Abstract
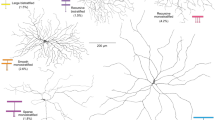
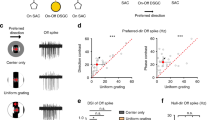
Directionally selective retinal ganglion cells respond strongly when a stimulus moves in their preferred direction, but respond little or not at all when it moves in the opposite direction1,2. This selectivity represents a classic paradigm of computation by neural microcircuits, but its cellular mechanism remains obscure. The directionally selective ganglion cells receive many synapses from a type of amacrine cell termed ‘starburst’ because of its regularly spaced, evenly radiating dendrites3,4. Starburst amacrine cells have a synaptic asymmetry that has been proposed as the source of the directional response in the ganglion cells5,6. Here we report experiments that make this unlikely, and offer an alternative concept of the function of starburst cells. We labelled starburst cells in living retinas, then killed them by targeted laser ablation while recording from individual directionally selective ganglion cells. Ablating starburst cells revealed no asymmetric contribution to the ganglion cell response. Instead of being direction discriminators, the starburst cells appear to potentiate generically the responses of ganglion cells to moving stimuli. The origin of direction selectivity probably lies with another type of amacrine cell.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Barlow, H. B. & Levick, W. R. The mechanism of directionally selective units in rabbit's retina. J. Physiol. (Lond.) 178, 477–504 (1965).
Amthor, F. R. & Grzywacz, N. M. in Visual Motion and its Role in the Stabilizaiton of Gaze (eds Miles, F. A. & Wallman, J.) (Elsevier, New York, (1993)).
Famiglietti, E. V. J Synaptic organizaiton of starburst amacrine cells in rabbit retina: analysis of serial thin sections by electron microscopy and graphic reconstruction. J. Comp. Neurol. 309, 40–70 (1991).
Masland, R. H. & Tauchi, M. The cholinergic amacrine cell. Trends Neurosci. 9, 218–223 (1986).
Vaney, D. I. The mosaic of amacrine cells in the mammalian retina. Prog. Retin. Res. 9, 1–28 (1991).
Borg-Graham, L. J. & Grzywacz, N. M. in Single Neuron Computation (eds McKenna, T., Davis, J. & Zornetzer, S. F.) 347–376 (Academic, New York, (1992)).
Masland, R. H. & Ames, A. II Responses to acetylcholine of ganglion cells in an isolated mammalian retina. J. Neurophysiol. 39, 1220–1235 (1976).
Ariel, M. & Daw, N. W. Pharmacological analysis of directionally selective rabbit retinal ganglion cells. J. Physiol. (Lond.) 324, 161–185 (1982).
Brecha, N., Johnson, D., Peichl, L. & Wässle, H. Cholinergic amacrine cells of the rabbit retina contain glutamate decarboxylase and gamma-aminobutyrate immunoreactivity. Proc. Natl Acad. Sci. USA 85, 6187–6191 (1988).
O'Malley, D. M., Sandell, J. H. & Masland, R. H. Co-release of acetylcholine and GABA by the starburst amacrine cells. J. Neurosci. 12, 1394–1408 (1992).
Masland, R. H., Mills, J. W. & Hayden, S. A. Acetylcholine-synthesizing amacrine cells: identification and selective staining by using autoradiography and fluorescent markers. Proc. R. Soc. Lond. B 223, 79–100 (1984).
Yang, G. & Masland, R. H. Receptive fields and dendritic structure of directionally selective retinal ganglion cells. J. Neurosci. 14, 5267–5280 (1994).
Sulston, J. E. & White, J. G. Regulation and cell autonomy during postembryonic development of Caenorhabditis elegans. Dev. Biol. 78, 577–597 (1980).
Bargmann, C. I. & Avery, L. Laser killing of cells in Caenorhabditis elegans. Methods Cell. Biol. 48, 225–250 (1995).
Jacobs, G. A., Miller, J. P. & Murphey, R. K. Integrative mechanisms controlling directional sensitivity of an identified sensory interneuron. J. Neurosci. 6, 2298–2311 (1986).
Grzywacz, N. M. & Amthor, F. R. Facilitation in ON–OFF directionally selective ganglion cells of the rabbit retina. J. Neurophysiol. 69, 2188–2199 (1993).
Marchiafava, P. L. The responses of retinal ganglion cells to stationary and moving visual stimuli. Vision Res. 19, 1203–1211 (1979).
Torre, V. & Poggio, T. Asynaptic mechanism possibly underlying directional selectivity to motion. Proc. R. Soc. Lond. 202, 409–416 (1978).
Cohen, E. D. & Miller, R. F. Quinoxalines block the mechanism of directional selectivity in ganglion cells of the rabbit retina. Proc. Natl Acad. Sci. USA 92, 1127–1131 (1995).
Kittila, C. A. & Massey, S. C. The pharmacology of directionally selective ganglion cells in the rabbit retina. J. Neurophysiol. 77, 675–689 (1997).
Ariel, M. & Daw, N. W. Effects of cholinergic drugs on receptive field properties of rabbit retinal ganglion cells. J. Physiol. (Lond.) 324, 135–160 (1982).
Caldwell, J. H., Daw, N. W. & Wyatt, H. J. Effects of picrotoxin and strychnine on rabbit retinal ganglion cells: lateral interactions for cells with more complex receptive fields. J. Physiol. (Lond.) 276, 277–298 (1978).
Vardi, N., Masarachia, P. H. & Sterling, P. Structure of the starburst amacrine network in the cat retina and its association with alpha ganglion cells. J. Comp. Neurol. 288, 601–611 (1989).
Schmidt, M., Humphrey, M. F. & Wässle, H. Action and localization of acetylcholine in the cat retina. J. Neurophysiol. 58, 997–1015 (1987).
Strettoi, E. & Masland, R. H. The number of unidentified amacrine cells in the rabbit's retina. Proc. Natl Acad. Sci. USA 93, 14906–14911 (1996).
Ames, A. I. & Nesbett, F. B. In vitro retina as an experimental model of the central nervous system. J. Neurochem. 37, 867–877 (1996).
Huba, R., Schneider, H. & Hofmann, H. D. Voltage-gated currents of putative GABAergic amacrine cells in primary cultures and in retinal slice preparations. Brain Res. 577, 10–18 (1992).
Tauchi, M. & Masland, R. H. The shape and arrangement of the cholinergic neurons in the rabbit retina. Proc. R. Soc. Lond. 223, 101–119 (1984).
Masland, R. H. & Mills, J. W. Autoradiographic identification of acetylcholine in the rabbit retina. J. Cell Biol. 83, 159–178 (1979).
Acknowledgements
We thank M. Chiang for help in developing the laser system, and S. Wallenstein for preparing cultured cells.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
He, S., Masland, R. Retinal direction selectivity after targeted laser ablation of starburst amacrine cells. Nature 389, 378–382 (1997). https://doi.org/10.1038/38723
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/38723
This article is cited by
-
Dendro-dendritic cholinergic excitation controls dendritic spike initiation in retinal ganglion cells
Nature Communications (2017)
-
Chi-Ju-Di-Huang-Wan protects rats against retinal ischemia by downregulating matrix metalloproteinase-9 and inhibiting p38 mitogen-activated protein kinase
Chinese Medicine (2016)
-
Direction selectivity in the retina: symmetry and asymmetry in structure and function
Nature Reviews Neuroscience (2012)
-
How neurons compute direction
Nature (2002)
-
Directionally selective calcium signals in dendrites of starburst amacrine cells
Nature (2002)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.