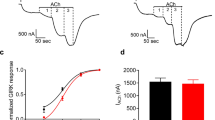
Abstract
Guanine nucleotide binding proteins, interchangeably called N or G proteins, seem to be tbe primary signal-transducing components of various agonist-induced cell membrane functions1–3. In the heart, G proteins have been implicated in β-adrenergic modulation of the slow inward Ca2+ current. We have investigated the role of G proteins in muscarinic activation of an inwardly rectifying, acetylcholine (ACh)-induced K+ current (IACh), and β-adrenergic activation of an (isoprenaline)-induced Ca2+ current (Isi). Here we report that intracellular application of the non-hydrolysable GTP analogue 5′-guanylylimidodiphosphate (GppNHp) brought about an agonist-induced, antagonist-resistant, persistent activation of IACh and Isi. This functional uncoupling of channel from receptor suggests that the muscarinic receptor and the IAch channel are separate molecular structures. Membrane conductance responses to sequential activation of muscarinic and β-adrenergic receptors demonstrate that in contrast to the muscarinic inhibition of Isi, muscarinic stimulation of IACh is mediated by a G protein via a pathway that does not involve adenylate cyclase. Taken together, the results support the notion that agonist is required to induce GppNHp binding and/or activation of the G proteins. Once triggered by agonist, the control system remains maximally activated, thereby transforming the cell so that it no longer responds to subsequent homologous receptor-mediated signals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Rodbell, M. Nature 284, 17–22 (1980).
Gilman, A. G. Cell 36, 577–579 (1984).
Birnbaumer, L. et al. Recent Prog. Horm. Res. 41 (in the press).
Sperelakis, N. Membrane Biochem. 5, 131–166 (1984).
Hamill, O. P., Marty, A., Neher, E., Sakmann, B. & Sigworth, F. J. Pflügers Arch. ges. Physiol. 391, 85–100 (1981).
Hume, J. R. & Giles, W. J. gen. Physiol. 78, 18–43 (1981).
Momose, Y., Giles, W. & Szabo, G. Biophys. J. 45, 20–21 (1984).
Galper, J. B. & Ganz, P. in Receptor Science in Cardiology (eds Haft, J. I. & Karliner, J. S.) (Futura, New York, 1984).
Galper, J. B., Dziekan, L. C. & Smith, T. W. J. biol. Chem. 259, 7382–7390 (1984).
Burgisser, E., De Lean, A. & Lefkowitz, R. J. Proc. natn. Acad. Sci. U.S.A. 79, 1732–1736 (1982).
Pappano, A. I. & Inoue, D. Fedn Proc. 43, 2607–2612 (1984).
Hume, J. R. & Giles, W. J. gen. Physiol. 81, 153–194 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Breitwieser, G., Szabo, G. Uncoupling of cardiac muscarinic and β-adrenergic receptors from ion channels by a guanine nucleotide analogue. Nature 317, 538–540 (1985). https://doi.org/10.1038/317538a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/317538a0
This article is cited by
-
The voltage-sensitive cardiac M2 muscarinic receptor modulates the inward rectification of the G protein-coupled, ACh-gated K+ current
Pflügers Archiv - European Journal of Physiology (2018)
-
Cross-signaling in metabotropic glutamate 2 and serotonin 2A receptor heteromers in mammalian cells
Pflügers Archiv - European Journal of Physiology (2016)
-
X-ray structure of the mammalian GIRK2–βγ G-protein complex
Nature (2013)
-
Regulation of the muscarinic K+ channel by extracellular ATP through membrane phosphatidylinositol 4,5‐bisphosphate in guinea‐pig atrial myocytes
British Journal of Pharmacology (2005)
-
Anti-cholinergic effect of verapamil on the muscarinic acetylcholine receptor-gated K+ channel in isolated guinea-pig atrial myocytes
Naunyn-Schmiedeberg's Archives of Pharmacology (2004)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.