Abstract
Germline mutations in the LKB1/STK11 tumour suppressor gene cause Peutz–Jeghers syndrome (PJS), a rare dominant disorder. In addition to typical hamartomatous gastrointestinal polyps and pigmented perioral lesions, PJS is associated with an increased risk of tumours at multiple sites. Follow-up information on carriers is limited and genetic heterogeneity makes counselling and management in PJS difficult. Here we report the analysis of the LKB1/STK11 locus in a series of 33 PJS families, and estimation of cancer risks in carriers and noncarriers. Germline mutations of LKB1/STK11 were identified in 52% of cases. This observation reinforces the hypothesis of a second PJS locus. In carriers of LKB1/STK11 mutations, the risk of cancer was markedly elevated. The risk of developing any cancer in carriers by age 65 years was 47% (95% CI: 27–73%) with elevated risks of both gastrointestinal and breast cancer. PJS with germline mutations in LKB1/STK11 are at a very high relative and absolute risk of multiple gastrointestinal and nongastrointestinal cancers. To obtain precise estimates of risk associated with PJS requires further studies of genotype–phenotype especially with respect to LKB1/STK11 negative cases, as this group is likely to be heterogeneous.
Similar content being viewed by others
Main
Peutz–Jeghers syndrome (PJS; MIM 175200) is an autosomal dominant disorder characterised by a specific form of hamartomatous polyposis of the gastrointestinal tract, and by melanin pigmentation of the lips, perioral region and buccal mucosa, fingers and toes, and other sites (Tomlinson and Houlston, 1997). Approximately three-quarters of PJS are familial, the remainder resulting from new mutations or low-penetrance variants. PJS typically presents in early childhood with pigmentation or with complications of small bowel polyps–intussusception, obstruction or bleeding.
Although PJS polyps are seen most commonly in the small bowel, they can occur throughout the gastrointestinal tract (Tomlinson and Houlston, 1997) and at other extra-intestinal sites such as the kidney, ureter, gall bladder, bronchus and nasal passage (Westerman et al, 1999; Sommerhaug et al, 1970; Wada et al, 1987). The polyps seen in PJS have a muscular core and are generally classified as being hamartomas. Nevertheless, adenomatous change may occur in polyps and they may become malignant, and an increased risk of jejunal and other small bowel tumours is recognised (Gruber et al, 1998).
In addition to an elevated risk of gastrointestinal malignancies, an increased risk of cancers at other sites is recognized; in particular, breast, pancreas, ovary, uterus, cervix, lung and testicular cancers have been reported (Giardello et al, 1987, 2000; Spigelman et al, 1989). Testicular sex cord and Sertoli-cell tumours may occur in prepubertal boys affected with PJS leading to sexual precocity and gynaecomastia (Wilson et al, 1986; Coen et al, 1991; Young et al, 1995). The production of oestrogen in ovarian tumours in girls with PJS has also been reported causing isosexual precocity (Sohl et al, 1983).
Germline mutations in the serine/threonine kinase gene (LKB1/STK11) on chromosome 19p13.3 have been shown to cause PJS (Hemminki et al, 1997; Hemminki et al, 1998; Jenne et al, 1998). This gene has a putative coding region of ∼1.3 kb, composed of nine exons, and functions as a tumour suppressor.
Previous studies have shown that between 30 and 82% of patients have no detectable germline mutations in LKB1/STK11 (Mehenni et al, 1998; Nakagawa et al, 1998; Jiang et al, 1999; Wang et al., 1999; Westerman et al, 1999b; Ylikorkala et al, 1999; Boardman et al, 2000; Yoon et al, 2000; Olschwang et al, 2001). Families with PJS unlinked to 19p13.3 have also been reported, suggesting that the disease is heterogeneous (Jiang et al, 1999; Westerman et al, 1999b; Yoon et al, 2000). Furthermore, a second PJS locus on chromosome 19q13.4 has been proposed on the basis of linkage in one family (Mehenni et al, 1998).
The clinical features of PJS are variable especially with respect to cancer risks. It is likely that inter- and intrafamilial differences in disease expression reflect in part the influence of different germline mutations.
To further our knowledge about the relation between genotype and cancer risk in PJS, we have related disease expression to LKB1/STK11 status in 33 families.
Patients and methods
Patients
Thirty-three index patients with PJS were ascertained through colorectal surgeons, gastroenterologists and geneticists within the UK. Clinical information was collected on all patients using a standard proforma and through access to patients' medical records. PJS was defined according to published diagnostic criteria (Giardello et al, 1987)–histopathologically verified hamartomatous polyps with at least two of the following: small bowel polyposis, mucocutaneous melanotic pigmentation and family history of the disease. Patients were asked to provide details of any cancer in first- and second-degree relatives. There was no selection of cases for a family history of cancer. Clinical information and samples were obtained with informed consent and Local Ethical Review Board approval in accordance with the tenets of the Declaration of Helsinki.
Mutational analysis of LKB1/STK11
Genomic DNA from PJS patients was isolated from EDTA venous blood samples using a standard sucrose lysis protocol. The search for germline mutations in LKB1/STK11 was performed using conformational sensitive gel electrophoresis (CSGE) as described by Ganguly et al (1993). Published oligonucleotide primer sequences were used to amplify each of the nine exons of LKB1/STK11 (Bignell et al, 1998). Any fragments showing migration shifts were reamplified and sequenced directly using the ABI Prism dRhodamine Terminator Cycle Sequencing Ready Reaction Kit and an ABI377 Genetic Analyser. For all samples with possible mutations, sequencing was replicated in forward and reverse orientation using an additional affected family member (or using the original patient if no other affected individual had been sampled) in order to confirm the presence of the mutation. A search for large-scale deletions in LKB1/STK11 was made by long-range PCR. Amplification of exons 3–8 of LKB1/STK11 was undertaken using the Expand Long Template PCR System (Roche Diagnostics, UK).
Nucleotide changes identified were coded according to the published sequence of LKB1/STK11 (Genbank accession numbers: exon 1, AF032984; exons 2–8, AF032985; exon 9, AF032986) and referenced to the Human Gene Mutation Database (http://archive.uwcm.ac.uk/uwcm/mg).
A search of the literature was made using the electronic database MEDLINE (National Library of Medicine, USA) for additional mutations reported to be associated with PJS which were not referenced in the Human Gene Mutation Database. LKB1/STK11 protein sequences of Homo sapiens (GenBank accession number NP 000446), Mus musculus (NP 035622) and XEEK1 (Q91604) were obtained from the NCBI protein database. They were aligned using Clustal W (1.82) multiple sequence alignment program (http://www.ebi.ac.uk/clustalw/).
Statistical analyses
Statistical analyses were performed using the statistical software program STATA Version 6 (Stata Corporation, TX, USA. http://www.stata.com). The 95% confidence interval (CI) of the estimate of the frequency of LKB1/STK11 mutations in PJS was estimated from the binomial distribution. The association between categorical variables was made using either Fisher or χ2 tests, and differences in the distribution of continuous variables were evaluated using the Mann–Whitney U-test. Estimation of cancer risks was made excluding cases that had developed neoplasms either before or at the time of presentation of PJS. Estimates of cancer risks were obtained from survival analyses and standardised mortality ratios (SMRs). SMRs for cancers were determined using life-table methods. Cases were considered at risk from age 5 until the date of diagnosis of cancer or date of ascertainment, censoring at age 65. Expected numbers of cancers were computed using age-, sex- and calendar period-specific mortality rates for England and Wales referenced to the International Classification of Diseases, ninth revision (ICD-9)–all cancers 140–208, cancers of the digestive organs and peritoneum 150–159 and breast carcinoma 174. Two-sided 95% CIs for relative risk estimates are based on the Poisson distribution. A P-value of 0.05 was considered statistically significant.
Results
Table 1 details the clinical characteristics and family histories of the 33 index patients analysed. Of these cases, 13 were familial and 20 sporadic. Germline LKB1/STK11 mutations were identified in 17 of the 33 (52%; 95% CI: 33–69%) patients (Table 1), in exons 1–8.
We cannot exclude the possibility that some mutations may have gone undetected; however, under test conditions, we have found that CSGE can detect all small insertions and deletions and ∼90% of single-base substitutions. In addition, we have examined for the possibility that some cases might harbour large-scale deletions in LKB1/STK11. It is therefore unlikely that we have failed to detect coding mutations, and, allowing for 90% sensitivity, the results suggest that the mutations in LKB1/STK11 account for at best 75% of PJS cases (the upper 95% confidence limit). Two patients carried the same mutation in exon 6 (PJ42 and PJ51) and two carried the same mutation at the 5′ splice site of exon 8 (PJ33 and PJ61). These four patients were ascertained from different centres and were not known to have any common ancestry. Nevertheless, as all are from the UK, it is probable that these mutations have a common origin, although identical LKB1/STK11 mutations without evidence of common ancestry have been reported (Hemminki et al, 1998; Resta et al, 1998; Wang et al, 1999; Westerman et al, 1999b; Ylikorkala et al, 1999). None of the patients studied were shown to harbour large-scale deletions of LKB1/STK11.
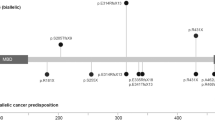
No significant bias towards mutations in exons 1 or 6 was observed, but no exon 9 mutations were identified. Seven of the 15 different mutations identified have not been reported previously–336delG (Q112fsX17), 369delG (Q123fsX6), 427delG (V143fsX144), 718_719insA (S240fsX26), G725A (G242E), 815_816insA (Y272X), IVS8-2A>G (altered splicing). In all, 11 of the mutations are predicted to lead to a truncated protein (four nonsense mutations, four frameshift deletions, one frameshift insertion and two splice site mutations). The other mutations identified were missense mutations, three of which have previously been reported to be pathogenic (Resta et al, 1998; Westerman et al, 1999b; Ylikorkala et al, 1999). All are nonconservative amino-acid changes that are highly conserved among human, mouse and Xenopus homologues of LKB1/STK11 and reside within the protein kinase core of LKB1/STK11 (Collins et al, 2000).
Table 2 shows the positions of the mutations observed in our study and in previously published reports (Hemminki et al, 1998; Gruber et al, 1989; Jenne et al, 1998; Mehenni et al, 1998; Nakagawa et al, 1998; Resta et al, 1998; Jiang et al, 1999; Kruse et al, 1999; Wang et al, 1999; Westerman et al, 1999b; Ylikorkala et al, 1999; Boardman et al, 2000; Miyaki et al, 2000; Yoon et al, 2000; Olschwang et al, 2001; Abed et al, 2001). Overall, most mutations reported to date have been frameshift or nonsense mutations and thus result in a truncated protein. In-frame deletions or missense mutations appear to occur less frequently generally at conserved amino acids in the kinase core.
Very few cases of PJS appear to be the consequence of large-scale deletions of LKB1/STK11; however, not all studies have systematically searched for such genetic changes (Table 2).
Disease expression in PJS is well documented to display inter- and intrafamilial variation (Burdick et al, 1982; Foley et al, 1998). Establishing a relationship between a number of the features of the disease and genotype is, however, inherently problematic because features typical of the disease are criteria for ascertainment. Nevertheless, there was no evidence that the ages at diagnosis are significantly different in carriers and noncarriers–mean ages of index cases, 13.9 y and 13.6 years, respectively. Furthermore, the distribution of polyps and rates of laparotomy were not significantly different between the groups.
Some previously reported studies have reported no association between detectable LKB1/STK11 mutation and family history (Hemminki et al, 1998; Wang et al, 1999; Ylikorkala et al, 1999). In our study, 13 of the 33 index cases had a family history of PJS (39%). Of these 10 were carriers of mutations in LKB1/STK11 (77%), but only seven (35%) patients with sporadic disease had mutations in LKB1/STK11. The higher prevalence of LKB1/STK11 mutations in PJS patients with a family history of the disease compared with sporadic cases is statistically significant (P=0.03).
Extra-gastrointestinal polyps are a recognised feature of PJS. Four of the patients in our study had extra-intestinal polyps: one of these harboured an LKB1/STK11 mutation and three did not.
Two patients had developed breast cancer since the diagnosis of PJS had been made–at ages 52 and 35 years. Both are carriers of an LKB1/STK11 mutation. In addition, one patient had presented at age 6 with a Sertoli–Leydig cell stromal tumour. He did not harbour an, LKB1/STK11 mutation. A high frequency of cancer was seen in the relatives of the familial cases–stomach (n=2, ages 32, 33 years), breast (n=2, ages 39, 51 years), colorectal (n=2, ages 43, 67 years), pancreas (n=1; age 50 years) and adenoma malignum of the cervix (n=1, age 43 years). All but the one case of stomach cancer was associated with LKB1/STK11 mutations. Excluding the case presenting with a Sertoli–Leydig cell tumour, the index cases and their relatives provided a total of 70 individuals with PJS from which cancer risks could be estimated. These individuals provided a total of 2120 years at risk.
The probability of developing cancer by age 65 years in all PJS patients was 37% (95% CI: 21–61%). The observation of seven cancer deaths, four from gastrointestinal disease, between ages 5 and 65 years, equates to the SMR for all cancer of 9.9 (95% CI: 0.4–20.4; P<0.001) and for gastrointestinal cancer of 24.8 (95% CI: 0.7–63.6; P<0.001). Confining the analysis to LKB1/STK11 mutation carriers, the probability of developing cancer by age 65 is 47% (95% CI: 27–73%), SMR of all and gastrointestinal cancers of 13.2 (95% CI: 0.5–27.1, P<0.001) and 32.0 (95% CI: 0.5–81.8, P<0.001), respectively. The risk of breast cancer in carriers was markedly increased, 29% by age 65 (95% CI: 12–62%); SMR, 13.9 (95% CI: 0.2–50.3, P<0.001).
Discussion
It is now well recognised that cancer risks are markedly elevated in PJS (Giardello et al, 1987, 2000; Spigelman et al, 1989). Diagnosing PJS in the absence of mutation data, especially in those without a prior family history of the disease, can however be difficult as pigmentation may not always be present or can fade and polyposis is not always an invariable feature. Moreover, there is substantial phenotypic overlap with other syndromes such as Carney complex (Stratakis et al, 1998).
Over 75% of LKB1/STK11 mutations reported have been frameshift or nonsense mutations and thus result in a truncated protein (Hemminki et al, 1998; Jenne et al, 1998; Mehenni et al, 1998; Nakagawa et al, 1998; Resta et al, 1998; Jiang et al, 1999; Kruse et al, 1999; Wang et al, 1999; Westerman et al, 1999b; Ylikorkala et al, 1999; Boardman et al, 2000; Miyaki et al, 2000; Yoon et al, 2000; Olschwang et al, 2001). In-frame deletions or missense mutations appear to occur less commonly at conserved amino acids within the kinase core of the expressed protein. Mutations reported to date have been scattered across exons 1–8. The distribution of mutations within the protein kinase core encoding region of LKB1/STK11 does not appear to be random (P<0.05) and exons 1 and 6 appear to be preferentially involved accounting for ∼38% of all reported mutations. Only one mutation has been described in exon 9 (Wang et al, 1999)–a nonsense mutation removing 56 residues from the protein of 434 amino acids and as such resides outside the protein kinase core. Although the case was familial, other members of the family were not evaluated and hence the pathological significance of this mutation is questionable.
Our study showed that the risk for cancer, gastrointestinal and breast, associated with germline LKB1/STK11 mutations is high and supports recent implementation of screening protocols suggested for patients (Wirtzfeld et al, 2001). In contrast to a number of other inherited cancer syndromes, cancer risks associated with germline LKB1/STK11 mutations cancer risks are not so site specific. LKB1/STK11 functions as a tumour suppressor in hamartomous polyps and in neoplasms. Some neoplasms develop from hamartomas; however, as LKB1/STK11 has a role in a number of pathways involved in control of cell growth, it is likely that some mutations may confer an increased cancer risk through alternative mechanisms.
In our study, cancers were found in association with mutations in most exons. From studies published so far, there does not seem to be a specifically higher prevalence of any cancer associated with mutations in specific exons (Figure 1). However, one of the mutations we detected, R304W, appeared to be associated with a high risk of breast cancer. It is highly conceivable that certain mutations may be associated with higher risks of cancer at certain sites, as seen with BRCA2 (The Breast Cancer Linkage Consortium 1999; Murphy et al, 2002). To formally assess such relationships will require a large number of observations.
Since Hemminki et al (1998) first showed that germline mutations in LKB1/STK11 cause PJS, a number of studies have examined the prevalence of mutations in the syndrome. In our study, we identified the LKB1/STK11 mutation in 52% of our patients, implying that approximately half of the cases are not caused by mutations in this gene, reinforcing the suggestion that the disease is genetically heterogeneous. Other studies have reported similar estimates for the prevalence of germline LKB1/STK11 mutations in PJS patients (Wang et al, 1999; Westerman et al, 1999b; Yoon et al, 2000; Olschwang et al, 2001). Some mutations may have gone undetected such as those in regulatory elements which may be undetectable in some PCR-based assays; however, families with PJS unlinked to 19p13.3 have been reported confirming that the disease is heterogeneous (Mehenni et al, 1998; Jiang et al, 1999; Westerman et al, 1999b; Yoon et al, 2000).
Studies that have formally estimated cancer risks in PJS have not computed separate estimates according to LKB1/STK11 status. Olschwang et al (2001) recently reported a high frequency of proximal bile duct adenocarcinomas in PJS who did not carry LKB1/STK11 mutations. Similarly, Boardman et al (2000) reported a high frequency of cancer in this group of patients, although no cases of bile duct cancers were observed. In our study, we had few familial cases not caused by LKB1/STK11 mutations to enable us to compute a separate estimate of risk for noncarriers.
In conclusion, our results confirm that there is significant genetic heterogeneity in PJS. Future studies characterising the mutational status and disease manifestation in large numbers of PJS patients will allow better genotype–phenotype correlation to be made, which should assist clinicians in formulating cancer surveillance and individual predictive genetic testing.
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Abed AA, Gunther K, Kraus C, Hohenberger W, Ballhausen WG (2001) Mutation screening at the RNA level of the STK11/LKB1 gene in the Peutz–Jeghers syndrome reveals complex splicing abnormalities and a novel mRNA isoform (STK11 c.597∧598insIVS4). Hum Mutat 18: 397–410
Bignell GR, Barfoot R, Seal S, Collins N, Warren W, Stratton MR (1998) Low frequency of somatic mutations in the LKB1/Peutz–Jeghers syndrome gene in sporadic breast cancer. Cancer Res 58: 1384–1386
Boardman LA, Couch FJ, Burgart LJ, Schwartz D, Berry R, McDonnell SK, Schaid DJ, Hartmann LC, Schroeder JJ, Stratakis CA, Thibodeau SN (2000) Genetic heterogeneity in Peutz–Jeghers syndrome. Hum Mut 16: 23–30
Burdick D, Prior JT (1982) Peutz–Jeghers syndrome. A clinicopathological study of a large family with a 27-year follow-up. Cancer 50: 2139–2146
Coen P, Kulin H, Ballantine T, Zaino R, Frauenhoffer E, Boal D, Inkster S, Brodie A, Santen R (1991) An aromatase-producing sex-cord tumor resulting in prepurbertal gynaecomastia. New Engl J Med 324: 317–322
Collins SP, Reoma JL, Gamm DM, Uhler MD (2000) LKB1, a novel serine/threonine protein kinase and potential tumour suppressor, is phosphorylated by cAMP-dependent protein kinase (PKA) and prenylated in vivo. Biochem J 345: 673–680
Foley TR, McGarrity TJ, Abt AB (1998) Peutz–Jeghers syndrome: a clinicopathological survey of the ‘Harrisburg family’ with a 49-year follow-up. Gastroenterology 95: 1535–1540
Ganguly A, Rock MJ, Prockop DJ (1993) Conformation-sensitive gel electrophoresis for rapid detection of single base differences in double stranded PCR products and DNA fragments: evidence for solvent induced bends in DNA heteroduplexes. Proc Natl Acad Sci USA 90: 10325–10329
Giardello FM, Brensinger JD, Tersmette AC, Goodman SN, Petersen GM, Booker SV, Cruz-Correa M, Offerhaus GJA (2000) Very high risk of cancer in familial Peutz–Jeghers syndrome. Gastroenterology 119: 1447–1453
Giardello FM, Welsh SB, Hamilton SR, Offerhaus GJA, Gittelsohn AM, Booker SV, Krush AJ, Yardley JH, Luk GD (1987) Increased risk of cancer in the Peutz–Jeghers syndrome. N Engl J Med 316: 1511–1514
Gruber SB, Entius MM, Petersen GM, Laken SJ, Longo PA, Boyer R, Levin AM, Mujumdar UJ, Trent JM, Kinzler KW, Vogelstein B, Hamilton SR, Polymeropoulos MH, Offerhaus GJ, Giardello FM (1998) Pathogenesis of adenocarcinoma in Peutz–Jeghers syndrome. Cancer Res 58: 5267–5270
Hemminki A, Markie D, Tomlinson I, Avizienyte E, Roth S, Loukola A, Bignell G, Warren W, Aminoff M, Hoglund P, Jarvinen H, Kristo P, Pelin K, Ridanpaa M, Salovaara R, Toro T, Bodmer W, Olschwang S, Olsen AS, Stratton MR, de la Chapelle A, Aaltonen LA (1998) A Serine/threonine kinase gene defective in Peutz–Jeghers syndrome. Nature 391: 184–187
Hemminki A, Tomlinson I, Markie D, Jarvinen H, Sistonen P, Bjorkqvist AM, Knuutila S, Salovaara R, Bodmer W, Shibata D, de la Chapelle A, Aaltonen LA (1997) Localization of a susceptibility locus for Peutz–Jeghers syndrome to 19p using comparative genomic hybridisation and targeted linkage analysis. Nat Genet 15: 87–90
Jenne DE, Reimann H, Nezu J, Friedel W, Loff S, Jeschke R, Muller O, Back W, Zimmer M (1998) Peutz–Jeghers syndrome is caused by mutations in a novel serine threonine kinase. Nat Genet 18: 38–43
Jiang CY, Esufali S, Berk T, Gallinger S, Cohen Z, Tobi M, Redston M, Bapat B (1999) STK11/LKB1 germline mutations are not identified in most Peutz–Jeghers syndrome patients. Clin Genet 56: 136–141
Kruse R, Uhlhaas S, Lamberti C, Keller KM, Jackisch C, Steinhard J, Knöpfle G, Loff S, Back W, Stolte M, Jungck M, Propping P, Friedl W, Jenne DE (1999) Peutz–Jeghers syndrome: four novel inactivating germline mutations in the STK11 gene. Hum Mut 13: 257–258
Mehenni H, Gehrig C, Nezu J, Oku A, Shimane M, Rossier C, Guex N, Blouin JL, Scott HS, Antonarakis SE (1998) Loss of LKB1 kinase activity in Peutz–Jeghers syndrome and evidence for allelic and locus heterogeneity. Am J Hum Genet 63: 1641–1650
Miyaki M, Iijima T, Hosono K, Ishii R, Yasuno M, Mori T, Toi M, Hishima T, Shitara N, Tamura K, Utsunomiya J, Kobayashi N, Kuroki T, Iwama T (2000) Somatic mutations of LKB1 and beta-catenin genes in gastrointestinal polyps from patients with Peutz–Jeghers syndrome. Cancer Res 60: 6311–6313
Murphy KM, Brune KA, Griffin C, Sollenberger JE, Petersen GM, Bansal R, Hruban RH, Kern SE (2002) Evaluation of candidate genes MAP2K4, MADH4, ACVR1B, and BRCA2 in familial pancreatic cancer. Cancer Res 62: 3789–3793
Nakagawa H, Koyama K, Miyoshi Y, Ando H, Baba S, Watatani M, Yasutomi M, Matsuura N, Monden M, Nakamura Y (1998) Nine novel germline mutations of STK11 in ten families with Peutz–Jeghers syndrome. Hum Genet 103: 168–172
Olschwang S, Boisson C, Thomas G (2001) Peutz–Jeghers families unlinked to STK11/LKB1 gene mutations are highly predisposed to primitive biliary adenocarcinoma. J Med Genet 38: 356–360
Resta N, Simone C, Mareni C, Montera M, Gentile M, Susca F, Gristina R, Pozzi S, Bertario L, Bufo P, Carlomagno N, Ingrosso M, Rossini FP, Tenconi R, Guanti G (1998) STK11/LKB1 mutations in Peutz–Jeghers syndrome and sporadic colon cancer. Cancer Res 58: 4799–4801
Sohl HM, Azoury RS, Najjar SS (1983) Peutz–Jeghers syndrome associated with precocious puberty. J Pediatr 103: 593–595
Sommerhaug RG, Mason T (1970) Peutz–Jeghers syndrome and ureteral polyposis. J Am Med Assoc 211: 120–122
Spigelman AD, Murday V, Phillips RK (1989) Cancer and the Peutz–Jeghers syndrome. Gut 30: 1588–1590
Stratakis CA, Kirschner LS, Taymans SE, Tomlinson IPM, Marsh DJ, TOrpy DJ, Giatzakis C, Eccles DM, Theaker J, Houlston RS, Blouin J-L, Antonarakis SE, Basson CT, Eng C, Carney JA (1998) Carney Complex, Peutz–Jeghers Syndrome, Cowden Disease, and Bannayan–Zonana Syndrome share cutaneous and endocrine manifestation, but not genetic loci. J Clin End Met 8: 2972–2976
The Breast Cancer Linkage Consortium (1999) Cancer risks in BRCA2 mutation carriers. JNCI 91: 1310–1316
Tomlinson IPM, Houlston RS (1997) Peutz–Jeghers syndrome. J Med Genet 34: 1007–1011
Wada K, Tanaka M, Yamaguchi K (1987) Carcinoma and polyps of the gallbladder associated with Peutz–Jeghers syndrome. Dig Dis Sci 32: 943–946
Wang Z-J, Churchman M, Avizienyte E, McKeown C, Davies S, Evans DGR, Ferguson A, Ellis I, Xu W-H, Yan Z-Y, Aaltonen LA, Tomlinson IPM (1999) Germline mutations of the LKB1 (STK11) gene in Peutz–Jeghers patients. J Med Genet 36: 365–368
Westerman AM, Entius MM, Boor PPC, Koole R, de Baar E, Offerhaus GJA, Lubinski J, Lindhout D, Halley DJJ, de Rooij FWM, Wilson JHP (1999) Novel mutations in the LKB1/STK11 gene in Dutch Peutz–Jeghers families. Hum Mut 13: 476–481
Westerman AM, Wilson JHP (1999) Peutz–Jegher syndrome: risks of a hereditary condition. A clinical review. Scand J Gastro S230: 64–70
Wilson DM, Pitts WC, Hintz RL, Rosenfeld RG (1986) Testicular tumors with Peutz–Jeghers syndrome. Cancer 57: 2238–2240
Wirtzfeld DA, Petrelli NJ, Rodriguez-Bigas MA (2001) Hamartomatous polyposis syndrome: molecular genetics, neoplastic risk, and surveillance recommendations. Ann Surg Oncol; 8: 319–327
Ylikorkala A, Avizienyte E, Tomlinson IPM, Tiainen M, Roth S, Loukola A, Hemminki A, Johansson M, Sistonen P, Markie D, Neale K, Phillips R, Zauber P, Twama T, Sampson J, Jarvinen H, Makela TP, Aaltonen LA (1999) Mutations and impaired function of LKB1 in familial and non familial Peutz–Jeghers syndrome and a sporadic testicular cancer. Hum Mol Genet 8: 45–51
Yoon K-A, Ku J-L, Choi HS, Heo SC, Jeong S-Y, Park YJ, Kim NK, Kim JC, Jung PM, Park J-G (2000) Germline mutations of the gene in Korean Peutz–Jeghers syndrome patients. Br J Cancer 82: 1403–1406
Young S, Gooneratne S, Straus II FH, Zeller WP, Bulun SE, Rosenthal IM (1995) Feminizing Sertoli cell tumors in boys with Peutz–Jeghers syndrome. Am J Surg Path 19: 50–58
Acknowledgements
Funding for this work was undertaken with support from Cancer Research UK. W Lim was in receipt of a grant from the Epsom Hospital NHS Trust Gastroenterology R & D fund. We are grateful to the patients who participated in this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Lim, W., Hearle, N., Shah, B. et al. Further observations on LKB1/STK11 status and cancer risk in Peutz–Jeghers syndrome. Br J Cancer 89, 308–313 (2003). https://doi.org/10.1038/sj.bjc.6601030
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6601030
Keywords
This article is cited by
-
Progress report: Peutz–Jeghers syndrome
Familial Cancer (2024)
-
A novel missense mutation of the STK11 gene in a Chinese family with Peutz-Jeghers syndrome
BMC Gastroenterology (2022)
-
AMPK activation by ASP4132 inhibits non-small cell lung cancer cell growth
Cell Death & Disease (2021)
-
Low-level parental mosaicism in an apparent de novo case of Peutz–Jeghers syndrome
Familial Cancer (2019)
-
An exploration of genotype-phenotype link between Peutz-Jeghers syndrome and STK11: a review
Familial Cancer (2018)