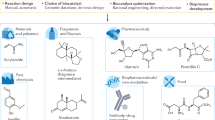
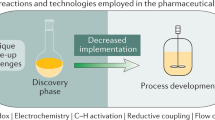
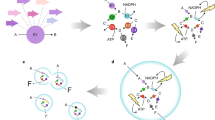
Abstract
The use of biocatalysis for industrial synthetic chemistry is on the verge of significant growth. Biocatalytic processes can now be carried out in organic solvents as well as aqueous environments, so that apolar organic compounds as well as water-soluble compounds can be modified selectively and efficiently with enzymes and biocatalytically active cells. As the use of biocatalysis for industrial chemical synthesis becomes easier, several chemical companies have begun to increase significantly the number and sophistication of the biocatalytic processes used in their synthesis operations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Faber, K. Biotransformations in Organic Chemistry: A Textbook (Springer, Berlin, 1997).
Dordick, J. S. Biocatalysts for Industry (Plenum Press, New York, 1991).
Jensen, V. J. & Rugh, S. Industrial scale production and application of immobilized glucose isomerase. Methods Enzymol. 136, 356–370 (1987).
Boesten, W. H.J., Moody, H. M. & Roos, E. C. Process for the recovery of ampicillin from enzymatic acylation of 6-aminopenicillanic acid (Chemferm; patent no. WO.9630376; The Netherlands; 1996).
Schulze, B., Broxterman, R., Shoemaker, H. & Boesten, W. Review of biocatalysis in the production of chiral fine chemicals. Spec. Chem. 18, 244, 246 ( 1998).
Kaplan, D. L., Dordick, J. S., Gross, R. A. & Swift, G. Enzymes in polymer science: an introduction. Am. Chem. Soc. Symp. Ser. 684, 2–16 ( 1998).
Pokora, A. R. & Cyrus, W. L. Phenolic developer resins (The Mead Corp., Dayton, OH: US patent no. 4,647,952; 1987 ).
Nagasawa, T. & Yamada, H. Application of nitrile converting enzymes for the production of useful compounds. Pure Appl. Chem. 62, 1441–1444 ( 1990).
Buchholz, H. & Kasche, V. Biokatalysatoren und Enzymtechnologie (VCH, New York, 1997).
Tramper, J. Chemical versus biochemical conversion: when and how to use biocatalysts. Biotechnol. Bioeng. 52, 290–295 (1996).
Arnold, F. H. & Volkov, A. A. Directed evolution of biocatalysts . Curr. Opin. Chem. Biol. 3, 54– 59 (1999).
Stemmer, W. P. C. DNA shuffling by random fragmentation and reassembly: in vitro recombination for molecular evolution. Nature 370, 389 –391 (1994).
Blinkovsky, A. M., Martin, B. D. & Dordick, J. S. Enzymology in monophasic organic media. Curr. Opin. Biotechnol. 3, 124–129 (1992).
Klibanov, A. M. Asymmetric transformations catalyzed by enzymes in organic solvents. Acc. Chem. Res. 23, 114–120 (1990).
Russell, A. J., Beckman, E. J. & Chaudhary, A. K. Studying enzyme activity in supercritical fluids . Chemtech 3, 33–37 (1994).
Lalonde, J., Navia, M. A. & Margolin, A. L. Crosslinked enzyme crystals of lipases as catalysts for kinetic resolution of acids and alcohols. Methods Enzymol. 286, 443–464 ( 1997).
Park, O., Kim, D. Y. & Dordick, J. S. Enzyme-catalyzed synthesis of sugar-containing monomers and linear polymers. Biotechnol. Bioeng. 70, 208–216 (2000).
Khmelnitsky, Y. L., Welch, S. H., Dlark, D. S. & Dordick, J. S. Salts dramatically enhance activity of enzymes suspended in organic solvents . J. Am. Chem. Soc. 116, 2647– 2648 (1994).
Ru, M. T., Dordick, J. S., Reimer, J. A. & Clark, D. S. Optimizing the salt-induced activation of enzymes in organic solvents: effects of lyophilization time and water content. Biotechnol. Bioeng. 63, 233–241 (1999).
Ru, M. T. et al. On the salt-induced activation of lyophilized enzymes in organic solvents: effect of salt kosmotropicity on enzyme activity. J. Am. Chem. Soc. 122, 1565–1571 (2000).
Engbersen, J. F. J., Broos, J., Verboom, W. & Reinhoudt, D. N. Effects of crown ethers and small amounts of cosolvent on the activity and enantioselectivity of α-chymotrypsin in organic solvents. Pure Appl. Chem. 68, 2171–2178 (1996).
Slade, C. J. & Vulfson, E. N. Induction of catalytic activity in proteins by lyophilization in the presence of a transition state analogue . Biotechnol. Bioeng. 57, 211– 215 (1998).
Russel, A. J. & Klibanov, A. M. Inhibitor-induced enzyme activation in organic solvents. J. Biol. Chem. 263, 11626–11626 (1988).
Khmelnitski, Y. L. et al. Synthesis of water soluble Paclitaxel derivatives by enzymatic acylation. J. Am. Chem. Soc. 119, 11554– 11555 (1997).
Paradkar, V. M. & Dordick, J. S. Aqueous-like activity of α-chymotrypsin dissolved in nearly anhydrous organic solvents . J. Am. Chem. Soc. 116, 5009– 5010 (1994).
Wang, P., Sergeeva, M. V., Lim, L. & Dordick, J. S. Biocatalytic plastics as active and stable materials for biotransformations. Nature Biotechnol. 15, 789–793 (1997).
Yang, Z. et al. Activity and stability of enzymes incorporated into acrylic polymers . J. Am. Chem. Soc. 117, 4843– 4850 (1995).
Chaudhary, A., Lopez, J., Beckman, E. J. & Russell, A. J. Biocatalytic solvent-free polymerization to produce high molecular weight polyesters. Biotechnol. Prog. 13, 318– 325 (1997).
Wubbolts, M. G., Hoven, J., Melgert, B. & Witholt, B. Efficient production of optically active styrene epoxides in two-liquid phase cultures. Enzyme Microb. Technol. 16, 887–894 (1994).
Wubbolts, M. G., Favre-Bulle, O. & Witholt, B. Biosynthesis of synthons in two-liquid phase media. Biotechnol. Bioeng. 52, 301–308 (1996).
Held, M. et al. Preparative scale production of 3-substituted catechols using a novel monooxygenase from Pseudomonas azelaica HBP1. J. Mol. Catal. Enzymatic 5, 87–93 (1998).
De Smet, M. J., Wijnberg, H. & Witholt, B. Synthesis of 1,2-epoxyoctane by Pseudomonas oleovorans during growth in a two phase system containing high concentrations of 1-octene. Appl. Environ. Microbiol. 42, 811–816 (1981).
De Smet, M. J., Witholt, B. & Wijnberg, H. Practical approach to high yield enzymatic stereospecific organic synthesis in multiphase systems. J. Org. Chem. 46, 3128–3131 (1981).
De Smet, M. J., Kingma, J., Wijnberg, H. & Witholt, B. Pseudomonas oleovorans as a tool in bioconversions of organic substrates: growth, morphology and conversion characteristics in different two-phase systems. Enzyme Microb. Technol. 5, 352–360 (1983).
Favre-Bulle, O., Schouten, T., Kingma, J. & Witholt, B. Bioconversion of n-octane to octanoic acid by a recombinant Escherichia coli cultured in a two-liquid phase bioreactor. Biotechnology 9, 367–71 (1991).
Laane, C., Boeren, S., Vos, K. & Veeger, C. Rules for optimization of biocatalysts in organic solvents. Biotechnol. Bioeng. 30, 81–87 (1987).
Nikolova, P. & Ward, O. P. Whole cell biocatalysis in nonconventional media. J. Ind. Microbiol. 12, 76– 86 (1993).
Buckland, B. C. et al. Microbial conversion of indene to indanol: a key intermediate in the synthesis of Crixivan. Metabol. Eng. 1, 63–74 (1999).
Schmid, A., Kollmer, A., Mathys, R. G. & Witholt, B. Developments toward large scale bacterial bioprocesses in the presence of bulk amounts of organic solvents. Extremophiles 2, 249–245 (1998).
Mathys, R. G., Schmid, A. & Witholt, B. Integrated two-liquid phase bioconversion and product-recovery processes for the oxidation of alkanes: process design and economic evaluation . Biotechnol. Bioeng. 64, 459– 477 (1999).
Panke, S., Witholt, B., Schmid, A. & Wubbolts, M. G. Towards a biocatalyst for (S)-styrene oxide production: characterization of the styrene degradation pathway of Pseudomonas sp. strain VLB120. Appl. Environ. Microbiol. 64, 2032–2043 (1998). [Published erratum appears in Appl. Environ. Microbiol. 64, 3546 (1998).]
Kiener, A. Enzymatic oxidation of methyl groups on aromatic heterocycles: a versatile method for the preparation of heteroaromatic carboxylic acids. Angw. Chem. Int. Ed. Engl. 31, 774– 775 (1992). (Patent no. EP-B 0442430.)
Lamare, S. & Legoy, M.-D. Biocatalysis in the gas phase. Trends Biotechnol. 11, 413–418 (1993).
Held, M. et al. An integrated process for the production of toxic catechols from toxic phenols based on a designer biocatalyst. Biotechnol. Bioeng. 62, 641–648 ( 1999).
Li, Z., Feiten, H.-J., Van Beilen, J. B., Duetz, W. & Witholt, B. Preparation of optically active N-benzyl-3-hydroxy-pyrrolidine by enzymatic hydroxylation. Tetrahedr. Asym. 10, 1323–1333 (1999).
Schmid, A., Sonnleitner, B. & Witholt, B. Medium chain length alkane solvent-cell transfer rates in two-liquid phase, Pseudomonas oleovorans cultures. Biotechnol. Bioeng. 60, 10–23 ( 1998).
Schmid, A., Kollmer, A., Mathys, R. G. & Witholt, B. Developments toward large-scale bacterial bioprocesses in the presence of bulk amounts of organic solvents. Extremophiles 2, 249–256 (1998).
Panke, S., Wubbolts, M. G., Schmid, A. & Witholt, B. Production of enantiopure styrene oxide by recombinant Escherichia coli synthesizing a two-component styrene monooxygenase. Biotechnol. Bioeng. 69, 91–100 ( 2000).
Panke, S., de Lorenzo, V., Kaiser, A., Witholt, B. & Wubbolts, M. G. Engineering of a stable whole-cell biocatalyst capable of (S)-styrene oxide formation for continuous two-liquid-phase applications. Appl. Environ. Microbiol. 65, 5619–5623 (1999).
Yamada, H. in New Frontiers in Screening for Microbial Biocatalysts (eds Kieslich, K., van der Beek, C. P., de Bont, J. A. M. & van den Tweel, W. J. J.) 13–17 (Elsevier, Amsterdam, 1998).
Lye, G. J. & Woodley, J. M. Application of in situ product-removal techniques to biocatalytic processes. Trends Biotechnol. 17, 395–402 (1999).
Witholt, B. et al. Bioconversions of aliphatic compounds by Pseudomonas oleovorans in multiphase bioreactors: background and economic potential. Trends Biotechnol. 8, 46–52 (1990).
Mathys, R. G., Schmid, A., Kut, O. M. & Witholt, B. Alkanol removal from the apolar phase of a two-liquid fermentation system. 2. Effect of fermentation medium on distillation. J. Chem. Technol. Biotechnol. 71, 326–334 (1998).
Mathys, R. G., Kut, O. M. & Witholt, B. Alkanol removal from the apolar phase of a two-liquid fermentation system. 1. Comparison of a less volatile and more volatile in situ extraction solvent for separation of 1-octanol by distillation . J. Chem. Tech. Biotech. 71, 315– 325 (1998).
Balkenhohl, F., Ditrich, K., Hauer, B. & Ladner, W. Optically active amines via lipase-catalyzed methoxyacetylation. J. prakt. Chemie 339, 381–384 ( 1997).
Hauer, B. et al. The development of enzymes for the preparation of chemicals. Chimia 53, 613–616 ( 1999).
Ress-Löschke, M., Friedrich, T., Hauer, B., Mattes, R. & Engels, D. Verfahren zur Herstellung chiraler Carbonsäuren (BASF; German patent no. DE 198 448 129 A1; 1998)
Boesten, W. H. J. Enzymic Division of DL-phenylglycinamide into its optically active antipodes (German patent no. DE 2526594; 1976).
Boesten, W. H. J. & Meyer-Hoffman, L. R. M. Enzyme product with L-α-aminoacylamidase activity (NOVO, GB 1577087; 1977).
Broxterman, R., Sonke, T., Wories, H. & van den Tweel, W. Biocatalytic production of unnatural amino acids. Pharm. Manuf. Int. 61(2000).
Broxterman, Q. B., Boesten, W. H. J., Schoemaker, H. E. & Schulze, B. DSM gets the biocatalysis bug. Spec. Chem. 17, 186–193 (1997).
Boesten, W. H. J., Broxterman, Q. B. & Plaum, M. J. M. Process for preparing optically active 2-amino-ω-oxoalkanoic acid derivatives (DSM, EP 905257; 1998).
Kaptein, B. et al. Preparation and use of enantiomerically pure Cα-tetrasubstituted α-amino acids. Chim. Oggi 14, 9– 12 (1996).
Kaptein, B. et al. Enzymic resolution of α,α-disubstituted α-amino acid esters and amides. Tetrahedr. Asym. 4, 1113–1116 (1993).
Kruizinga, W. H. et al. Synthesis of optically pure α-alkylated α-amino acids and a single-step method for enantiomeric excess determination. J. Org. Chem. 53, 5392 (1988).
Roos, E. C. et al. Synthesis of racemic α-amino carboxamides via Lewis acid-mediated reactions of α-methoxyglycinamide derivatives with allylsilanes: enzymic resolution to optically active α-amino acids. J. Org. Chem. 57, 6769–6778 (1992).
Schoemaker, H. E. et al. Application of enzymes in industrial organic synthesis. Chimia 51, 308–310 ( 1997).
Kirchner, G., Salzbrenner, E., Werenka, C. & Boesten, W. H. J. Process for the preparation of the disodium salt of Z-L-aspartic acid from fumaric acid (DSM Chemie and Holland Sweetner Company, EP 832982; 1997).
Bruggink, A. Biocatalysis and process integration in the synthesis of semisynthetic antibiotics . Chimia 50, 431–432 (1996).
Demain, A. L. & Baez-Vasquez, M. A. Immobilized Streptomyces clavuligerus NP1 cells for biotransformation of penicillin G into deacetoxycephalosporin G. Appl. Biochem. Biotechnol. 87, 135– 140 (2000).
Boesten, W. H. J., van Dooren, T. J. & Smeets, J. C. M. Process for the preparation of a β-lactam antibiotic (Chemferm, WO 9623897; 1996).
Bruggink, A., Roos, E. C. & de Vroom, E. Penicillin acylase in the industrial production of β-lactam antibiotics. Org. Process Res. Dev. 2, 128 –133 (1998).
Kulla, H. G. Enzymatic hydroxylations in industrial application. Chimia 45, 81–85 (1991). (Lonza, EP-B 0152948; EP-B 0152949.)
Wieser, M., Heinzmann, K. & Kiener, A. Bioconversion of 2-cyanopyrazine to 5-hydroxypyrazine-2-carboxylic acid with Agrobacterium sp. DSM 6336. Appl. Microbiol. Biotechnol 48, 174–176 ( 1997). (Lonza, EP-B 0578137.)
Bergmann, K. E., Cynamon, M. H. & Welch, J. T. Quantitative structure-activity relationships for the in-vitro antimycobacterial activity for pyrazinoic acid esters. J. Med. Chem. 39, 3394–3400 (1996).
Spande, T. F. et al. Epibatidine: A novel (chloropyridyl) azabicycloheptane with potent analgesic activity from an Ecuadoran poison frog. J. Am. Chem. Soc. 114, 3475–3478 (1992).
Roduit, J.-P., Wellig, A. & Kiener, A. Renewable functionalized pyridines derived from microbial metabolites of the alkaloid (S)-nicotine. Heterocycles 46, 1687–1702 (1997). (Lonza, EP-B 0713869.)
Eichhorn, E., Roduit, J.-P., Shaw, N., Heinzmann, K. & Kiener, A. Preparation of (S)-piperazine-2-carboxylic acid, (R)-piperazine-2-carboxylic acid, and (S)-piperidine-2-carboxylic acid by kinetic resolution of the corresponding racemic carboxamides with stereoselective amidases in whole bacterial cells . Tetrahedr. Asym. 8, 2533– 2536 (1997). (Lonza, EP-A 0686698; WO 96/35775.)
Reipa, V., Mayhew, M. P. & Vilker, V. L. A direct electrode driven P450 cycle for biocatalysis . Proc. Natl Acad. Sci. USA 94, 13554– 13558 (1997).
Certik, M. & Shimizu, S. Biosynthesis and regulation of microbial polyunsaturated fatty acid production. J. Biosci. Bioeng. 87, 1–14 (1999).
Koizumi, S., Endo, T., Tabata, K. & Ozaki, A. Large scale production of UDP-galactose and globotriose by coupling metabolically engineered bacteria . Nature Biotechnol. 16, 847– 850 (1998).
Kim, Y., Dordick, J. S. & Clark, D. S. Biotechnol. Bioeng. (in the press).
MacBeath, G. & Schreiber, S. L. Printing proteins as microarrays for high-throughput function determination. Science 289, 1760–1763 (2000).
Michels, P. C., Khmelnitsky, Y. L., Dordick, J. S. & Clard, D. S. Combinatorial biocatalysis: a natural approach to drug discovery. Trends Biotechnol. 16, 210–215 (1998).
Krstenansky, J. L. & Khmelnitsky, Y. L. Biocatalytic combinatorial synthesis. Bioorg. Med. Chem. 7, 2157–2162 (1999).
Simon, H., Bader, J., Günther, H., Neumann, S. & Thanos, J. Chiral compounds synthesized by biocatalytic reductions. Angew. Chem. Int. Ed. 24, 539 –553 (1985).
Adlercreutz, P. Cofactor regeneration in biocatalysis in organic media. Biocatal. Biotransform. 14, 1–30 ( 1996).
Chenault, H. K., Simon, E. S. & Whitesides, G. M. Cofactor regeneration for enzyme catalysed synthesis . Biotechnol. Gen. Eng. Rev. 6, 221– 270 (1988).
Bommarius, A. S., Drauz, K., Groeger, U. & Wandrey, C. in Chirality in Industry (eds Collins, A. N., Sheldrake, G. N. & Crosby, J.) 371–397 (Wiley, Chichester, 1992).
Clair, N. S., Wang, Y.-F. & Margolin, A. L. Cofactor-bound cross linked enzyme crystals (CLEC) of alcohol dehydrogenase. Angew. Chem. Int. Ed. 39, 380–383 (2000).
Westerhausen, D., Herrmann, S., Hummel, W. & Steckhan, E. Formate driven, non-enzymatic NAD(P)H regeneration for the alcohol dehydrogenase catalyzed stereoselective reduction of 4-phenylbutanone. Angew. Chem. Int. Ed. 31, 1529–1531 ( 1992).
Hollmann, F., Schmid, A. & Steckhan, E. First synthetic application of a monooxygenase employing indirect electrochemical NADH-regeneration. Angew. Chem. Int. Ed. (in the press).
Günther, H. & Simon, H. Artificial electron carriers for preparative biocatalytic redox reactions forming reversibly carbon hydrogen bonds with enzymes present in strict or facultative anaerobes. Biocatal. Biotransform. 12, 1–26 (1995).
Resnick, S. & Zehnder, A. J. B. In vitro ATP regeneration from polyphosphate and AMP by polyphosphate:AMP phosphotransferase and adenylate kinase from Acinetobacter johnsonii 210A. Appl. Environ. Microbiol. 66, 2045–2051 (2000).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schmid, A., Dordick, J., Hauer, B. et al. Industrial biocatalysis today and tomorrow. Nature 409, 258–268 (2001). https://doi.org/10.1038/35051736
Issue Date:
DOI: https://doi.org/10.1038/35051736
This article is cited by
-
Two-substrate enzyme engineering using small libraries that combine the substrate preferences from two different variant lineages
Scientific Reports (2024)
-
Semi-rational engineering an aldo–keto reductase for stereocomplementary reduction of α-keto amide compounds
Microbial Cell Factories (2023)
-
Encapsulating and stabilizing enzymes using hydrogen-bonded organic frameworks
Nature Protocols (2023)
-
Hierarchically Structured CA@ZIF-8 Biohybrids for Carbon Dioxide Mineralization
Applied Biochemistry and Biotechnology (2023)
-
Arylacetonitrilases: Potential Biocatalysts for Green Chemistry
Applied Biochemistry and Biotechnology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.