Abstract
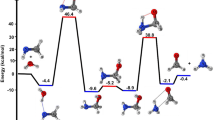
HYPERCOORDINATE carbonium ions can be formed by protonating saturated hydrocarbons with superacids1–3. As this leaves a deficiency of bonding electrons, the resulting non-classical carbocations contain bonds in which two electrons are shared between three nuclei. Protonated methane, CH+5, might be seen as the prototype of such species1–3. But recent calculations4,5 have suggested that all five C–H bonds are effectively equivalent and exchange dynamically very rapidly. It was therefore concluded4 that CH+5 is a highly fluxional molecule without a definite structure, in which the representation in terms of three-centre two-electron bonding is misleading. Here we use a recently developed technique6 to perform ab initio electronic structure calculations that include quantum effects of the nuclei. We find that, although there are prominent quantum-mechanical effects on the structure, including fluxional-ity, pseudo-rotations and hydrogen scrambling, the quantum ground state is nevertheless dominated on average by configurations in which an H2 moiety is attached to a CH3 group forming a three-centre two-electron bond. To this extent, CH+5 should therefore resemble other carbonium ions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Olah, G. A. Carbocations and Electrophilic Reactions (Chemie, Weinheim, 1974).
Olah, G. A., Prakash, G. K. S., Williams, R. E., Field, L. D. & Wade, K. Hypercarbon Chemistry (Wiley, New York, 1987).
Olah, G. A., Prakash, G. K. S. & Sommer, J. Superacids (Wiley, New York, 1985).
Schreiner, P. R., Kim, S.-J., Schaefer, H. F. & Schleyer, P. v. R. J. chem. Phys. 99, 3716–3720 (1993).
Scuseria, G. A. Nature. 366, 512–513 (1993).
Marx, D. & Parrinello, M. Z. Phys. B (Rapid Note) 95, 143–144 (1994).
Boo, D. W. & Lee, Y. T. Chem. Phys. Lett. 211, 358–363 (1993).
Hiraoka, K., Kudaka, I. & Yamabe, S. Chem. Phys. Lett. 184, 271–276 (1991).
Heck, A. J. R., de Koning, L. J. & Nibbering, N. M. M. J. Am. Soc. Mass Spectrom. 2, 453–458 (1991).
Klopper, W. & Kutzelnigg, W. J. phys. Chem. 94, 5625–5630 (1990).
Schleyer, P. v. R. & Carneiro, J. W. d. M. J. comput. Chem. 13, 997–1003 (1992).
Car, R. & Parrinello, M. Phys. Rev. Lett. 55, 2471–2474 (1985).
Jones, R. O. & Gunnarsson, O. Rev. mod. Phys. 61, 689–746 (1989).
Remler, D. K. & Madden, P. A. Molec. Phys. 70, 921–966 (1990).
Payne, M. C., Teter, M. P., Allan, D. C., Arias, T. A. & Joannopoulos, J. D. Rev. mod. Phys. 64, 1045–1097 (1992).
Feynman, R. P. & Hibbs, A. R. Quantum Mechanics and Path integrals (McGraw-Hill, New York, 1965).
Chandler, D. in Liquids, Freezing, and Glass Transition (eds Hansen, J. P., Levesque, D. & Zinn-Justin, J.) 193–285 (Elsevier, Amsterdam, 1991).
Becke, A. D. Phys. Rev. A38, 3098–3100 (1988).
Boo, D. W., Liu, Z. F., Tse, J. S., Suits, A. G. & Lee, Y. T. Phys. Rev. Lett. (submitted).
Tse, J. S., Klug, D. D. & Laasonen, K. Phys. Rev. Lett. 74, 876–879 (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Marx, D., Parrinello, M. Structural quantum effects and three-centre two-electron bonding in CH+5. Nature 375, 216–218 (1995). https://doi.org/10.1038/375216a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/375216a0
This article is cited by
-
Dilute carbon in H3S under pressure
npj Computational Materials (2022)
-
Electronic-structure methods for materials design
Nature Materials (2021)
-
About the structure of CH5 + and structural variations in related systems
Structural Chemistry (2017)
-
The two-component quantum theory of atoms in molecules (TC-QTAIM): the unified theory of localization/delocalization of electrons, nuclei, and exotic elementary particles
Theoretical Chemistry Accounts (2013)
-
Quantum-induced symmetry breaking explains infrared spectra of CH5+ isotopologues
Nature Chemistry (2010)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.