Abstract
We studied three possible genotypes at 10 well-defined blood pressure (BP) QTLs using congenic rat lines. The central question was whether the hypertensive or normotensive allele is dominant, or whether there is partial dominance. The congenic strains were employed to investigate the BP effects of alleles originating from normotensive rats in the background of hypertensive Dahl salt-sensitive (DSS) rats. The normotensive alleles at eight QTLs were fully dominant over DSS alleles, which we tentatively interpreted as indicating that DSS rats incurred a loss of function at these loci and that the QTLs produced BP-reducing agents. In contrast, the normotensive allele of only one QTL was recessive over its DSS counterpart, implying a gain of function at this QTL or a null allele involved in generating a BP-elevating agent. Only one locus, C17QTL, had alleles exhibiting partial dominance. These estimates of dominance differ considerably from those obtained by QTL analysis in a F2 cross. This disagreement demonstrates the importance of establishing a cause–effect relationship between a QTL and its phenotypic effect via congenic strains. The dominance relationships suggest pertinent strategies for gene identification and pharmaceutical intervention.
Similar content being viewed by others
Introduction
It has been suggested that blood pressure (BP) is determined by the cumulative influence of QTLs, which, on average, have minor effects (Abiola et al., 2003; Caulfield et al., 2003; Harrap, 2003; Flint et al., 2005). This proposal implies that a phenotypic ‘threshold’ might need to be overcome by amassing multiple QTLs (Falconer and Mackey, 1996) before a BP effect is visible. This model would explain why no single human QTL with major BP effects has been found. But is this outcome actually due to the absence of such major QTLs, or to the difficulty of detecting them in population-based genetic analyses? Among the many factors impacting on QTL detection in human populations, genetic heterogeneity, incomplete penetrance and environmental influences are considered major stumbling blocks (Caulfield et al., 2003; Harrap, 2003).
As an alternative to human studies, inbred mammalian models can be experimentally manipulated under environmentally controlled conditions and, thus, can be more readily exploited to identify BP QTLs (Deng, 1998; Rapp, 2000; Flint et al., 2005). Moreover, mammalian models can be utilized to test if QTLs with minor effects might cumulatively determine BP (Caulfield et al., 2003; Harrap, 2003; Flint et al., 2005) or if some QTLs simply exhibit ‘major’ BP effects. One of these models is Dahl salt-sensitive (DSS) rats (Dahl et al., 1962). We previously (Garrett et al., 1998) analyzed a single F2 population derived from crosses of DSS with normotensive Lewis (LEW) rats and localized multiple QTLs, each of which accounted for 4.8–17.8% of total variance in the population. These results suggested that certain QTLs might have minor (e.g. 4.8%) BP effects, while others might manifest major (e.g. 17.8%) BP effects.
There are, however, major limitations to QTL mapping in mammals when based on linkage in a F2 population. First, QTLs are usually localized to segments of 20 cM or longer, even when a large number of animals are used and the QTLs exert major effects (Darvasi et al., 1993). Consequently, F2 linkage analysis cannot reveal how many QTLs are present within such a 20-cM intervals: it is incapable of separating closely linked QTLs. For example, our previous work revealed the presence of a QTL in a 46-cM region on rat Chr 10 (Deng and Rapp, 1995; Garrett et al., 1998), whereas, in fact, there are four QTLs in this segment. Three of them are clustered within a section of less than 15 cM (Palijan et al., 2003b; Charron et al., 2005a). Second, false negatives or false positives can appear. A false negative can be exemplified by the localization of C2QTL1. Linkage analyses detected no QTLs near the gene encoding neutral endopeptidase (Mme) (Deng et al., 1994b), yet it actually marked the location of C2QTL1 (Dutil et al., 2005). The gene encoding the inducible form of nitric oxide synthase (Nos2) is an example of a false positive. Nos2 was initially identified as marking a probable BP QTL location (Deng and Rapp, 1995), but was subsequently excluded (Palijan et al., 2003b).
To overcome the limitations imposed by linkage in QTL definition, congenic strains (Snell, 1948) have been utilized. The distinct value of rat congenic strains is firstly (Deng, 1998) the negligible genetic differences among individuals: they are more than 99.9% identical. Second, the small genetic differences that do exist between two congenic strains are well known, precisely defined and uniform. Thus, any observable phenotypic differences can be attributed directly to genetic differences. Two novel functional aspects of BP QTLs have become apparent from the analyses of congenic strains. First, each of them accounts for at least 33 mm Hg/88 mm Hg=38% of the total BP difference between DSS and LEW parental strains, as measured directly by telemetry (Moujahidine et al., 2004; Dutil et al., 2005; Grondin et al., 2005; Charron et al., 2005a), exhibiting the status of a major QTL. Second, each QTL appears have an individual influence on BP: no combination seems to be necessary for any one of them to affect BP, despite QTLQTL epistatic interactions (Deng and Rapp, 1992; Rapp et al., 1998; Palijan et al., 2003a; Charron et al., 2005a, 2005b; Dutil et al., 2005).
Similar results have been obtained with QTLs trapped in congenic strains constructed by replacing DSS alleles with homologous alleles from Milan normotensive (MNS) rats (Dutil et al., 2005). No previous studies have addressed the domiance of the QTL alleles. In our analyses, we studied the three possible genotypes at 10 well-defined BP QTLs, namely, homozygous for normotensive alleles, heterozygous, and homozygous for hypertensive alleles. The congenic strains chosen were those that carry the smallest number of genes in a QTL interval, thereby minimizing the probability that it contains an additional QTL. The results represent the first such analyses to investigate individual BP QTLs in well-defined and homogeneous genetic backgrounds, that is using congenic strains. Their initial detection by linkage analysis is summarized in Table 1. The central question that we addressed is: which allele of a QTL, that is, hypertensive or normotensive, is dominant, recessive or partially dominant? Classifying QTLs in this fashion may provide insights into their genetic bases and, consequently, facilitate research prioritization and the establishment of an appropriate strategy in gene discovery for each QTL.
Materials and methods
Animals
Protocols for handling and maintaining the animals were approved by our institutional animal protection committee. All experimental procedures were in accordance with institutional, provincial and federal regulations. DSS, LEW and MNS rats have previously been described (Palijan et al., 2003a, 2003b; Ariyarajah et al., 2004; Moujahidine et al., 2004; Charron et al., 2005a, 2005b; Dutil et al., 2005; Eliopoulos et al., 2005; Grondin et al., 2005). Nine congenic strains had trapped BP QTLs and were largely based on those isolated previously (Palijan et al., 2003b; Ariyarajah et al., 2004; Moujahidine et al., 2004; Charron et al., 2005a; Dutil et al., 2005; Grondin et al., 2005).
Congenic construction to define a QTL on DSS Chr 1
An additional congenic strain was produced by substituting a DSS chromosome segment of interest with a homologous section from LEW. The breeding procedure and screening protocol were essentially the same as those reported previously (Palijan et al., 2003a, 2003b; Ariyarajah et al., 2004; Charron et al., 2005b; Eliopoulos et al., 2005; Grondin et al., 2005). This new congenic strain for Chr 1 was designated as DSS.LEW-(D1Rat268-D1Chm2)/Lt (abbreviated as C1S.L1).
Generation of heterozygotes
A male of a congenic strain known to harbor a BP QTL was mated with a female DSS rat to produce F1 progeny. As all the QTLs to be studied were located on autosomes, a reciprocal cross was also performed, with a female of the same congenic strain mating with a DSS male. In so doing, the effect of X or Y chromosomes on the BP of a congenic strain could be excluded. BPs were measured in male heterozygous rats of F1 progeny from both reciprocal crosses along with the DSS and the congenic strain in question. The distribution of homozygote 1, heterozygote and homozygote 2 for each QTL followed an 1:2:1 Mendelian ratio in F2s (data not shown).
BP measurements
BP studies of the congenic strains were essentially made in the same way as previous studies (Palijan et al., 2003a, 2003b; Ariyarajah et al., 2004; Charron et al., 2005b; Eliopoulos et al., 2005; Grondin et al., 2005). In brief, male rats were weaned at 21 days of age, maintained on a low-salt diet (0.2% NaCl, Harlan Teklad 7034) and then fed a high-salt diet (2% NaCl, Harlan Teklad 94217), starting from 35 days of age until the end of the experiment. Telemetry probes were implanted when the rats were 56 days old (i.e. after 3 weeks on the high-salt diet) with their body weights between 250 and 320 g. BPs for all strains were measured at least at two different times to exclude seasonal and environmental influences. Thus, the BP data were pooled from separately reproducible measurements for each strain. All BP components such as systolic (SAPs), diastolic (DAPs) and mean arterial pressures (MAPs) were measured, but for the sake of easy comparisons, only MAPs are reported. DAPs and SAPs were consistent with MAPs of each strain (data not shown). As they were not significantly different, BP data from reciprocal crosses were pooled (results not presented) for all heterozygotes of the same strain.
Statistical analyses
Repeated measures' analysis of variance (ANOVA), followed by the Tukey test (Palijan et al., 2003a, 2003b; Ariyarajah et al., 2004; Charron et al., 2005b; Eliopoulos et al., 2005; Grondin et al., 2005), were used to compare the differences between homozygote 1, that is DSS in Figure 1 and homozygote 2 (either LL or MM), homozygote 1 and heterozygotes, and homozygote 2 and heterozygotes for each congenic strain harboring a QTL.
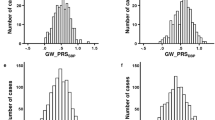
Comparison of MAPs among homozygotes and heterozygotes of 10 QTLs defined by congenic strains: to the left of (a–j) are chromosome maps and QTL interval definitions. Hatched, dark and open bars symbolize the genomes of DSS, LEW (or MNS) and crossover ambiguity respectively in each congenic strain. The size of the QTL interval in megabases (Mb) is indicated in parentheses after each QTL designation. The rest of the genome in each congenic strain and heterozygote of congenic strain was DSS. To the right of (a–j) are MAP comparisons among three genotypes of a QTL, that is homozygote SS (for DSS), homozygote LL (for LEW) or homozygote MM (for MNS) and heterozygote LS or heterozygote MS, depending on the congenic compositions. (a) Analysis of C1QTL; (b) analysis of C16QTL; (c) analysis of C17QTL; (d) analysis of C10QTL1; (e) analysis of C10QTL2; (f) analysis of C10QTL3; (g) analysis of C10QTL4; (h) analysis of C2QTL1; (i) analysis of C2QTL2; and (j) analysis of C2QTL3. Error bars in BP tracings represent s.e.m. n refers to the number of rats. The BP response patterns such as diurnal variations, among all the congenic strains were not different (data not shown). The BPs of each strain were measured, at least, during two different time periods, and then pooled to produce the final BP data. Only a 24-h average of BP was taken as 1 data point on the graph for each strain. MAP refers to averaged mean arterial pressure during the period of measurement for each strain and heterozygote. ANOVA with the Tukey test (P) compares MAPs between DSS and each of the congenic strains, DSS and a heterozygote of a congenic strain, and a congenic strain and its heterozygote. C1QTL represents the definition of a QTL on Chr 1 based on the newly generated congenic strain C1S.L1; C16QTL, a QTL on Chr 16 (Moujahidine et al., 2004) defined by a new congenic substrain C16S.L5; C17QTL, a QTL on Chr 17 as defined previously (Grondin et al., 2005); C10QTL1 to C10QTL4, QTLs on Chr 10 (Palijan et al., 2003b; Charron et al., 2005a); and C2QTL1 to C10QTL3, QTLs on Chr 2 as defined previously (Dutil et al., 2005).
Results
QTL locations, congenic strains and their BPs in both homozygotes and heterozygotes are presented side-by-side in Figure 1 for 10 QTLs. Assuming that 1 QTL is present in each congenic interval, alleles of C1QTL, C16QTL, C10QTL1, C10QTL2, C10QTL3 and C10QTL4 from LEW acted in a dominant mode (Figure 1a, b, d–g). Alleles of C2QTL1 and C2QTL2 from MNS were also dominant (Figure 1h and i). DSS alleles for these QTLs acted recessively. These data demonstrate that one copy of the MNS or LEW allele for each of these QTLs was sufficient to lower BP as much as two copies. A notable exception is that the MNS allele of C2QTL3 was recessive to its DSS counterpart (Figure 1j), indicating that two copies of MNS alleles were required to lower BP.
As the three QTLs on Chr 2 were closely linked (Dutil et al., 2005), it was essential to separate them by fine congenic mapping. Otherwise, the differing modes of action, that is dominance of C2QTL1 and C2QTL2 and recessivity of C2QTL3 for MNS alleles, would not have been distinguishable. In contrast, LEW alleles of C17QTL showed a dosage effect, that is BP of the heterozygotes (SL) fell in between those of homozygotes DSS (SS) and LEW (LL) (Figure 1c), although the differences between SL and LL were only of borderline significance (P<0.056). Thus, the functionality of C17QTL appears to be partially dominant. Table 2 compares the BP effect for each of the 10 QTLs, and calculates its percentage effect versus the total BP difference between two parental strains.
Discussion
The major findings of this study are that:
-
a)
Alleles at 8 QTLs showed dominance of the normotensive LEW or MNS strain, suggesting that the product for each of these QTL alleles leads to a reduction of BP. The DSS alleles at these QTLs are most readily interpreted as loss of function mutations, whereas the MNS or LEW alleles correspond to functional wild-type alleles. However, it should be borne in mind that there are other interpretations of dominance, including haploinsufficiency. Haploinsufficiency refers to cases where the amount of product produced by heterozygotes is insufficient to fulfill the function of the gene and, consequently, the null allele appears dominant over a normal allele (Wilkie, 1994; Zlotogora, 1997). Based on the working hypothesis that dominance indicates a loss or gain of function, the future research direction will be to search systematically, in QTL-containing intervals, for mutations that are responsible for a loss or gain of function, including little or no expression of the contrasting strain's product.
-
b)
For C17QTL, the LEW allele appears to be partially dominant. In this case, the identification of C17QTL will focus on the detection of mutations controlling the levels and functions of gene products of genes residing in the C17QTL interval.
Insights gained into the physiological functions of BP QTLs
Eight out of 10 QTLs documented in the current study (Figure 1) demonstrated dominance of normotensive alleles, corresponding to BP diminution. DSS QTL alleles are correspondingly recessive, indicating a loss of function or a lack of gene product. Thus, the products of MNS or LEW alleles for these eight QTLs are probably involved in generating, either directly or indirectly, BP-decreasing agents. Consequently, the products of these eight QTLs might impact treatments of hypertension, by pharmacological addition of the BP-decreasing agents specified by the MNS and LEW alleles. In contrast, the DSS allele of C2QTL3 is dominant, correlating with higher BP, whereas its MNS allele is recessive (Figure 1j). Therefore, the DSS C2QTL3 allele could represent a gain of function or haploinsufficiency. It follows that hypertension in DSS might have resulted, in part, from the existence of the C2QTL3 product of DSS alleles, which could be targeted for inhibition by pharmacological intervention to lower BP. The alleles of C17QTL showed partial dominance. C17QTL likely encodes a BP increasing agent. Among other approaches Suppressing C17QTL expression might be effective in lowering BP.
Molecular bases of genetic dominance: a threshold, or the presence or absence of a functional product
Mechanistically, genetic dominance has been interpreted, sufficiently although not completely (Keightley, 1996; Porteous, 2004), as a kinetic property of metabolic systems (Kacser and Burns, 1981). Although the molecular bases of dominance for the BP QTLs in question are not known, there are, in general, two paradigms: a threshold (Bhattacharyya et al., 1990) including haploinsufficiency and the presence (or absence) of a functional gene product (INGRAM, 1957).
Molecular bases of genetically partial-dominance: gene dosage or a hybrid complex
Regarding the C17QTL results (Figure 1c), it is reasonable to propose that a dosage effect could be the underlying molecular cause of the partial dominance, as has been found in the case of other genes. For example, the Rad18-2 gene is involved in DNA repair and mitotic recombination in yeast (Mayer and Goin, 1984). Heterozygotes are intermediate in their phenotype. The gene dosage effect is thought to account for this partial dominance (Mayer and Goin, 1984). Another possibility is that a mutation occurring in the coding region could produce a functionally abnormal product. When combined with the wild-type product, a hybrid complex could be formed and, consequently, show partial dominance. For example, a homeobox gene, HESX1, responsible for a mild form of septo-optic dysplasia, manifests partial dominance in heterozygotes (Thomas et al., 2001). It is the result of a reduction in DNA-binding activity in the hybrid complex of wild-type and mutant HESX1 products. Another example is the Mg chelatase mutations in barley seedlings (Hansson et al., 1999). When wild-type and mutant subunits were joined as a heterodimer in heterozygotes, the phenotype showed partial dominance.
Linkage versus congenic results
Of the 10 QTLs analyzed here, only five had previously been detected by linkage studies (Tables 1 and 2). The remaining five (Table 2) have been resolved only by congenic fine mapping (Charron et al., 2005a; Dutil et al., 2005). C1QTL, C16QTL, C10QTL and C2QTL showed partial dominance in linkage analyses, that is the BP of the heterozygotes fell between the two homozygotes. Only C17QTL exhibited dominance of the DSS allele.
In contrast to the extensive partial dominance suggested linkage analyses, our current results show that eight of the10 QTLs exhibit complete dominance (Figure 1). Two QTLs, C2QTL2 and C2QTL3, were previously interpreted as a locus with partial dominance and C2QTL1 was not previously detected (Deng et al., 1994b). We now infer that the DSS alleles for C2QTL2 and C2QTL3 exert opposite effects on BP, that for C2QTL2 being recessive, and that for C2QTL3 being dominant (Figure 1i and j). In addition, the C17QTL allele appears to show partial dominance, rather than the complete dominance suggested by linkage studies (Deng et al., 1994a).
The stark difference between the results of linkage and congenic studies can be explained by an obvious fact. In an F2 population, all animals are heterogeneous genetically, whereas in a congenic strain, all of them are homogeneous. Furthermore, linkage results are correlative in nature. They do not show a cause–effect relationship between a QTL and BP effect. In contrast, a congenic strain can be used to evaluate directly the effect of a chromosome fragment on a trait (i.e. BP in this study). Thus, a congenic strain harboring a QTL can be viewed as a ‘monogenic’ isolate without interference from other factors, such as undefined QTL–QTL interactions and regulatory elements (Charron et al., 2005b). As such, a congenic strain provides a more reliable test for Mendelian behavior than linkage analyses.
Over-abundance of QTLs needed to determine the polygenic BP trait
It should be noted that alleles from the normotensive LEW or MNS strain for all 10 QTLs studied in the current work lowered BP (Figure 1). Their cumulative effects exceeded 400 mm Hg (Figure 1). This value, however, cannot, and does not, reflect the true BP difference between the two parental strains, DSS and LEW (or MNS), which is around 88 mm Hg (Palijan et al., 2003b; Ariyarajah et al., 2004; Charron et al., 2005a, 2005b; Dutil et al., 2005). Apparently, the real physiological effects of the 10 QTLs do not combine additively. Epistasis between QTLs certainly plays an important role (Palijan et al., 2003a; Charron et al., 2005a, 2005b; Dutil et al., 2005) and genetic or epigenetic inhibition could be involved (Charron et al., 2005b).
Use of heterozygotes as a deductive functional test of QTLs
The search for differential gene expression has recently received considerable attention as a means of finding candidate genes for QTLs (Liang et al., 2003; McBride et al., 2003; Moujahidine et al., 2004; Garrett et al., 2005; Hubner et al., 2005; Yagil et al., 2005). It is easy to understand the tantalizing appeal of such an approach, because it generates gene candidates directly, rapidly and immediately, with apparent performance differentials. Although this approach has met with success in another field (Aitman et al., 1999), there are examples of false positives in the study of genetic hypertension. The SA gene is a case in point. It was first found to be differentially expressed in the kidneys, a prominent candidate organ for BP regulation, in comparisons between Wistar-Kyoto and spontaneously hypertensive rat strains. More promising was the fact that it was found later to co-segregate with BP in linkage analyses (Iwai et al., 1992; Samani et al., 1993). However, subsequent physical mapping has ruled it out: the SA gene was contained in congenic intervals, yet the congenic strains failed to show any BP effect (Hubner et al., 1999; St Lezin et al., 2000). Attempts to identify BP QTLs by gene profiling again proved to be disappointing in experiments based on the comparison of renal gene expression of congenic and DSS strains. In these cases, differentially expressed candidates fell outside the precisely defined QTL intervals in the congenic strains (Moujahidine et al., 2004; Garrett et al., 2005). Thus, they could not be the QTLs in question. These examples illustrate that such rapid screening approaches for identifying BP QTLs must go hand in hand with genetic verification (Pravenec et al., 2003; Deng, 2005). The present study suggests that the use of heterozygotes obtained from congenic strains could offer such corroboration.
Prospective molecular characterization of QTLs
Based on the dominance relationships for the nine out of 10 QTLs studied (Figure 1), genetic rescue experiments could conceivably be employed to restore the phenotype by transgenically introducing the dominant allele into the background of the recessive allele. In doing so, one could authenticate the function of the gene in question as a BP QTL (Deng, 2005). Although BP as a whole is a polygenic trait and there is epistasis between the QTLs, we interpret the combined data to mean that its individual genetic determinants consist of functionally integrated ‘monogenic’ components.
References
Abiola O, Angel JM, Avner P, Bachmanov AA, Belknap JK, Bennett B et al. (2003). The nature and identification of quantitative trait loci: a community's view. Nat Rev Genet 4: 911–916.
Aitman TJ, Glazier AM, Wallace CA, Cooper LD, Norsworthy PJ, Wahid FN et al. (1999). Identification of Cd36 (Fat) as an insulin-resistance gene causing defective fatty acid and glucose metabolism in hypertensive rats. Nat Genet 21: 76–83.
Ariyarajah A, Palijan A, Dutil J, Prithiviraj K, Deng Y, Deng AY (2004). Dissecting quantitative trait loci into opposite blood pressure effects on Dahl rat chromosome 8 by congenic strains. J Hypertens 22: 1495–1502.
Bhattacharyya MK, Smith AM, Ellis TH, Hedley C, Martin C (1990). The wrinkled-seed character of pea described by Mendel is caused by a transposon-like insertion in a gene encoding starch-branching enzyme. Cell 60: 115–122.
Bianchi G, Fox U, Imbasciati E (1974). The development of a new strain of spontaneously hypertensive rats. Life Sci 14: 339–347.
Caulfield M, Munroe P, Pembroke J, Samani N, Dominiczak A, Brown M et al. (2003). Genome-wide mapping of human loci for essential hypertension. Lancet 361: 2118–2123.
Charron S, Duong C, Menard A, Roy J, Eliopoulos V, Lambert R et al. (2005a). Epistasis, not numbers, regulates functions of clustered Dahl rat quantitative trait loci applicable to human hypertension. Hypertension 46: 1300–1308.
Charron S, Lambert R, Eliopoulos V, Duong C, Menard A, Roy J et al. (2005b). A loss of genome buffering capacity of Dahl salt-sensitive model to modulate blood pressure as a cause of hypertension. Hum Mol Genet 14: 3877–3884.
Dahl LK, Heine M, Tassinari L (1962). Role of genetic factors in susceptibility to experimental hypertension due to chronic excess salt ingestion. Nature 194: 480–482.
Darvasi A, Weinreb A, Minke V, Weller JI, Soller M (1993). Detecting marker-QTL linkage and estimating QTL gene effect and map location using a saturated genetic map. Genetics 134: 943–951.
Deng AY (1998). In search of hypertension genes in Dahl salt-sensitive rats. J Hypertens 16: 1707–1717.
Deng AY (2005). Functional genomics of blood pressure determination: dissecting and assembling a polygenic strait by experimental genetics. Curr Hypertens Rev 1: 35–50.
Deng AY, Dene H, Pravenec M, Rapp JP (1994a). Genetic mapping of two new blood pressure quantitative trait loci in the rat by genotyping endothelin system genes. J Clin Invest 93: 2701–2709.
Deng AY, Dene H, Rapp JP (1994b). Mapping of a quantitative trait locus for blood pressure on rat chromosome 2. J Clin Invest 94: 431–436.
Deng Y, Rapp JP (1992). Cosegregation of blood pressure with angiotensin converting enzyme and atrial natriuretic peptide receptor genes using Dahl salt-sensitive rats. Nat Genet 1: 267–272.
Deng AY, Rapp JP (1995). Locus for the inducible, but not a constitutive, nitric oxide synthase cosegregates with blood pressure in the Dahl salt-sensitive rat. J Clin Invest 95: 2170–2177.
Dutil J, Eliopoulos V, Tremblay J, Hamet P, Charron S, Deng AY (2005). Multiple quantitative trait loci for blood pressure interacting epistatically and additively on Dahl rat chromosome 2. Hypertension 45: 557–564.
Eliopoulos V, Dutil J, Deng Y, Grondin M, Deng AY (2005). Severe hypertension caused by alleles from normotensive Lewis for a quantitative trait locus on chromosome 2. Physiol Genom 22: 70–75.
Falconer DS, Mackey TFC (1996). Introduction to Quantitative Genetics, 4th edn. Longman: Essex, England.
Flint J, Valdar W, Shifman S, Mott R (2005). Strategies for mapping and cloning quantitative trait genes in rodents. Nat Rev Genet 6: 271–286.
Garrett MR, Dene H, Walder R, Zhang Q, Cicila GT, Assadnia S et al. (1998). Genomic scan and congenic strains for blood pressure quantitative trait loci using Dahl salt-sensitive rats. Genome Res 8: 711–723.
Garrett MR, Meng H, Rapp JP, Joe B (2005). Locating a blood pressure quantitative trait locus within 117 kb on the rat genome: substitution mapping and renal expression analysis. Hypertension 45: 451–459.
Grondin M, Eliopoulos V, Lambert R, Deng Y, Ariyarajah A, Moujahidine M et al. (2005). Complete and overlapping congenics proving the existence of a quantitative trait locus for blood pressure on Dahl rat chromosome 17. Physiol Genom 21: 112–116.
Hansson A, Kannangara CG, Von Wettstein D, Hansson M (1999). Molecular basis for semidominance of missense mutations in the XANTHA-H (42-kDa) subunit of magnesium chelatase. Proc Natl Acad Sci USA 96: 1744–1749.
Harrap SB (2003). Where are all the blood-pressure genes? Lancet 361: 2149–2151.
Hubner N, Lee YA, Lindpaintner K, Ganten D, Kreutz R (1999). Congenic substitution mapping excludes Sa as a candidate gene locus for a blood pressure quantitative trait locus on rat chromosome 1. Hypertension 34: 643–648.
Hubner N, Wallace CA, Zimdahl H, Petretto E, Schulz H, Maciver F et al. (2005). Integrated transcriptional profiling and linkage analysis for identification of genes underlying disease. Nat Genet 37: 243–253.
Ingram VM (1957). Gene mutations in human haemoglobin: the chemical difference between normal and sickle cell haemoglobin. Nature 180: 326–328.
Iwai N, Kurtz TW, Inagami T (1992). Further evidence of the SA gene as a candidate gene contributing to the hypertension in spontaneously hypertensive rat. Biochem Biophys Res Commun 188: 64–69.
Kacser H, Burns JA (1981). The molecular basis of dominance. Genetics 97: 639–666.
Keightley PD (1996). A metabolic basis for dominance and recessivity. Genetics 143: 621–625.
Liang M, Yuan B, Rute E, Greene AS, Olivier M, Cowley Jr AW (2003). Insights into Dahl salt-sensitive hypertension revealed by temporal patterns of renal medullary gene expression. Physiol Genom 12: 229–237.
Mayer VW, Goin CJ (1984). Semidominance of rad18-2 for several phenotypic characters in Saccharomyces cerevisiae. Genetics 106: 577–589.
Mcbride MW, Carr FJ, Graham D, Anderson NH, Clark JS, Lee WK et al. (2003). Microarray analysis of rat chromosome 2 congenic strains. Hypertension 41: 847–853.
Moujahidine M, Lambert R, Dutil J, Palijan A, Sivo Z, Ariyarajah A et al. (2004). Combining congenic coverage with gene profiling in search of candidates for blood pressure quantitative trait loci in Dahl rats. Hypertens Res 27: 203–212.
Palijan A, Dutil J, Deng AY (2003a). Quantitative trait loci with opposing blood pressure effects demonstrating epistasis on Dahl rat chromosome 3. Physiol Genom 15: 1–8.
Palijan A, Lambert R, Dutil J, Sivo Z, Deng AY (2003b). Comprehensive congenic coverage revealing multiple blood pressure quantitative trait loci on Dahl rat chromosome 10. Hypertension 42: 515–522.
Porteous JW (2004). A rational treatment of Mendelian genetics. Theor Biol Med Model 1: 6.
Pravenec M, Wallace C, Aitman TJ, Kurtz TW (2003). Gene expression profiling in hypertension research: a critical perspective. Hypertension 41: 3–8.
Rapp JP (2000). Genetic analysis of inherited hypertension in the rat. Physiol Rev 80: 135–172.
Rapp JP, Garret MR, Deng AY (1998). Construction of a double congenic strain to prove an epistatic interaction on blood pressure between rat chromosomes 2 and 10. J Clin Invest 101: 1591–1595.
Samani NJ, Lodwick D, Vincent M, Dubay C, Kaiser MA, Kelly MP et al. (1993). A gene differentially expressed in the kidney of the spontaneously hypertensive rat cosegregates with increased blood pressure. J Clin Invest 92: 1099–1103.
Snell GD (1948). Methods for the study of histocompatibility genes. J Genet 49: 87–103.
St Lezin E, Liu W, Wang JM, Yang Y, Qi N, Kren V et al. (2000). Genetic analysis of rat chromosome 1 and the Sa gene in spontaneous hypertension. Hypertension 35: 225–230.
Thomas PQ, Dattani MT, Brickman JM, McNay D, Warne G, Zacharin M et al. (2001). Heterozygous HESX1 mutations associated with isolated congenital pituitary hypoplasia and septo-optic dysplasia. Hum Mol Genet 10: 39–45.
Wilkie AO (1994). The molecular basis of genetic dominance. J Med Genet 31: 89–98.
Yagil C, Hubner N, Monti J, Schulz H, Sapojnikov M, Luft FC et al. (2005). Identification of hypertension-related genes through an integrated genomictranscriptomic approach. Circ Res 96: 617–625.
Zlotogora J (1997). Dominance and homozygosity. Am J Med Genet 68: 412–416.
Acknowledgements
This work was supported by a grant from the Canadian Institutes for Health Research (CIHR) to AY Deng. S Charron is supported by a fellowship from the Societé Québecoise d'hypertension arterielle. We thank Mr Ovid Da Silva, Research Support Office, Research Centre, CHUM, for his editorial assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Duong, C., Charron, S., Deng, Y. et al. Individual QTLs controlling quantitative variation in blood pressure inherited in a Mendelian mode. Heredity 98, 165–171 (2007). https://doi.org/10.1038/sj.hdy.6800920
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.hdy.6800920