Keough K, Shiloach J, Shapiro R, Folkman J . Zinc-binding of endostatin is essential for its antiangiogenic activity Biochem Biophys Res Commun 1998 252: 190–194
Kong HLHD, Song W, Kovesdi I, Hackett NR, Yayon A, Crystal RG . Regional suppression of tumor growth by in vivo transfer of a cDNA encoding a secreted form of the extracellular domain of the flt-1 vascular endothelial growth factor receptor Hum Gene Ther 1998 9: 823–833
Pengnian Lin JAB, Acheson A, Czeslaw, et al . Antiangiogenic gene therapy targeting the endothelium-specific receptor tyrosine kinase Tie2 Proc Natl Acad Sci USA 1998 95: 8829–8834
Sauter BVMO, Zhang WJ, Mandeli J, Woo SL . Adenovirus-mediated gene transfer of endostatin in vivo results in high level of transgene expression and inhibition of tumor growth and metastases Pro Nat Acad Sci USA 2000 97: 4802–4807
Chen QRKD, Stass SA, Mixson AJ . Liposomes complexed to plasmids encoding angiostatin and endostatin inhibit breast cancer in nude mice Cancer Res 1999 59: 3308–3312
Feldman ALRN, Alexander HR, Bartlett DL, Hwu P, Seth P, Libutti SK . Antiangiogenic gene therapy of cancer utilizing a recombinant adenovirus to elevate systemic endostatin levels in mice Cancer Res 2000 60: 1503–1506
Blezinger PWJ, Gondo M, Quezada A, et al . Systemic inhibition of tumor growth and tumor metastases by intramuscular administration of the endostatin gene Nat Biotechnol 1999 17: 343–348
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jin, X., Bookstein, R., Wills, K. et al. Evaluation of endostatin antiangiogenesis gene therapy in vitro and in vivo. Cancer Gene Ther 8, 982–989 (2001). https://doi.org/10.1038/sj.cgt.7700396
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7700396
Keywords
This article is cited by
-
MR Reporter Gene Imaging of Endostatin Expression and Therapy
Molecular Imaging and Biology (2010)
-
Liposome–DNA complexes infused intravenously inhibit tumor angiogenesis and elicit antitumor activity in dogs with soft tissue sarcoma
Cancer Gene Therapy (2006)
-
Endostatin therapy reveals a U-shaped curve for antitumor activity
Cancer Gene Therapy (2006)
-
Endostatin gene transfection using a cationic lipid: advantages of transfection before tumor cell inoculation and repeated transfection
Cancer Gene Therapy (2004)
-
Friends or foes — bipolar effects of the tumour stroma in cancer
Nature Reviews Cancer (2004)