Abstract
Mature B-cell acute lymphoblastic leukemia (ALL) is typically associated with the FAB-L3 morphology and rearrangement of the MYC gene, features characteristic of the leukemic phase of Burkitt's lymphoma. However, the term ‘mature’ has also been used to describe other rare cases of B-ALL with light-chain surface immunoglobulin expression. In contrast, infantile B-cell ALL is generally characterized by rearrangement of the MLL gene, an immature pro-B-cell phenotype, and CD10 negativity. We describe two unusual cases of infantile B-ALL with non-L3 morphology, expressing a mature B-cell phenotype (λ sIg+, CD19+, CD10−, TdT−, and CD34−), and showing MLL rearrangement without MYC rearrangement at presentation. Both infants relapsed after months of morphologic and genetic remission. At relapse, the t(9;11) translocation was detected in both cases by spectral karyotyping. After the initial relapse, both cases followed a rapid and aggressive course. Literature search identified few similar cases, all expressed λ surface immunoglobulin and showed MLL rearrangement (majority with the t(9;11) translocation). These cases show that B-ALL with MLL rearrangement, especially the t(9;11) translocation, can express a ‘mature’ B-cell phenotype and may represent a distinct subset. Identification of additional cases will further clarify the significance of MLL rearrangements in mature B-ALL.
Similar content being viewed by others
Main
Acute lymphoblastic leukemia (ALL) is a heterogeneous group of hematopoietic neoplasms. Precursor B-ALL, characterized by FAB-L1 or L2 morphology and surface immunoglobulin (sIg)−, TdT+, CD34+ immature B-cell phenotype,1, 2 comprises 90% of ALL. The less common mature B-ALL, characterized by a FAB-L3 morphology and sIg+ and TdT− phenotype, is usually associated with MYC gene rearrangements and represents the leukemic phase of Burkitt's lymphoma. However, rare cases of B-ALL with non-FAB-L3 morphology and without MYC rearrangements have also been reported in both adult and pediatric patients as ‘mature B-ALL’.3, 4, 5, 6, 7, 8, 9 These cases may or may not express TdT and CD34, but uniformly express sIg to some degree.
Infantile B-ALL, generally a precursor B-ALL, has a poor prognosis and is frequently associated with rearrangement of the MLL gene10, 11, 12 through different genetic aberrations involving chromosome 11q23. Reciprocal translocations are most common and the following are most common in decreasing order of frequency: t(4;11)(q21;q23), t(11;19)(q23;p13.3) and t(9;11)(p21–22;q23).10, 11, 12 MLL gene rearrangements are seen in both B-ALL and T-cell ALL, and in acute myelogenous leukemia (AML), especially DNA-topoisomerase inhibitor therapy-related AML.13, 14
Infantile B-ALL with MLL gene rearrangements or MLL+ precursor B-ALL has typically a CD10−, CD20−, CD34+, TdT+, and HLA-DR+ pro-B-cell immunophenotype. In addition, myeloid and natural-killer cell antigens, such as CD15 and CD56, can frequently be expressed.12 This is in contrast to B-ALL in infants without MLL gene rearrangement, which frequently expresses CD10. In gene expression profile studies, MLL+ precursor B-ALL shows a profile consistent with an early hematopoietic progenitor that is distinct from conventional B-ALL and AML, suggesting that MLL+ precursor B-ALL is a clinically and molecularly unique entity.15
Among the cases of mature B-ALL of non-FAB-L3 morphology reported in children, rare cases contain MLL gene rearrangements. Of the cases studied, all have been reported as the t(9;11) translocation. We describe two additional cases of MLL+ mature B-ALL in infants, both showing a mature B-cell phenotype with λ sIg+, CD19+, CD10−, CD34−, and TdT− and containing a t(9;11) translocation. These cases show that MLL+ B-ALL, especially those with a t(9;11) translocation, can express a mature B-cell phenotype and may represent a distinct subset of B-ALL. Identification of additional cases will further clarify the significance of MLL rearrangements in mature B-ALL.
Materials and methods
Morphology Assessment
Bone marrow aspirate smear slides were air dried and stained with Wright–Giemsa stain. Bone marrow biopsies were fixed in Bouin's solution and then decalcified. Paraffin-embedded tissue sections were stained with hematoxylin and eosin for routine histomorphology.
Immunophenotyping by immunohistochemistry
Immunohistochemical analysis (IHC) was performed on formalin-fixed and paraffin-embedded sections using the Dako Envision plus system for detection (DAKO, Carpinteria, CA, USA). Immunostains included CD20, CD79a, and Bcl-6 (DAKO, Carpinteria, CA, USA), CD10 (Novocastra, Burlingame, CA, USA), Bcl-2 (Biogenex, San Ramon, CA, USA), TdT (Supertechs, Bethesda, MD, USA) and c-MYC (Santa Cruz Biotechnology, Santa Cruz, CA, USA).
Immunophenotyping by flow cytometry
Immunophenotyping was performed by flow cytometric analysis (FACScan; Becton Dickinson, San Diego, CA, USA) using the Cell Quest software (Becton Dickinson) on bone marrow aspirate and peripheral blood samples collected in ethylene diamine tetraacetic acid (EDTA) according to standardized procedures. Antibodies included CD2, CD3, CD4, CD5, CD7, CD8, CD10, CD11c, CD13, CD14, CD16+56, CD19, CD20, cCD22, CD33, CD34, CD45, CD64, CD79a, CD117, TdT, MPO, Kappa, Lambda, and cIgM.
Cytogenetic Analysis
Conventional G-banded chromosome analysis was performed by standard methods, and the karyotype was described according to ISCN 1995.16
Fluorescence in situ hybridization (FISH) and spectral karyotype (SKY) analysis
FISH was performed using LSI MLL dual color break apart and LSI C-MYC probes obtained from VYSIS (Downers Grove, IL, USA) and hybridized by standard methods. Hybridization signals were scored on Nikon Eclipse 600 microscope attached to CytoVision imaging system (Applied Imaging, Santa Clara, CA, USA). SKY was performed on metaphase preparations using human SKYPaint kit obtained from Applied Spectral Imaging (Carlsbad, CA, USA) according to the manufacturer's protocol. SKY images were acquired with SD200 Spectra cube mounted on Nikon Eclipse 800 microscope by using SKY optical filter (Chroma Technology, Brattleboro, VT, USA) analyzed using the SKY View software.
Molecular Analysis
Molecular analysis performed on the JH region of the Ig heavy chain gene (JH) and T-cell receptor gamma (TCR γ) gene using polymerase chain reaction (PCR)-heteroduplex analysis with polyacrylamide gel electrophoresis with the FRIIIB and JH-B primers and V1 V9, V10/11, J1/2, JP, and JP1/P2 primers, respectively, as reported by Sioutos et al17 and Bottaro et al,18 respectively.
Case reports
Case #1
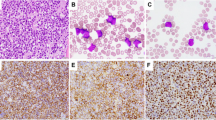
The patient was a 4-month-old male who presented with a patchy, erythematous raised macular rash, multiple subcutaneous nodules on the scalp and trunk, massive hepatosplenomegaly and bilateral testicular enlargement. At admission, the patient had leukocytosis (33 200/ml, reference range 5000–16 000/ml) and thrombocytopenia (136 000/ml, reference range 165 000–415 000/ml). Peripheral blood smears showed small to medium-sized blast cells with high nuclear:cytoplasmic ratio, variably irregular nuclei with fine chromatin and inconspicuous nucleoli, and scanty blue nonvacuolated cytoplasm consistent with a FAB non-L3 morphology (Figure 1a). Bone marrow, skin, and testicular biopsies showed extensive disease. No central nervous system involvement was present at this time. The patient was treated according to the Interfant-99 Protocol (vincristine, daunorubicin, prednisone, L-asparaginase, and low-dose Ara-C followed by alternating high-dose methotrexate with high-dose Ara-C) with localized radiation for the testicular involvement. Rapid response (significant decreases in leukocyte count to <500/ml and resolution of organomegaly) to prephase and induction chemotherapy was seen. Complete remission was documented with follow-up bone marrow biopsies, flow cytometric studies, and cytogenetic studies including FISH analyses. At 5 months after initial diagnosis, the patient relapsed with central nervous system, soft tissue and bone marrow involvement. Reinduction therapy included vincristine, idarubicin, L-asparaginase, and dexamethasone with intrathecal Ara-C, hydrocortisone, and methotrexate, and localized radiation for soft-tissue disease. However, the patient did not respond and the disease progressed with increasing leukocytosis (peak 79 000/ml). Despite additional courses of mitoxantrone, Ara-C, and monoclonal anti-CD52 antibodies, the patient died 11 months after diagnosis.
Case #2
The patient was an 8-month-old girl presenting with high fevers, fatigue, pallor, massive hepatosplenomegaly, generalized lymphadenopathy, and a diffuse rash with several subcutaneous nodules. At admission, the patient had marked leukocytosis (160 900/ml) and thrombocytopenia (19 000/ml). Peripheral blood smears showed numerous blasts with non-FAB-L3 similar to the previous case. Bone marrow and skin involvement were documented on biopsies. The patient was pretreated with steroids and then placed on the Interfant-99 Protocol. The patient responded with rapid reduction in peripheral leukocyte count and resolution of the organomegaly, lymphadenopathy, and skin lesions. Complete remission was documented on bone marrow biopsies, flow cytometric studies and cytogenetic studies including FISH analyses. Subsequently, the patient received a 4/6 HLA-matched allogeneic cord blood transplant. However, 65 days after receiving the unrelated allogeneic cord blood transplant, the patient had recurrent acute leukemia, approximately 6 months after initial diagnosis and died soon after from respiratory failure associated with progressive disease.
Results
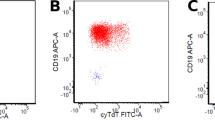
Immunophenotypic analysis by IHC and flow cytometry performed on bone marrow aspirates/biopsies and peripheral blood showed similar findings in both patients. Blast cells were found to be CD45+ (weakly), CD19+, CD20+/− (variable), cytoplasmic CD22+, cytoplasmic CD79a+, cytoplasmic IgM+, λ sIg+, and Bcl-2+ (Figure 2a–c and f). CD10, TdT, and CD34 were negative (Figure 2c–e). All other markers performed by flow cytometry and IHC, including all myelomonocytic markers, were negative.
Scatter plots of three-color flow cytometric immunophenotyping showing CD45 dim leukemic blasts located in R2 gate (a). Gated scatter plots showing leukemic blasts with CD19, λ surface immunoglobulin light chain, CD20 and cytoplasmic μ heavy chain expression and lack of CD10, CD34, and TdT expression (b–f).
Molecular analysis of the JH and TCR γ genes showed monoclonal rearrangement of the JH and polyclonal rearrangement of the TCR γ in both cases. Both patients had normal karyotypes by conventional karyotyping, but FISH analyses using an MLL probe detected MLL translocations. No evidence of MYC rearrangements was present at this time.
In both patients, the recurrent leukemic blasts had a different immunophenotype from the original. In the first patient, recurrent blast cells showed monocytic morphology (Figure 1b) and phenotype (weak CD45+, CD14+, CD64+, HLA-DR+, CD33+, CD13+, CD11c+, and negative for all B-cell markers). However, analysis of the JH gene by PCR and DNA sequencing showed monoclonal JH rearrangement identical to the initial leukemia. In addition, MLL rearrangement continued to be present by FISH analyses. SKY analysis identified the following karyotype: 46,XY,t(9;11)(p21;q23)[3]/46,XY,der(1)t(1;8)(p36.3;q13),t(9;11)(p21;q23)[16]/46,XY[1] (Figure 3a). The additional translocation der(1)t(1;8)(p36.3;q13) identified resulted in a partial trisomy of 8q, an additional copy of the MYC gene (confirmed by FISH), and overexpression of MYC protein by IHC. In contrast, the initial lymphoblastic cells did not overexpress the MYC protein by IHC. In the second patient, blast cells lost expression of λ sIg and gained expression of the CD2 and CD7 T-cell antigens. Interestingly, molecular analysis showed monoclonal JH rearrangement identical to the original leukemia and a monoclonal TCR γ rearrangement not present in the initial leukemia. SKY analysis identified a very complex karyotype that included a t(9;11) translocation in all cells examined (Figure 3b).
Discussion
The term ‘mature B-ALL’ has been used in the literature to describe rare cases of B-ALL, other than Burkitt's lymphoma, characterized by expression of pan-B-cell markers and sIg with light-chain restriction. 9 These cases can show non-FAB-L3 morphology with sIg expression and MYC rearrangement,7, 8, 9 L3 morphology with sIg expression but no MYC rearrangement,19 and rare cases showing sIg expression with non-FAB-L3 morphology and no MYC rearrangement.3, 4, 5, 6, 7, 8, 9 Some of these sIg+ B-ALL cases may even express TdT and CD343, 4, 9 and may represent an intermediate differentiation stage between the pre-B-cell and mature B-cell.20 These cases should probably be more appropriately named ‘surface immunoglobulin-positive B-ALL.’
The most common cytogenetic abnormalities associated with mature B-ALL are translocations involving the MYC gene, including t(8;14), t(2;8), and t(8;22).8 Rare cases with additional or other cytogenetic abnormalities have also been reported, including t(14;18) and t(8;9)(q24;p13);21 t(8;14)(q24;q32) and −15,der(1;15)(p10;q10);22 t(1;19) and t(14;18);23, 24 and even normal karyotype.19 Cytogenetic abnormalities involving the MLL gene at 11q23 have very rarely been reported in mature B-ALL. Literature review reveals four cases of MLL+ mature B-ALL.3, 4, 5, 6 Two additional cases are identified, one of ALL and the other of lymphoblastic lymphoma25 (both presented at an international meeting, cases #106 and #139, Society for Hematopathology/European Association of Hematopathology workshop, Pediatric Hematopathology, Memphis, TN, USA).
We describe two additional cases of MLL+ mature B-ALL in infants, both with non-FAB-L3 morphology, showing the typical sIg+, CD10-, CD34-, and TdT- mature phenotype and no MYC rearrangement. MLL rearrangement was detected by FISH analysis in both cases. The presence of a t(9;11) translocation was demonstrated by SKY analysis upon recurrence in the first case, and by both conventional karyotyping and SKY analysis upon recurrence in the second case. Clinically, the first case followed the typical MLL+ precursor B-ALL course with multiple relapses resulting in early death of the patient. Similarly, the second case eventually relapsed after bone marrow transplantation, also resulting in early death.
Interestingly, all MLL+ mature B-ALL (including ours, 8/8) express λ-restricted sIg and all cases studied (6/6) contain the t(9:11) translocation (Table 1). In two cases, the underlying translocation was not reported. The predominance of the t(9;11) translocation and λ-isotype in MLL+ mature B-ALL cases is very peculiar, however, the reason is not quite clear. Predominance of λ-isotype has also been reported in MLL-negative mature B-ALL of non-FAB-L3 morphology.7, 8 However, the t(9;11) translocation, although the most common 11q23 chromosomal abnormality associated with de novo AML with monocytic differentiation (FAB M4 and M5) and DNA-topoisomerase inhibitor therapy-related AML, is only rarely seen in precursor B-ALL.10, 11, 12, 26, 27
Although the exact fusion gene in our cases was not determined, studies of the t(9;11) translocation in AML most commonly results in a fusion of the MLL and AF9 genes.26 Additional studies have suggested that the resultant MLL/AF9 fusion gene is involved in myeloproliferation13 and leukemogenesis.26, 27, 28 Double knock in mice expressing the t(9;11) fusion gene develop predominantly AML with only a minority displaying features of ALL.27 Studies of the MLL-AF9 genomic breakpoint junction in AML, DNA-topoisomerase inhibitor therapy-related AML and ALL have suggested that the fusion point within the AF9 gene may influence the phenotype of the resulting leukemia.26 However, the exact mechanism of leukemogenesis is still unclear, as is its role in MLL+ mature B-ALL.
In our first case, the original mature B-ALL-phenotype switched into a morphologically and phenotypically monocytic lineage in subsequent relapses. PCR and DNA sequencing of the JH gene showed identical clonal rearrangement to the initial lymphoblastic leukemia. In addition, the MLL rearrangement was retained and shown to be a t(9;11) translocation by SKY. These findings suggest a leukemic transformation rather than the presence of two leukemic lineages. The phenomenon of lineage switching has been previously reported in cases of MLL+ precursor B-ALL that also switched to a monoblastic leukemia.29 The presence of bipotential B-macrophage progenitor cells in adult bone marrow30, 31 and that MLL+ B-ALL shows gene expression profiles consistent with early hematopoietic progenitors15 may provide some explanation for these lineage switching events. The additional t(1;8) translocation, subsequently identified by conventional karyotyping, FISH and SKY analysis, resulted in an additional copy of MYC gene with subsequent overexpression of MYC protein demonstrated by IHC. However, neither MYC protein overexpression nor the t(1;8) translocation was present in the initial MLL+ mature B-ALL, suggesting that this translocation is secondary to tumor progression.
In our second case, the leukemic cells changed immunophenotype upon recurrence, losing λ sIg expression and acquiring the CD2 and CD7 antigens. Molecular analysis by PCR showed similar JH rearrangement to the original leukemic cells, and an additional clonal rearrangement of the TCR γ gene, not initially present. However, the CD2 and CD7 antigens are not lineage specific and can be expressed in other leukemias, especially myeloid leukemias. In addition, rearrangement of the TCR γ gene is also not always indicative of a T-cell lineage, since it is also reported in other acute leukemias.
Clinically, MLL+ precursor B-ALL is typically associated with a poor prognosis due to recurrent relapses. Similarly, MLL+ mature B-ALL also shows a poor prognosis (4/7) with a tendency for multiple relapses (3/7) (Table 2). Of the three cases achieving complete remission, one is reported in abstract form where follow-up information is not included, one has only 4 months of follow-up, and one is reported as a precursor B-lymphoblastic lymphoma with 3.5 years of follow-up (Table 2).
In summary, we present two cases of MLL+ mature B-ALL in infants with the typical findings of non-FAB-L3 morphology, expression of monoclonal λ sIg, and 11q23 translocation involving the MLL gene. Six other similar cases have been previously reported. Interestingly, all cases analyzed show the presence of the t(9;11) translocation and express monoclonal λ sIg. The majority of cases appear to have a poor prognosis. The significance and the reason for the predominance of the t(9;11) translocation and the exclusive expression of λ-isotype sIg in MLL+ mature B-ALL is unclear. However, the consistent association between the t(9;11) translocation and the sIg+ mature phenotype suggests that mature B-ALL with t(9;11) is a distinct subset of MLL+ B-ALL. Additional cases need to be identified and studied to further clarify the significance of the t(9;11) translocation in ALL of mature B-cell phenotype.
References
Bene MC, Castoldi G, Knapp W et al. Proposals for the immunological classification of acute leukemias. European Group for the Immunological Characterization of Leukemias (EGIL). Leukemia 1995;9:1783–1786.
Rubnitz JE, Behm FG, Downing JR . 11q23 rearrangements in acute leukemia. Leukemia 1996;10:74–82.
Behm FG, Head DR, Pui C-H et al. B-precursor ALL with unexpected expression of surface immunoglobulin (sIg) mu and lambda (abstract). Lab Invest 1995;72:106A.
Lorenzana AN, Rubin CM, Le Beau MM et al. Immunoglobulin gene rearrangements in acute lymphoblastic leukemia with the 9;11 translocation. Genes Chromosomes Cancer 1991;3:74–77.
Talmant P, Berger R, Robillard N et al. Childhood B-cell acute lymphoblastic leukemia with FAB-L1 morphology and a t(9;11) translocation involving the MLL gene. Hematol Cell Ther 1996;38:265–268.
Li S, Lew G . Is B-lineage acute lymphoblastic leukemia with a mature phenotype and L1 morphology a precursor B-lymphoblastic leukemia/lymphoma or Burkitt leukemia/lymphoma? Arch Pathol Lab Med 2003;127:1340–1344.
Vasef MA, Brynes RK, Murata-Collins JL et al. Surface immunoglobulin light chain-positive acute lymphoblastic leukemia of FAB L1 or L2 type: a report of 6 cases in adults. Am J Clin Pathol 1998;110:143–149.
Hammami A, Chan WC, Michels SD et al. Mature B-cell acute leukemia: a clinical, morphological, immunological, and cytogenetic study of nine cases. Hematol Pathol 1991;5:109–118.
Chan NP, Ma ES, Wan TS et al. The spectrum of acute lymphoblastic leukemia with mature B-cell phenotype. Leuk Res 2003;27:231–234.
Taki T, Ida K, Bessho F et al. Frequency and clinical significance of the MLL gene rearrangements in infant acute leukemia. Leukemia 1996;10:1303–1307.
Chessells JM, Harrison CJ, Kempski H et al. Clinical features, cytogenetics and outcome in acute lymphoblastic and myeloid leukaemia of infancy: report from the MRC Childhood Leukaemia working party. Leukemia 2002;16:776–784.
Borkhardt A, Wuchter C, Viehmann S et al. Infant acute lymphoblastic leukemia—combined cytogenetic, immunophenotypical and molecular analysis of 77 cases. Leukemia 2002;16:1685–1690.
Ernst P, Wang J, Korsmeyer SJ . The role of MLL in hematopoiesis and leukemia. Curr Opin Hematol 2002;9:282–287.
Moorman AV, Richards S, Harrison CJ . Involvement of the MLL gene in T-lineage acute lymphoblastic leukemia. Blood 2002;100:2273–2274.
Armstrong SA, Staunton JE, Silverman LB et al. MLL translocations specify a distinct gene expression profile that distinguishes a unique leukemia. Nat Genet 2002;30:41–47.
Mitelman F (ed.) An International System for Human Cytogenetic Nomenclature (1995), Recommendations of the International Standing Committee on Human Cytogenetic Nomenclature; 1995 October; Memphis, TN S. Karger, Basel, 1995.
Sioutos N, Bagg A, Michaud GY et al. Polymerase chain reaction versus Southern blot hybridization. Detection of immunoglobulin heavy-chain gene rearrangements. Diagn Mol Pathol 1995;4:8–13.
Bottaro M, Berti E, Biondi A et al. Heteroduplex analysis of T-cell receptor gamma gene rearrangements for diagnosis and monitoring of cutaneous T-cell lymphomas. Blood 1994;83:3271–3278.
Michiels JJ, Adriaansen HJ, Hagemeijer A et al. TdT positive B-cell acute lymphoblastic leukaemia (B-ALL) without Burkitt characteristics. Br J Haematol 1988;68:423–426.
Ohtsuki T, Ogawa Y, Izumi T et al. Two cases of mature B-cell acute lymphocytic leukemia with normal karyotype in adults. Acta Haematol 1996;96:258–261.
Dunphy CH, van Deventer HW, Carder KJ et al. Mature B-cell acute lymphoblastic leukemia with associated translocations (14;18)(q32;q21) and (8;9)(q24;p13). A Burkitt variant? Arch Pathol Lab Med 2003;127:610–613.
Gunduz C, Cogulu O, Cetingul N et al. New chromosome rearrangement in acute lymphoblastic leukemia. Cancer Genet Cytogenet 2002;137:150–152.
Rowe D, Devaraj PE, Irving JA et al. A case of mature B-cell ALL with coexistence of t(1;19) and t(14;18) and expression of the E2A/PBX1 fusion gene. Br J Haematol 1996;94:133–135.
Kouides PA, Phatak PD, Wang N et al. B-cell acute lymphoblastic leukemia with L1 morphology and coexistence of t(1;19) and t(14;18) chromosome translocations. Cancer Genet Cytogenet 1994;78:23–27.
Trupiano JK, Bringelsen K, Hsi ED . Primary cutaneous lymphoblastic lymphoma presenting in an 8 week-old infant. J Cutan Pathol 2002;29:107–112.
Super HG, Strissel PL, Sobulo OM et al. Identification of complex genomic breakpoint junctions in the t(9;11) MLL-AF9 fusion gene in acute leukemia. Genes Chromosomes Cancer 1997;20:185–195.
Dobson CL, Warren AJ, Pannell R et al. The MLL-AF9 gene fusion in mice controls myeloproliferation and specifies acute myeloid leukaemogenesis. EMBO J 1999;18:3564–3574.
Corral J, Lavenir I, Impey H et al. An MLL-AF9 fusion gene made by homologous recombination causes acute leukemia in chimeric mice: a method to create fusion oncogenes. Cell 1996;85:853–861.
Dunphy CH, Gardner LJ, Evans HL et al. CD15(+) acute lymphoblastic leukemia and subsequent monoblastic leukemia: persistence of t(4;11) abnormality and B-cell gene rearrangement. Arch Pathol Lab Med 2001;125:1227–1230.
Montecino-Rodriguez E, Leathers H, Dorshkind K . Bipotential B-macrophage progenitors are present in adult bone marrow. Nat Immunol 2001;2:83–88.
Montecino-Rodriguez E, Dorshkind K . Identification of B/macrophage progenitors in adult bone marrow. Semin Immunol 2002;14:371–376.
Acknowledgements
We thank Dr Giorgio Cattoretti and his lab for performing the MYC immunohistochemical staining.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tsao, L., Draoua, H., Osunkwo, I. et al. Mature B-cell acute lymphoblastic leukemia with t(9;11) translocation: a distinct subset of B-cell acute lymphoblastic leukemia. Mod Pathol 17, 832–839 (2004). https://doi.org/10.1038/modpathol.3800128
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.3800128
Keywords
This article is cited by
-
Mature B cell acute lymphoblastic leukaemia with KMT2A-MLLT3 transcripts in children: three case reports and literature reviews
Orphanet Journal of Rare Diseases (2021)
-
Heterogeneity of childhood acute leukemia with mature B-cell immunophenotype
Journal of Cancer Research and Clinical Oncology (2019)
-
Hematopoietic stem cell transplantation for pediatric mature B-cell acute lymphoblastic leukemia with non-L3 morphology and MLL-AF9 gene fusion: three case reports and review of the literature
International Journal of Hematology (2016)
-
Mature B-cell lymphoblastic leukemia with MLL rearrangement: an uncommon and distinct subset of childhood acute leukemia
Leukemia (2008)