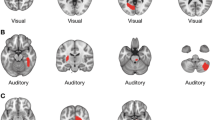
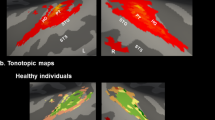
Abstract
HALLUCINATIONS, perceptions in the absence of external stimuli, are prominent among the core symptoms of schizophrenia. The neural correlates of these brief, involuntary experiences are not well understood, and have not been imaged selectively. We have used new positron emission tomography (PET) methods1,2 to study the brain state associated with the occurrence of hallucinations in six schizophrenic patients. Here we present a group study of five patients with classic auditory verbal hallucinations despite medication, demonstrating activations in subcortical nuclei (thalamic, stri-atal), limbic structures (especially hippocampus), and paralimbic regions (parahippocampal and cingulate gyri, as well as orbito-frontal cortex). We also present a case study of a unique, drug-naive patient with visual as well as auditory verbal hallucinations, demonstrating activations in visual and auditory/linguistic association cortices as part of a distributed cortical–subcortical network. Activity in deep brain structures, identified with group analysis, may generate or modulate hallucinations, and the particular neo-cortical regions entrained in individual patients may affect their specific perceptual content. The interaction of these distributed neural systems provides a biological basis for the bizarre reports of schizophrenic patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Silbersweig, D. A. et al. J. cereb. Blood Flow Metab. 13, 617–629 (1993).
Silbersweig, D. A. et al. J. cereb. Blood Flow Metab. 14, 771–782 (1994).
Friston, K. J. et al. Hum. Brain Mapping 2, 189–210 (1995).
Bogerts, B. Schizophr. Bull. 19, 431–445 (1993).
Marsh, L., Suddath, R. L., Higgins, N. & Weinberger, D. R. Schizophr. Res. 11, 225–38 (1994).
Andreasen, N. C. et al. Science 266, 294–298 (1994).
LIinas, R. R. & Pare, D. Neuroscience 44, 521–535 (1991).
Trimble, M. R. The Psychoses of Epilepsy (Raven, New York, 1991).
Csernansky, J. G., Murphy, G. M. & Faustman, W. O. Biol Psychiat. 30, 383–400 (1991).
Olney, J. W. & Farber, N. B. Biol. Psychiat. 37, 667 (1995).
Friston, K. J., Liddle, P. F. & Frith, C. D. Brain 115, 367–382 (1992).
Zeki, S. et al. J. Neurosci. 11, 641–649 (1991).
Barbur, J. L., Watson, J. D., Frackowiak, R. S. & Zeki, S. Brain 116, 1293–1302 (1994).
Farah, M. J. Trends Neurosci. 12, 395–399 (1989).
Dolan, R. J. et al. Hum. Brain Mapping (Suppl.) 1, 417 (1995).
McGuire, P. K. et al. Psychol. Med. (in the press).
Friston, K. J. et al. in Biology of Schizophrenia and Affective Disease (ed. Watson, S. J.) (Raven, New York) (in the press).
Weinberger, D. R. et al. J. Neuropsychiat. clin. Neurosci. 6, 419–427 (1994).
Mesulam, M. M. in Principles of Behavioral Neurology (ed. Mesulam, M. M.) 1–58 (Davis, Philadelphia, 1985).
Volkow, N. D. et al. Am. J. Psychiat. 144, 151–158 (1987).
DeLisi, L. E. et al. Biol. Psychiat. 25, 835–851 (1989).
Cleghorn, J. M. et al. Br. J. Psychiat. 157, 562–570 (1990).
Cleghorn, J. M. et al. Am. J. Psychiat. 149, 1062–1069 (1992).
Matsuda, H. et al. Clin. nucl. Med. 13, 53–55 (1988).
Notardonato, H. et al. Clin. nucl. Med. 14, 319–321 (1989).
Kurachi, M. et al. Eur. Neurol. 24, 176–181 (1985).
Masulek, M. et al. Comp. Psychiat. 30, 99–108 (1989).
McGuire, P. K., Shah, G. M. S. & Murray, R. M. Lancet 342, 703–706 (1993).
Talairach, J. & Tournoux, P. Co-Planar Stereotaxic Atlas of the Human Brain (Thieme, Stuttgart, 1989).
Woods, R. P., Mazziotta, J. C. & Cherry, S. R. J. Comput. Assist. Tomogr. 17, 536–546 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Silbersweig, D., Stern, E., Frith, C. et al. A functional neuroanatomy of hallucinations in schizophrenia. Nature 378, 176–179 (1995). https://doi.org/10.1038/378176a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/378176a0
This article is cited by
-
Transcranial Magnetic Stimulation in the Treatment of Positive, Negative, and Cognitive Symptoms of Psychosis
Current Behavioral Neuroscience Reports (2023)
-
An evolutionary account of impairment of self in cognitive disorders
Cognitive Processing (2023)
-
Excitatory and inhibitory imbalances in the trisynaptic pathway in the hippocampus in schizophrenia: a postmortem ultrastructural study
Journal of Neural Transmission (2023)
-
Alpha transcranial alternating current stimulation reduces depressive symptoms in people with schizophrenia and auditory hallucinations: a double-blind, randomized pilot clinical trial
Schizophrenia (2022)
-
Immune Activation in Pregnant Rats Affects Brain Glucose Consumption, Anxiety-like Behaviour and Recognition Memory in their Male Offspring
Molecular Imaging and Biology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.