Abstract
THIS PAPER HAS BEEN RETRACTED; see retraction.
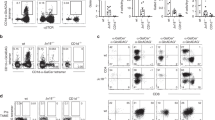
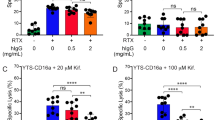
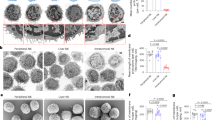
A diversity of high-affinity Oligosaccharide ligands are identified for NKR-P1, a membrane protein on natural killer (NK) cells which contains an extracellular Ca2+-dependent lectin domain. Interactions of such oligosaccharides on the target cell surface with NKR-P1 on the killer cell surface are crucial both for target cell recognition and for delivery of stimulatory or inhibitory signals linked to the NK cytolytic machinery. NK-resistant tumour cells are rendered susceptible by preincubation with liposomes expressing NKR-P1 ligands, suggesting that purging of tumour or virally infected cells in vivo may be a therapeutic possibility.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Feizi, T. Nature 314, 53–57 (1985).
Hakomori, S. Cancer Res. 45, 2405–2414 (1985).
Feizi, T. & Childs, R. A. Biochem. J. 245, 1–11 (1987).
Feizi, T. in Carbohydrate Recognition in Cellular Function Ciba Found. Symp. 145 (eds Bock, G. & Harnett, S.) 62–79 (Wiley, Chichester, 1989).
Drickamer, K. Curr. Opin. struct. Biol. 3, 393–400 (1993).
Feizi, T. Curr. Opin. struct. Biol. 3, 701–710 (1993).
Bevilacqua, M. P. & Nelson, R. M. J. clin. Invest. 91, 379–387 (1993).
Lasky, L. A. et al. Cell 56, 1045–1055 (1989).
McEver, R. P. J. Cell Biochem. 45, 156–161 (1991).
Chambers, W. H., Adamkiewicz, T. & Houchins, J. P. Glycobiology 3, 9–14 (1993).
Yokoyama, W. M. Curr. Opin. Immun. 5, 67–73 (1993).
Trinchieri, G. Adv. Immun. 47, 187–376 (1989).
Giorda, R. et al. Science 249, 1298–1300 (1990).
Ryan, J. C., Turck, J., Niemi, E. C., Yokoyama, W. M. & Seaman, W. E. J. Immun. 149, 1631–1635 (1992).
Yokoyama, W. M. & Seaman, W. E. A. Rev. Immun. 11, 613–635 (1993).
Chambers, W. H. et al. J. exp. Med. 169, 1373–1389 (1989).
Ryan, J. C., Niemi, E. C., Goldfien, R. D., Hiserodt, J. C. & Seaman, W. E. J. Immun. 147, 3244–3250 (1991).
Bezouška, K. et al. J. biol. Chem. 269, 16945–16952 (1994).
Kjellen, L. & Lindahl, U. A. Rev. Biochem. 60, 443–475 (1991).
Scudder, P. et al. Eur. J. Biochem. 157, 365–373 (1986).
Karlhofer, F. M., Ribaudo, R. K. & Yokoyama, W. M. Nature 358, 66–70 (1992).
Ryan, J. C., Niemi, E. C. & Seaman, W. E. Nat. Immun. Cell Growth Reg. 11, 279 (abstr.) (1992).
McCoy, J. P. Jr & Chambers, W. H. Glycobiology 1, 321–328 (1991).
Ando, I. et al. Int. J. Cancer 40, 12–17 (1987).
Grayson, G. & Ladisch, S. Cell. Immun. 139, 18–29 (1992).
Yamamoto, H., Fuyama, S., Arai, S. & Sendo, F. Cell. Immun. 96, 409–417 (1985).
Yogeeswaran, G. et al. Int. J. Cancer 28, 517–526 (1981).
Young, W. M. Jr, Durdik, J. M., Urdal, D., Hakomori, S. & Henney, C. S. J. Immun. 126, 1–6 (1981).
Forbes, J. T., Bretthauer, R. K. & Oeltmann, T. N. Proc. natn. Acad. Sci. U.S.A. 78, 5797–5801 (1981).
Chambers, W. H. & Oeltmann, T. N. J. Immun. 137, 1469–1474 (1986).
Haubeck, H.-D., Kolsch, E., Imort, M., Hasilik, A. & von Figura, K. J. Immun. 134, 65–69 (1985).
von Figura, K. & Hasilik, A. A. Rev. Biochem. 55, 167–193 (1986).
Young, W. W. Jr, Hakomori, S., Durdik, J. M. & Henney, C. S. J. Immun. 124, 199–201 (1980).
Chou, D. K. H. et al. J. biol. Chem. 261, 11717–11725 (1986).
Ohmori, K. et al. Blood 74, 255–261 (1989).
Stoll, M. S., Mizuochi, T., Childs, R. A. & Feizi, T. Biochem. J. 256, 661–664 (1988).
Gooi, H. C. et al. J. biol. Chem. 260, 13218–13224 (1985).
Larkin, M. et al. J. biol. Chem. 267, 13661–13668 (1992).
Yuen, C.-T, et al. J. biol. Chem. 269, 1595–1598 (1994).
Loveless, R. W., Floyd-O'Sullivan, G., Raynes, J. G., Yuen, C.-T. & Feizi, T. EMBO J. 11, 813–819 (1992).
Schwarzmann, G. & Sandhoff, K. Meth. Enzym. 138, 319–341 (1987).
Feizi, T., Stoll, M. S., Yuen, C.-T., Chai, W. & Lawson, A. M. Meth. Enzym. 230, 484–519 (1993).
Vujanovic, N. L., Herberman, R. B., Maghazachi, A. A. & Hiserodt, J. C. J. exp. Med. 167, 15–29 (1988).
Ward, J. M. & Reynolds, C. W. Am. J. Path. 111, 1–10 (1983).
Brunner, K. T., Manuel, J., Cerottini, J. C. & Chapuis, B. Immunology 14, 181–196 (1968).
Feizi, T., Childs, R. A., Watanabe, K. & Hakomori, S. J. exp. Med. 149, 975–980 (1979).
Chused, T. M. et al. Cytometry 8, 396–404 (1987).
Robinson, P. J. in Immunological Methods Vol. III (eds Lefkovitz, I. & Pernis, B.) 125–132 (Academic, Orlando, 1979).
Laemmli, U. K. Nature 227, 680–685 (1970).
Ebel, F., Schmitt, E., Peter-Katalinic, J., Kniep, B. & Mühlradt, P. F. Biochemistry 31, 12190–12197 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bezouška, K., Yuen, CT., O'Brien, J. et al. Oligosaccharide ligands for NKR-P1 protein activate NK cells and cytotoxicity. Nature 372, 150–157 (1994). https://doi.org/10.1038/372150a0
Issue Date:
DOI: https://doi.org/10.1038/372150a0
This article is cited by
-
Retraction Note: Oligosaccharide ligands for NKR-P1 protein activate NK cells and cytotoxicity
Nature (2013)
-
Antitumor activity of N-acetyl-d-glucosamine-substituted glycoconjugates and combined therapy with keyhole limpet hemocyanin in B16F10 mouse melanoma model
Folia Microbiologica (2010)
-
Genetically linked C-type lectin-related ligands for the NKRP1 family of natural killer cell receptors
Nature Immunology (2003)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.