Abstract
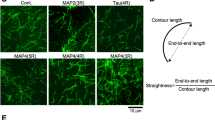
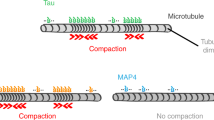
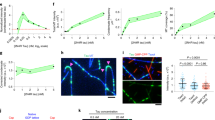
Here we report that the microtubule-associated proteins MAP2 and tau share two separable functional domains. One is the microtubule-binding site which serves to nucleate microtubule assembly; the second is a short C-terminal α-helical sequence which can crosslink microtubules by means of a hydrophobic zipper interaction into dense stable parallel arrays characteristic of axons or dendrites. Thus, interactions between molecules of a single type are capable of drastically reorganizing microtubules and completely suppressing their dynamic properties.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kirschner, M. & Mitchison, T. Cell 45, 329–342 (1986).
Saxton, W. M. et al. J. Cell Biol. 99, 2175–2186 (1984).
Schulze, E. & Kirschner, M. J. Cell Biol. 102, 1020–1031 (1986).
Salmon, E. D., Leslie, R. J., Saxton, W. M., Karow, M. L. & Mcintosh, J. R. J. Cell Biol. 99, 2165–2174 (1984).
Mitchison, T., Evans, L., Schulze, E. & Kirschner, M. W. Cell 45, 515–527 (1986).
Koshland, D. E., Mitchison, T. J. & Kirschner, M. W. Nature 331, 499–504 (1988).
Dustin, P. Microtubules (Springer, Berlin, 1984).
Olmsted, J. B. A. Rev. Cell Biol. 2, 419–455 (1986).
Bloom, G. S., Schoenfeld, T. A. & Vallee, R. B. J. Cell Biol. 98, 320–330 (1984).
Cleveland, D. W., Hwo, S. & Kirschner, M. W. J. molec. Biol. 116, 207–225 (1977).
Vallee, R. B. Proc. Natn. Acad. Sci U.S.A. 77, 3206–3210 (1980).
Murphy, D. B. & Borisy, G. G. Proc. natn. Acad. Sci. U.S.A. 72, 2696–270 (1975).
Paschal, B. M. & Vallee, R. B. Nature 330, 181–183 (1987).
Vallee, R. B., Wall, J. S., Paschal, B. M. & Schpetner, H. S. Nature 332, 561–563 (1988).
Binder, L. I., Frankfurter, A. & Rebhun, L. I. J. Cell Biol. 101, 1371–1378 (1985).
Bernhardt, R. & Matus, A. J. comp. Neurol. 226, 203–231 (1984).
Garner, C. C. & Matus, A. Nature 336, 674–676 (1988).
Sloboda, R. D., Dentler, W. L. & Rosenbaum, J. L. Biochemistry 15, 4497–4505 (1976).
Drubin, D. G. & Kirschner, M. W. J. Cell Biol. 103, 2739–2747 (1986).
Wischik, C. M. Curr. Opi. Cell Biol. 1, 115–122 (1989).
Lewis, S. A., Wang, D. & Cowan, N. J. Science 242, 936–939 (1988).
Lee, G., Cowan, N. J. & Kirschner, M. W. Science 239, 285–287 (1988).
Landschulz, W. H., Johnson, P. F. & McKnight, S. L. Science 240, 1759–1764 (1988).
Kouzarides, T. & Ziff, E. Nature 336, 646–651 (1988).
O'Shea, E. K., Rutkowski, R. & Kim, P. S. Science 243, 538–542 (1989).
Hope, I. A. & Struhl, K. EMBO J. 6, 2781–2784 (1987).
Yen, T. J., Machlin, P. S. & Cleveland, D. W. Nature 334, 580–585 (1989).
Schiff, P. B. & Horwitz, S. B. Nature 277, 665–667 (1979).
Tucker, R. P., Binder, L. I., Viereck, C., Hemmings, B. A. & Matus, A. J. Neurosci. 8, 4503–4515 (1988).
Himmler, A., Drechsel, D., Kirschner, M. W. & Martin, D. W. Molec. cell. Biol. 9, 1381–1388 (1989).
Chou, P. Y. & Fasman, G. T. Biochemistry 13, 222–228 (1974).
Garnier, J., Osguthorpe, D. J. & Robson, B. J. molec. Biol. 120, 97–106 (1978).
Solomon, F., Magendantz, M. & Salzman, A. Cell 18, 431–438 (1979).
Himmler, A. Molec. cell. Biol. 9, 1389–1396 (1989).
Bloom, G. S. & Vallee, R. B. J. cell Biol. 96, 1523–1531 (1983).
Hirokawa, N., Hisanaga, S. & Shiomura, Y. J. Neurosci. 8, 2769–2779 (1988).
Flynn, G., Jolly, J. C. & Purich, D. L. Biochem. biophys. Res. Commun. 148, 1453–1459 (1987).
Lewis, S. A. & Cowan, N. J. Molec. cell. Biol. 6, 1529–1534 (1986).
Levy, E., Liem, R. K. H., D'Eustachio, P. & Cowan, N. J. Eur. J. Biochem. 166, 71–77 (1987).
Vallee, R. B., DiBartolomeis, M. J. & Theurkauf, W. E. J. Cell Biol. 90, 568–576 (1981).
Shiomura, Y. & Hirokawa, N. J. Cell Biol. 104, 1575–1578 (1987).
Sloboda, R. D. et al. in Cell Mobility (eds Goldman, R. & Polland, T.) 1172–1212 (Cold Spring Harbor Laboratory, New York, 1976).
Hirokawa, N., Shiomura, Y. & Okabe, S. J. Cell Biol. 107, 1449–1459 (1988).
Mulligan, R. C. & Berg, P. Proc. natn. Acad. Sci. U.S.A. 78, 2072–2076 (1981).
Chen, C. & Okayama, H. Molec. cell. Biol 7, 2745–2752 (1987).
Lewis, S. A., Villasante, A., Sherline, P. & Cowan, N. J. J. Cell Biol. 102, 2098–2105 (1986).
Gu, W., Lewis, S. A. & Cowan, N. J. J. Cell Biol. 106, 2011–2022 (1988).
Shiomura, Y. & Hirokawa, N. J. Neurosci. 7, 1461–1469 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lewis, S., Ivanov, I., Lee, GH. et al. Organization of microtubules in dendrites and axons is determined by a short hydrophobic zipper in microtubule-associated proteins MAP2 and tau. Nature 342, 498–505 (1989). https://doi.org/10.1038/342498a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/342498a0
This article is cited by
-
Self protein-protein interactions are involved in TPPP/p25 mediated microtubule bundling
Scientific Reports (2015)
-
The MAP1 family of microtubule-associated proteins
Genome Biology (2006)
-
Regulation of microtubule assembly by human EB1 family proteins
Oncogene (2001)
-
MAP2c confers drug stability to microtubulesin vivo
Chinese Science Bulletin (1999)
-
Dlx-2 homeobox gene controls neuronal differentiation in primary cultures of developing basal ganglia
Journal of Molecular Neuroscience (1997)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.