Abstract
Some investigators consider small-volume prostate cancer (0.5 ml or less) without Gleason pattern 4/5 elements as clinically insignificant. The objective of this study was to characterize the anatomic distribution and pathologic features of small tumors (aggregate volume of 0.5 ml or less) in whole-mount prostatectomy specimens. Between 1999 and 2003, 371 consecutive patients underwent radical prostatectomy at the Indiana University Hospitals for localized prostate cancer. Patients who received hormonal or radiation therapy prior to the surgery were excluded from the study. A total of 62 specimens with total tumor volume of 0.5 ml or less were identified and included in this study. All specimens were embedded and whole-mounted. Tumor volume was measured using the grid method. The mean age at the time of surgery was 59 years (median, 61 years; range, 37–72 years). The mean preoperative prostate-specific antigen (PSA) was 6.5 ng/ml (range: 0.3–18 ng/ml). The mean prostate weight was 53 g (range: 16–132 g). The mean tumor volume was 0.29 ml (median, 0.35 ml; range, 0.02–0.48 ml). Tumor multifocality and bilaterality were present in 69 and 37% of cases, respectively. Three (5%) had positive surgical margins. The largest tumor was located in the peripheral zone, transitional zone, and central zone in 79, 16, and 5% of cases, respectively. The largest tumor was located in the anterior prostate in 10 cases (16%) and in the posterior prostate in 52 cases (84%). The distribution of Gleason scores was 5 (12 cases, 19 %), 6 (40 cases, 65 %), and 7 (10 cases, 16 %). One case had a primary Gleason pattern 4. None had extraprostatic extension, seminal vesicle invasion, or lymph node metastasis. Small-volume prostate cancers are often multifocal and bilateral, with predilection for the peripheral zone. Of these small-volume cases, 16% had Gleason pattern 4 and might, therefore, be clinically significant.
Similar content being viewed by others
Main
Prostate cancer is the most commonly diagnosed cancer in men in the United States.1 With widespread screening for prostate-specific antigen (PSA) supplemented with transrectal ultrasound guided systematic sextant needle biopsy, many more prostate cancers have been diagnosed. In all, 30–40% of men older than 50 years have prostate cancer, but only 8% of cancers become clinically significant.2 Small-volume prostate cancers (0.5 ml or less) without elements of Gleason grade pattern 4 or 5 are not thought to be clinically significant.2, 3, 4, 5, 6, 7, 8, 9, 10 However, tumor volume and other parameters, especially Gleason score, are closely related. Since many prostate cancer deaths occur more than 10 years after the initial diagnosis,11 the biological behavior of small-volume prostate cancer, which some consider to be clinically insignificant, may become important in patients with a relatively long postdiagnosis life expectancy. In a study of 223 patients with initially untreated early-stage prostate cancer, Johansson et al11 found a significant acceleration in the recurrence and mortality rates after 15 years of follow-up. It is not known how these findings relate to small-volume prostate cancers or cancers detected by PSA screening; however, the pathologic characterization of small-volume prostate cancer may help to clarify these issues.
Recognition of clinically significant prostate cancer is important for patient management. Extensive studies have been performed to identify clinically significant cancer using multiple clinical and pathologic parameters, including biopsy information, tumor volume, serum PSA level, PSA velocity and density, free/total PSA ratio, and patient's age at diagnosis.3, 4, 12, 13 Some authors consider tumor volume to be the single most important factor in predicting cancer progression.14, 15 As there is little information about the anatomic distribution and pathological features of small-volume prostate cancer, we sought, in this study, to characterize these parameters in small-volume tumors (total volume 0.5 ml or less) in whole-mount prostatectomy specimens.
Materials and methods
From 1999 to 2003, 371 patients underwent radical prostatectomy for clinically localized prostate cancer at the Indiana University Hospital. A total of 62 patients with total tumor volume of 0.5 ml or less were identified and included in this study. None had hormonal or radiation treatment prior to the surgery. Serum PSA was determined using the DPC chemiluminescent assay (Diagnostic Products Corp., Los Angeles, California, USA). This research was approved by the Indiana University Institutional Review Board.
Each radical prostatectomy specimen was totally embedded and processed by the whole-mount method as previously described.16, 17 Each prostate was weighed, measured, inked, and fixed in 10% buffered neutral formalin. After fixation, the apex and base were amputated and serially sectioned at approximately 3–5 mm intervals in the vertical parasagittal plane. The seminal vesicles were sectioned parallel to the junction of the prostate and submitted entirely for examination. The remaining prostate was serially sectioned perpendicular to the long axis of the gland from the apex of the prostate to the tip of the seminal vesicle, and whole-mount blocks were prepared. All slides were reviewed by a single urologic pathologist (LC) without knowledge of patient information. Total cancer volume was determined by the grid method and was the sum of the volume of individual cancer foci.18, 19, 20 Grading was performed according to the Gleason system.21 Surgical margins were considered positive if cancer cells were in contact with the inked margin.22 The 1997 TNM (tumor, node, metastasis) system was used for pathologic staging.23 The 1997 TNM criteria were used rather than the 2002 criteria because of the extreme rarity of 2002 pT2b tumors. In our previous study, we found that a true pT2b tumor probably does not exist.24
Results
Of the 371 patients who underwent radical prostatectomy for localized prostate cancer at the Indiana University Hospital between 1999 and 2003, 62 patients (17%) had small-volume prostate cancer (tumor volume <0.5 ml). Patient characteristics are shown in Table 1. The mean age at the time of surgery was 59 years, ranging from 37 to 72 years. The mean preoperative serum PSA level was 6.5 ng/ml (range: 0.3–18 ng/ml). Seven patients (11%) had preoperative serum PSA level >10 ng/ml.
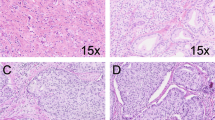
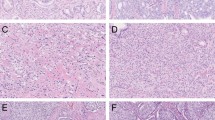
All the 62 specimens were totally embedded and whole-mounted. The mean prostate mass was 49 g, ranging from 16 to 122 g. The mean tumor volume was 0.29 ml (median, 0.35 ml; range, 0.02–0.48 ml). In all, 20 specimens had tumor volumes of less than 0.2 ml, and 42 specimens had volumes of 0.2–0.5 ml. The tumors were often multifocal (69%) and bilateral (37%) (Figure 1). Three (5%) had positive surgical margins (two in the left posterior and one in the right posterior region of the prostate), probably due to capsular incision, since none of these patients had extraprostatic extension. The largest tumor was located in the peripheral zone, transitional zone, and central zone in 79, 16, and 5% of cases, respectively. The largest tumor was located in the anterior prostate in 10 cases (16%) and in the posterior prostate in 52 cases (84%). The distribution of Gleason scores was 5 (12 cases, 19%), 6 (40 cases, 65%), and 7 (10 cases, 16%). One case had a primary Gleason pattern 4; nine had a secondary Gleason pattern 4. None of the cases had Gleason pattern 5 components. The mean tumor volume for the 12 tumors with a Gleason score of 5 is 0.32 ml (median, 0.39 ml; range, 0.06–0.46 ml). The mean tumor volume for the 40 tumors with a Gleason score of 6 was 0.26 ml (median, 0.31 ml; range, 0.02–0.48 ml). The mean tumor volume for the tumors with a Gleason score of 7 was 0.43 ml (median, 0.45 ml; range, 0.35–0.48 ml). None had extraprostatic extension, seminal vesicle invasion, or lymph node metastasis.
Discussion
Tumor volume has been considered to be one of the most important prognostic factors for prostate cancer. The clinical significance of small-volume (less than 0.5 ml) prostate cancer is controversial, although some consider small-volume cancer to be either clinically insignificant or minimally significant. In our study, we showed that even in small-volume prostate cancer, some specimens already had some pathological features indicative of a poor prognosis. Ten specimens (16%) contained Gleason pattern 4 components.
Prostate cancer is a progressive disease. As the tumor volume increases, so does the malignant potential. In 1986, McNeal et al25 found that only patients with large-volume prostate cancer (larger than 4 ml) with a Gleason pattern of 4 or 5 metastasized. Several other studies also found that metastasis was more likely to occur in patients with a large volume of prostate cancer.14, 26 Nevertheless, factors other than tumor volume must also be considered. According to the mathematical model proposed by Dugan et al,12 the cancer volume doubling time, patient's age, and Gleason score all must be taken into account in addition to tumor volume. Preoperative PSA levels and the anatomic distribution of the cancer are other variables that should be considered.
In general, the concept of tumor doubling time and its accurate measurement for prostate cancer are controversial issues. The tumor doubling time is difficult to predict. Several studies have suggested that it is between several months and 4 years.27, 28 If the tumor volume doubling time were three years, patients 60 years or younger with tumor volumes of 0.5 ml might be likely to develop clinically significant prostate cancer based on the average life expectancy. If tumors have higher Gleason scores, the potential for metastasis may increase and these patients may present with clinically significant prostate cancer sooner.12 Johansson et al11 studied 223 patients with early, localized prostate cancer who were managed by watchful waiting rather than undergoing definitive treatment at the time of diagnosis. These patients were followed for a mean observation period of 21 years. Most had an indolent course for the first 10 to 15 years of follow-up; however, there was a significant decrease in cumulative progression-free survival, survival without metastasis, and prostate cancer-specific survival during the 15–21 year follow-up period. Although tumor volume could not be assessed in this study, approximately half were clinically stage T0. In our patient population, about one-third of these patients are less than 60 years old. The vast majority are 65 years old or less (76%). Those small high-grade cancers detected in the current study would most likely progress over time. Considering the young age of our patient population, it is reasonable to predict that some patients may develop recurrent disease after radical prostatectomy. It is not uncommon for small prostate cancers to arise de novo as high grade lesions. Epstein et al29 examined 720 individual tumor foci and showed only a weak correlation between tumor volume and Gleason grade. Of 106 peripheral tumors with some Gleason pattern 4 or 5 components, 48 (45%) were less than 1 ml in volume. Therefore, prostate cancer can exhibit invasive potential when relatively small. For Gleason scores of 6 or below in small-volume cancers, the tumor's natural history is more unpredictable.
Eradication of cancer in its early stage offers the best chance for reducing cancer morbidity and mortality. Our findings may have important implications for future cancer prevention and detection strategies. We found that the majority of small-volume prostate cancers are multifocal, often involving both sides of the prostate. These tumors are located predominantly in the peripheral zone (79%) and posterior aspect (84%) of the prostate. These data suggest that prostatic carcinogenesis may be attributed to a field effect, supported by recent molecular evidence that multiple prostate cancers arise independently.30, 31 Our finding of frequent multifocality and bilaterality in small-volume prostate cancer is important for this reason. The fact that these patients have developed multifocal tumors even with a very low total tumor volume suggests that the field cancerization mechanism of carcinogenesis is already in effect30, 31 and that other tumors will likely arise in the future if these cancers are left untreated. Based on our findings and those of Epstein et al,29 it is not unreasonable to speculate that some of these future tumors might contain high-grade foci. It is also assumed that existing small-volume tumors will themselves grow in size and progress with time if left untreated. Using the current American Joint Committee on Cancer (AJCC) TNM staging classification,32 the small-volume prostate cancers in the current study with bilateral foci would be classified as pT2c. This relatively high stage for such a low-volume cancer highlights one of the major areas of controversy and uncertainty with the current staging system, the subclassification of pT2 tumors. In a recent study of a large series of whole-mount prostatectomy specimens, we could not identify any tumors that fulfilled the criteria for classification as a pT2b tumor (occupying greater than one-half of a single lobe without involvement of the other lobe), leading the authors to question whether this subclassification of prostate tumor (pT2b) actually exists.24 Djavan et al33 showed statistically significant differences between those patients with unifocal and multifocal disease with respect to preoperative PSA density of the transition zone, free/total PSA, as well as percentage of patients with organ confined disease. Multifocal prostate cancer has been shown to be associated with higher grade, stage, and recurrence rate than unifocal prostate cancer and may be a predictor of cancer recurrence and, therefore, of cancer-specific survival.33
While we acknowledge that the current study could be enhanced with either survival or PSA follow-up data, the postoperative follow-up period is of insufficient length for meaningful conclusions to be drawn. In addition, even with a long follow-up period, the natural course of these small-volume cancers could not be assessed as each patient in our study underwent definitive treatment (radical prostatectomy) rather than taking a more conservative management approach. It will, however, be important to compare outcomes among the patients with and without Gleason pattern 4 elements once adequate follow-up can be achieved. Recent studies suggest that the presence of high-grade components (Gleason pattern 4 or 5 tumor) may ultimately dictate the final clinical outcome in prostate cancer patients who underwent radical prostatectomy for cure.34, 35, 36 The purpose of this study was to analyze the pathologic characteristics and anatomic distribution of small-volume prostate cancers. We can only speculate as to how our findings of frequent multifocality and bilaterality and the finding of occasionally high Gleason grades will impact actual clinical behavior.
In summary, small-volume prostate cancers are often multifocal and bilateral, with predilection for the peripheral zone. Of these small-volume cancers, 16% had high Gleason grades and might be considered clinically significant if left untreated. Nonetheless, long-term clinical follow-up is warranted to determine the outcome differences among patients with and without small-volume prostate cancer.
References
Jemal A, Murray T, Ward E, et al. Cancer statistics, 2005. CA Cancer J Clin 2005;55:10–30.
Stamey TA, Freiha FS, McNeal JE, et al. Localized prostate cancer. Relationship of tumor volume to clinical significance for treatment of prostate cancer. Cancer 1993;71:933–938.
Epstein JI, Chan DW, Sokoll LJ, et al. Nonpalpable stage T1c prostate cancer: prediction of insignificant disease using free/total prostate specific antigen levels and needle biopsy findings. J Urol 1998;160:2407–2411.
Goto Y, Ohori M, Arakawa A, et al. Distinguishing clinically important from unimportant prostate cancers before treatment: value of systematic biopsies. J Urol 1996;156:1059–1063.
Irwin MB, Trapasso JG . Identification of insignificant prostate cancers: analysis of preoperative parameters. Urology 1994;44:862–867.
Epstein JI, Walsh PC, Carmichael M, et al. Pathologic and clinical findings to predict tumor extent of nonpalpable (stage T1c) prostate cancer. JAMA 1994;271:368–374.
Chan TY, Chan DY, Stutzman KL, et al. Does increased needle biopsy sampling of the prostate detect a higher number of potentially insignificant tumors? J Urol 2001;166:2181–2184.
Noguchi M, Stamey TA, McNeal JE, et al. Relationship between systematic biopsies and histological features of 222 radical prostatectomy specimens: lack of prediction of tumor significance for men with nonpalpable prostate cancer. J Urol 2001;166:104–109.
Elgamal AA, Van Poppel HP, Van de Voorde WM, et al. Impalpable invisible stage T1c prostate cancer: characteristics and clinical relevance in 100 radical prostatectomy specimens—a different view. J Urol 1997;157:244–250.
Augustin H, Hammerer PG, Graefen M, et al. Insignificant prostate cancer in radical prostatectomy specimens: time trends and preoperative prediction. Eur Urol 2003;43:455–460.
Johansson JE, Andren O, Andersson SO, et al. Natural history of early, localized prostate cancer. JAMA 2004;291:2713–2719.
Dugan JA, Bostwick DG, Myers RP, et al. The definition and preoperative prediction of clinically insignificant prostate cancer. JAMA 1996;275:288–294.
Cheng L, Poulos CK, Pan C, et al. Preoperative prediction of small volume cancer (<0.5 ml) in radical prostatectomy specimens. J Urol 2005 (in press).
Stamey TA, McNeal JE, Freiha FS, et al. Morphometric and clinical studies on 68 consecutive radical prostatectomies. J Urol 1988;139:1235–1241.
Bostwick DG, Graham Jr SD, Napalkov P, et al. Staging of early prostate cancer: a proposed tumor volume-based prognostic index. Urology 1993;41:403–411.
Poulos CK, Daggy JK, Cheng L . Preoperative prediction of Gleason grade in radical prostatectomy specimens: the influence of different Gleason grades from multiple positive biopsy sites. Mod Pathol 2005;18:228–234.
Emerson RE, Koch MO, Daggy J, et al. Closest distance between tumor and resection margin in radical prostatectomy specimens: lack of prognostic significance. Am J Surg Path 2005;29:224–229.
Cheng L, Bergstralh EJ, Cheville JC, et al. Cancer volume of lymph node metastasis predicts progression in prostate cancer. Am J Surg Pathol 1998;22:1491–1500.
Humphrey PA, Vollmer RT . Intraglandular tumor extent and prognosis in prostatic carcinoma: application of a grid method to prostatectomy specimens. Hum Pathol 1990;21:799–804.
Eichelberger LE, Koch MO, Daggy JK, et al. Predicting tumor volume in radical prostatectomy specimens from patients with prostate cancer. Am J Clin Pathol 2003;120:386–391.
Gleason DF, Mellinger GT . Prediction of prognosis for prostatic adenocarcinoma by combined histologic grading and clinical stage. J Urol 1974;111:58–64.
Cheng L, Slezak J, Bergstralh EJ, et al. Preoperative prediction of surgical margin status in prostate cancer patients treated by radical prostatectomy. J Clin Oncol 2000;18:2862–2868.
Fleming ID, Cooper JS, Henson DE, et al. American Joint Committee on Cancer Staging Manual. Lippincott Raven: Philadelphia, 1997, pp 219–222.
Eichelberger LE, Cheng L . Does pT2b prostate carcinoma exist? Critical appraisal of the 2002 TNM classification of prostate carcinoma. Cancer 2004;100:2573–2576.
McNeal JE, Bostwick DG, Kindrachuk RA, et al. Patterns of progression in prostate cancer. Lancet 1986;1:60–63.
Villers AA, McNeal JE, Redwine EA, et al. Pathogenesis and biological significance of seminal vesicle invasion in prostatic adenocarcinoma. J Urol 1990;143:1183–1187.
D'Amico AV, Hanks GE . Linear regressive analysis using prostate-specific antigen doubling time for predicting tumor biology and clinical outcome in prostate cancer. Cancer 1993;72:2638–2643.
Schmid HP, McNeal JE, Stamey TA . Observations on the doubling time of prostate cancer. The use of serial prostate-specific antigen in patients with untreated disease as a measure of increasing cancer volume. Cancer 1993;71:2031–2040.
Epstein JI, Carmichael MJ, Partin AW, et al. Small high grade adenocarcinoma of the prostate in radical prostatectomy specimens performed for nonpalpable disease: pathogenetic and clinical implications. J Urol 1994;151:1587–1592.
Bostwick DG, Shan A, Qian J, et al. Independent origin of multiple foci of prostatic intraepithelial neoplasia: comparison with matched foci of prostate carcinoma. Cancer 1998;83:1995–2002.
Cheng L, Song SY, Pretlow TG, et al. Evidence of independent origin of multiple tumors from patients with prostate cancer. J Natl Cancer Inst 1998;90:233–237.
Greene FL, Page DL, Flemming ID, et al. American Joint Committee on Cancer Staging Manual. Springer-Verlag: New York, 2002, pp 337–346.
Djavan B, Susani M, Bursa B, et al. Predictability and significance of multifocal prostate cancer in the radical prostatectomy specimen. Tech Urol 1999;5:139–142.
Pan C, Potter SR, Partin AW, et al. The prognostic significance of tertiary grade Gleason patterns of higher grade in radical prostatectomy specimens: a proposal to modify the Gleason grading system. Am J Surg Pathol 2000;24:563–569.
Cheng L, Koch MO, Daggy JK, et al. The combined percentage of Gleason pattern 4 and 5 is the best predictor of cancer progression after radical prostatectomy. J Clin Oncol (in press).
Stamey T, McNeal J, Yemoto C, et al. Biological determinants of cancer progression in men with prostate cancer. JAMA 1999;281:1395–1400.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cheng, L., Jones, T., Pan, CX. et al. Anatomic distribution and pathologic characterization of small-volume prostate cancer (<0.5 ml) in whole-mount prostatectomy specimens. Mod Pathol 18, 1022–1026 (2005). https://doi.org/10.1038/modpathol.3800431
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.3800431
Keywords
This article is cited by
-
Single-center, prospective phase 2 trial of high-intensity focused ultrasound (HIFU) in patients with unilateral localized prostate cancer: good functional results but oncologically not as safe as expected
World Journal of Urology (2023)
-
Posterior subcapsular prostate cancer: identification with mpMRI and MRI/TRUS fusion-guided biopsy
Abdominal Imaging (2015)
-
Re-evaluating the concept of “dominant/index tumor nodule” in multifocal prostate cancer
Virchows Archiv (2014)
-
MRI-Guided Biopsy for Prostate Cancer Detection: A Systematic Review of Current Clinical Results
Current Urology Reports (2013)