Abstract
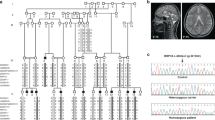
Primary or nonspecific X-linked mental retardation (MRX) is a heterogeneous condition in which affected patients do not have any distinctive clinical or biochemical features in common apart from cognitive impairment1. Although it is present in approximately 0.15–0.3% of males2, most of the genetic defects associated with MRX, which may involve more than ten different genes, remain unknown3. Here we report the characterization of a new gene on the long arm of the X-chromosome (position Xq12) and the identification in unrelated individuals of different mutations that are predicted to cause a loss of function. This gene is highly expressed in fetal brain and encodes a protein of relative molecular mass 91K, named oligophrenin-1, which contains a domain typical of a Rho-GTPase–activating protein (rhoGAP)4,5. By enhancing their GTPase activity, GAP proteins inactivate small Rho and Ras proteins, so inactivation of rhoGAP proteins might cause constitutive activation of their GTPase targets. Such activation is known to affect cell migration and outgrowth of axons and dendrites in vivo6,7,8,. Our results demonstrate an association between cognitive impairment and a defect in a signalling pathway that depends on a Ras-like GTPase.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Lubs, H. A. et al. XLMR genes: update 1996. Am. J. Med. Genet. 64, 147–157 (1996).
Herbst, D. S. & Miller, J. R. Nonspecific X-linked mental retardation II: the frequency in British Columbia. Am. J. Med. Genet. 7, 461–469 (1980).
Gedeon, A. K., Donnelly, A. J., Mulley, J. C., Kerr, B. & Turner, G. How many X-linked genes for non-specific mental retardation (MRX) are there? Am. J. Med. Genet. 64, 158–162 (1996).
Lamarche, N. & Hall, A. GAPs for rho-related GTPases. Trends Genet. 10, 436–440 (1994).
Hildebrand, J. D., Taylor, J. M. & Parsons, J. T. An SH3 domain-containing GTPase-activating protein for Rho and Cdc42 associates with focal adhesion kinase. Mol. Cell. Biol. 16, 3169–3178 (1996).
Zipkin, I. D., Kindt, R. M. & Kenyon, C. J. Role of a new rho family member in cell migration and axon guidance in C. elegans. Cell 90, 883–894 (1997).
Luo, L., Liao, Y. J., Jan, L. Y. & Jan, Y. N. Distinct morphogenetic functions of similar small GTPases: Drosophila Drac1 is involved in axonal outgrowth and myoblast fusion. Genes Dev. 8, 1787–1802 (1994).
Luo, L. et al. Differential effects of the Rac GTPase on Purkinje cell axons and dendritic trunks and spines. Nature 379, 837–840 (1996).
Bienvenu, T. et al. Mapping of the X-breakpoint involved in a balanced X;12 translocation in a female with mild mental retardation. Eur. J. Hum. Genet. 5, 105–109 (1997).
Kel, A. E. et al. SITEVIDEO: a computer system for functional site analysis and recognition. Investigation of the human splice sites. Comput. Appl. BioSci. 9, 617–627 (1993).
Solovyev, V. V., Salsmov, A. A. & Lawrence, C. B. Predicting internal exons by oligonucleotide composition and discriminant analysis of spliceable open reading frames. Nucleic Acids Res., 22, 5156–5163 (1994).
Lerman, L. S. & Silverstein, K. Computational simulation of DNA melting and its application to denaturing gradient gel electrophoresis. Meth. Enzymol. 155, 482–501 (1987).
Davies, H. R. et al. Androgen insensitivity with mental retardation: a contiguous gene syndrome? J. Med. Genet. 34, 158–160 (1997).
Kozak, M. Point mutations define a sequence flanking the AUG initiator codon that modulates translation by eukaryotic ribosomes. Cell 44, 283–292 (1986).
Nizetic, D. et al. Construction, arraying, and high-density screening of large insert libraries of human chromosomes X and 21: their potential use as reference libraries. Proc. Natl Acad. Sci. USA 88, 3233–3237 (1991).
Sambrook, J., Fritsch, E. F. & Maniatis, T. Molecular Cloning: A Laboratory Manual (Cold Spring Harbor Laboratory Press, New York, 1982).
Fernandez, E. et al. Use of chemical clamps in denaturing gradient gel electrophoresis: application in the detection of the most frequent Mediterranean beta-thalassemic mutations. PCR Meth. Appl. 3, 122–124 (1993).
Settleman, J., Albright, C. F., Foster, L. C. & Weinberg, R. A. Association between GTPase activators for Rho and Ras families. Nature 359, 153–154 (1992).
Boguski, M. S. & McCormick, F. Proteins regulating Ras and its relatives. Nature 366, 643–654 (1993).
Aelst, L. V. & D'Souza-Schorey, C. Rho GTPases and signaling networks. Genes Dev. 11, 2295–2322 (1997).
Acknowledgements
We thank the members of the families for their participation in this study; F. Francis, G. Gacon and O. Dorseuil for critically reading the manuscript; and T. Brüls for cosmid clones. This work was supported in part by grants from the AFM, AP-HP, and the Fondation Jérôme Lejeune. P.B is supported by a Ph.D. fellowship from Ministère de la Recherche.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Billuart, P., Bienvenu, T., Ronce, N. et al. Oligophrenin-1 encodes a rhoGAP protein involved in X-linked mental retardation. Nature 392, 923–926 (1998). https://doi.org/10.1038/31940
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/31940
This article is cited by
-
Key role of Rho GTPases in motor disorders associated with neurodevelopmental pathologies
Molecular Psychiatry (2023)
-
Drosophila Graf regulates mushroom body β-axon extension and olfactory long-term memory
Molecular Brain (2021)
-
Arhgap22 Disruption Leads to RAC1 Hyperactivity Affecting Hippocampal Glutamatergic Synapses and Cognition in Mice
Molecular Neurobiology (2021)
-
ARHGAP10, which encodes Rho GTPase-activating protein 10, is a novel gene for schizophrenia risk
Translational Psychiatry (2020)
-
FBP17 and CIP4 recruit SHIP2 and lamellipodin to prime the plasma membrane for fast endophilin-mediated endocytosis
Nature Cell Biology (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.