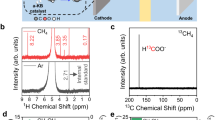
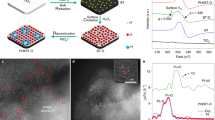
Abstract
As methane is extremely inert, its conversion into a form suitable for chemical use has been achieved by an indirect process in which methane is first converted to synthesis gas and then to methanol and to ethylene glycol1. One disadvantage of this route of conversion is that it requires conditions of high temperature and pressure2. Here we report the direct conversion of methane to methanol, chloromethane and dichloromethane at the three-phase interface (gas/solution/electrode) on illumination at room temperature. The key steps in this process are: (1) electrochemical oxidation of the chloride ion; (2) generation of the chlorine radical under illumination; and (3) formation of the methyl radical by the reaction of methane with the chlorine radical.
Similar content being viewed by others
Article PDF
References
Hatch, L. F. & Matar, S. From Hydrocarbons to Petrochemicals 58–61 (Gulf Publishing Company, Houston, 1981).
Kuhre, C. J. & Shearer, C. J. Oil Gas J. 85–90 (1971).
Nonaka, A. Analyt. Chem. 44, 271–276 (1972).
Bockris, J. O'M. J. chem. Phys. 24, 817–825 (1956).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ogura, K., Takamagari, K. Direct conversion of methane to methanol, chloromethane and dichloromethane at room temperature. Nature 319, 308 (1986). https://doi.org/10.1038/319308a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/319308a0
This article is cited by
-
Sustainable methane utilization technology via photocatalytic halogenation with alkali halides
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.