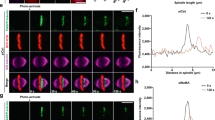
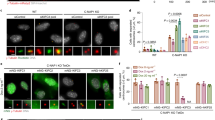
Abstract
Meiotic and mitotic spindles are required for the even segregation of duplicated chromosomes to the two daughter cells. The mechanism of spindle assembly is not fully understood, but two have been proposed that are not mutually exclusive1,2,3. The ‘search and capture’ model suggests that dynamic microtubules become progressively captured and stabilized by the kinetochores on chromosomes, leading to spindle assembly3,4. The ‘local stabilization’ model proposes that chromosomes change the state of the cytoplasm around them, making it more favourable to microtubule polymerization2,5,6,7,8,9. It has been shown10,11 that Stathmin/Op18 inhibits microtubule polymerization in vitro by interaction with tubulin12, and that overexpression in tissue culture cells of non-phosphorylatable mutants of Stathmin/Op18 prevents the assembly of mitotic spindles13. We have used Xenopus egg extracts and magnetic chromatin beads14 to show that mitotic chromatin induces phosphorylation of Stathmin/Op18. We have also shown that Stathmin/Op18 is one of the factors regulated by mitotic chromatin that governs preferential microtubule growth around chromosomes during spindle assembly.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Waters, J. C. & Salmon, E. D. Pathways of spindle assembly. Curr. Opin. Cell Biol. 9, 37–43 (1997).
Hyman, A. A. & Karsenti, E. Morphogenetic properties of microtubules and mitotic spindle assembly. Cell 84, 401 –411 (1996).
Nicklas, R. B. How cells get the right chromosomes. Science 275, 632–637 (1997).
Kirschner, M. W. & Mitchison, T. J. Beyond self assembly: from microtubules to morphogenesis. Cell 45, 329– 342 (1986).
Marek, L. F. Control of spindle form and function in Grasshopper spermatocytes. Chromosoma 68, 367–398 ( 1978).
Karsenti, E., Newport, J. & Kirschner, M. Respective roles of centrosomes and chromatin in the conversion of microtubule arrays from interphase to metaphase. J. Cell Biol. 99, 47s–57s (1984).
Nicklas, R. B. & Gordon, G. W. The total length of spindle microtubules depends on the number of chromosomes present. J. Cell Biol. 100, 1–7 (1985).
Zhang, D. & Nicklas, R. B. The impact of chromosomes and centrosomes on spindle assembly as observed in living cells. J. Cell Biol. 129, 1287–1300 (1995).
Zhang, D. & Nicklas, R. B. Chromosomes initiate spindle assembly upon experimental dissolution of the nuclear envelope in grasshopper spermatocytes. J. Cell Biol. 131, 1125– 1131 (1995).
Sobel, A. Stathmin: a relay phosphoprotein for multiple signal transduction? Trends Biochem. Sci. 16, 301–305 (1991).
Belmont, L., Mitchison, T. & Deacon, H. W. Catastrophic revelations about Op18/stathmin. Trends Biochem. Sci. 21, 197–198 (1996).
Belmont, L. D. & Mitchison, T. J. Identification of a protein that interacts with tubulin dimers and increases the catastrophe rate of microtubules. Cell 84, 623–631 (1996).
Marklund, U., Larsson, N., Gradin, H. M., Brattsand, G. & Gullberg, M. Oncoprotein 18 is a phosphorylation-responsive regulator of microtubule dynamics. EMBO J. 15, 5290–5298 (1996).
Heald, R. et al . Self-organization of microtubules into bipolar spindles around artificial chromosomes in Xenopus egg extracts. Nature 382, 420–425 (1996).
Maucuer, A., Moreau, J., Mechali, M. & Sobel, A. Stathmin gene family: phylogenetic conservation and developmental regulation in Xenopus. J. Biol. Chem. 268, 16420–16429 (1993).
Mayer-Jaekel, R. E. & Hemmings, B. A. Protein phosphatase 2A — a ‘ménage à trois’. Trends Cell Biol. 4, 287–291 ( 1994).
Tournebize, R. et al . Distinct roles of PP1 and PP2A-like phosphatases in control of microtubule dynamics during mitosis. EMBO J.(in the press).
Félix, M. A., Cohen, P. & Karsenti, E. Cdc2 H1 kinase is negatively regulated by a type 2A phosphatase in the Xenopus early embryonic cell cycle: evidence from the effects of okadaic acid. EMBO J. 9, 675– 683 (1990).
Larsson, N., Melander, H., Marklund, U., Osterman, O. & Gullberg, M. G2/M transition requires multisite phosphorylation of oncoprotein 18 by two distinct protein kinase systems. J. Biol. Chem. 270, 14175– 14183 (1995).
Sawin, K. E. & Mitchison, T. J. Poleward microtubule flux in mitotic spindles assembled in vitro. J. Cell Biol. 112, 941–954 (1991).
Walczak, C. E., Mitchison, T. J. & Desai, A. XKCM1: a Xenopus kinesin-related protein that regulates microtubule dynamics during mitotic spindle assembly. Cell 84, 37–47 ( 1996).
Samanta, A. & Greene, M. I. Akinase associated with chromatin that can be activated by ligand-p185c-Neu or epidermal growth factor-receptor interactions. Proc. Natl Acad. Sci. USA 92, 6582–6586 (1995).
Ookata, K. et al . Cyclin B interaction with microtubule-associated protein 4 (MAP4) targets p34CdC2 kinase to microtubules and is a potential regulator of M-phase microtubule dynamics. J. Cell Biol. 128, 849–862 (1995).
Masson, D. & Kreis, T. E. Binding of E-MAP-115 to microtubules is regulated by cell cycle-dependent phosphorylation. J. Cell Biol. 131, 1015–1024 ( 1995).
Andersen, S. S. L., Buendia, B., Domínguez, J. E., Sawyer, A. & Karsenti, E. Effect on microtubule dynamics of XMAP230, a microtubule-associated protein present in Xenopus laevis eggs and dividing cells. J. Cell Biol. 127, 1289–1299 (1994).
Vernos, I. et al . Xklp1, a chromosomal Xenopus kinesin-like protein essential for spindle organization and chromosome positioning. Cell 81, 117–127 (1995).
Lombillo, V. A., Nislow, C., Yen, T. J., Gelfand, V. I. & McIntosh, J. R. Antibodies to the kinesin motor domain and CENP-E inhibit microtubule depolymerizaiton-dependent motion of chromosomes in vitro . J. Cell Biol. 128, 107– 115 (1995).
Félix, M. A., Clarke, P., Coleman, J., Verde, F. & Karsenti, E. in The Cell Cycle: a Practical Approach (eds Fantes, P. & Brooks, R.) 253–283 (IRL, New York, (1994)).
Shamu, C. E. & Murray, A. W. Sister chromatin separation in frog egg extracts requires DNA topoisomerase II activity during anaphase. J. Cell Biol. 117, 921– 934 (1992).
Curmi, P. A. et al. The Stathmin tubulin interaction in vitro. J. Biol. Chem.(in the press).
Acknowledgements
We thank D. Chrétien, K. Dejgaard, J. Domínguez, R. Heald, J. Howard, T. J. Mitchison, I. T. Möst, A. Nebreda, M. Way, T. Wittmann and M. Zerial for discussions and reagents.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Andersen, S., Ashford, A., Tournebize, R. et al. Mitotic chromatin regulates phosphorylation of Stathmin/Op18. Nature 389, 640–643 (1997). https://doi.org/10.1038/39382
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/39382
This article is cited by
-
Combining optogenetics with sensitive FRET imaging to monitor local microtubule manipulations
Scientific Reports (2020)
-
Mitotic spindle assembly in animal cells: a fine balancing act
Nature Reviews Molecular Cell Biology (2017)
-
Stathmin 1 is a potential novel oncogene in melanoma
Oncogene (2013)
-
Involvement of PKCζ and GSK3β in the stability of the metaphase spindle
In Vitro Cellular & Developmental Biology - Animal (2012)
-
50 ways to build a spindle: the complexity of microtubule generation during mitosis
Chromosome Research (2011)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.