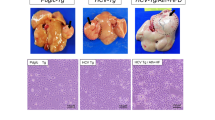
Abstract
The clinical potential of tumor therapies must be evaluated using animal models closely resembling human cancers. We investigated the impact of locally delivered interferon-γ (IFN-γ) on primary hepatocarcinoma spontaneously developed by T-SV40 transgenic mice. A single intratumor injection of adenovirus IFN-γ was sufficient enough to induce in vivo production of biologically active IFN-γ, as assessed by STAT1 activation. IFN-γ secretion led to the regression of primary tumor, principally by apoptosis of tumor hepatocytes. The lack of T-cells infiltrates in the liver upon treatment excluded a role of a specific immune response. In contrast, indirect pathways may include tumoricidal function of macrophages. Indeed, they were massively recruited in the entire liver under IFN-γ treatment; transmigration through hepatic blood vessels could be observed and co-localization with damaged hepatocytes was obvious. This correlated with nonparenchymal liver cell iNOS expression and high level of NO in hepatic extracts. Moreover, in vitro experiments showed that NO releasing agents induced cell death of freshly isolated tumor hepatocytes, suggesting that NO could be one of the major effector molecules. Altogether, these observations defined an important role of IFN-γ in controlling tumor development in a model of primary hepatocarcinoma. Cancer Gene Therapy (2001) 8, 193–202
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baratin, M., Ziol, M., Romieu, R. et al. Regression of primary hepatocarcinoma in cancer-prone transgenic mice by local interferon-γ delivery is associated with macrophages recruitment and nitric oxide production. Cancer Gene Ther 8, 193–202 (2001). https://doi.org/10.1038/sj.cgt.7700285
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7700285
Keywords
This article is cited by
-
Blood glutamate scavengers increase pro-apoptotic signaling and reduce metastatic melanoma growth in-vivo
Scientific Reports (2021)
-
Gene therapy of liver cancer: an update
Journal Africain du Cancer / African Journal of Cancer (2011)
-
Delivery of mengovirus-derived RNA replicons into tumoural liver enhances the anti-tumour efficacy of a peripheral peptide-based vaccine
Cancer Immunology, Immunotherapy (2008)
-
Histone deacetylase inhibitors enhance Ad5-TRAIL killing of TRAIL-resistant prostate tumor cells through increased caspase-2 activity
Apoptosis (2007)
-
A phase I trial of immunotherapy with intratumoral adenovirus-interferon-gamma (TG1041) in patients with malignant melanoma
Cancer Gene Therapy (2003)