Abstract
Study design: Prospective investigation using serial urodynamic studies.
Objective: To evaluate type of neurogenic bladder and to observe changes of autonomous detrusor contraction (ADC) after the normalization of the compliance and capacity of the bladder in cauda equina injury.
Setting:Spinal Cord Injury Unit, Yonsei Rehabilitation Hospital, Seoul, Korea.
Methods: Urodynamic studies were performed in 50 patients with complete cauda equina injury from trauma with an infusion ratio of 30 ml/min. Findings of urodynamic studies and clinical features of patients with low compliance were compared with those of the normal compliant patients. Fourteen patients with low compliance received oral administration of oxybutynin and propiverine and intravesical instillation of oxybutynin to increase the compliance and capacity of the bladder, and follow-up urodynamic studies to monitor the change were undertaken.
Results: Bladder compliance was decreased in 14 (28%) patients and normal in 36 (72%) patients. There was a significantly long time interval between the onset of injury and the initiation of rehabilitative treatment in the neurogenic bladder group with low compliance when compared to those of the normal compliance group (P<0.05). Clean intermittent catheterization was used as the voiding method, significantly less than the normal compliance group (P<0.05). ADC was observed in six out of fourteen patients with low compliance neurogenic bladder, but none in the normal compliance group. Upon the completion of conservative treatment, ADC disappeared in four patients whose compliance and capacity of the bladder were normalized on follow-up urodynamic studies.
Conclusion: ADC was only observed in the low compliant bladder and as ADC disappeared when compliance and capacity of the bladder was normalized; low compliance appeared to be the main cause of ADC. In addition, this study supports that the maintenance of compliance of the bladder may be the most important factor in the management of neurogenic bladder.
Similar content being viewed by others
Introduction
Effective management of the neurogenic bladder in patients with spinal cord injury is an important part of rehabilitation, since it does not only improve the quality of life but also shortens hospital stay due to complications. However, clinical manifestations of neurogenic bladder are variable depending upon the level and completeness of spinal cord injury. Methods of treatment and management are also variable, including behavior therapy, adjuvant therapy, medication and surgery.1,2
In general, an areflexic neurogenic bladder usually appears in patients with cauda equina injury due to damage of the pelvic nucleus and pudendal nucleus in the sacral cord.3 However several studies reported atypical findings such as autonomic detrusor contraction (ADC) or low compliance in cauda equina injury.4,5,6 This atypical neurogenic bladder causes a continuous increase of intravesical pressure during urination, and this, subsequently, leads to a deterioration of renal function.7,8,9 Therefore, it is important to increase the compliance of the bladder.10,11
In this study, we first evaluate the types of neurogenic bladder in patients with cauda equina injury using neurophysiologic and urodynamic studies. Second, in cases with a low compliance bladder, we re-evaluated the change in bladder status after the normalization of the maximal capacity and compliance of the bladder by conservative treatment, including oral administration and intravesical instillation of oxybutynin chloride, oral administration of propiverine hydrochloride, and intermittent catheterization. Lastly, the characteristics of the bladder with ADC were investigated, and the persistency of ADC after conservative treatment was also evaluated.
Materials and methods
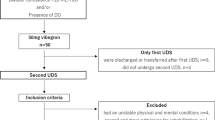
From 1998 to 2000, the 50 patients included were admitted to Yonsei Rehabilitation Hospital for traumatic cauda equina injury. Those who had active urinary tract infection during the study, an associated suprasacral lesion, incomplete cauda equina lesion, and non-traumatic origin were excluded. The subjects underwent electrophysiologic study; nerve conduction study, electromyography, pudendal nerve somatosensory evoked potential study, and bulbocavernous reflex study to confirm complete injury of cauda equina. There were 26 males and 24 females with the average age of 31.1 years. Informed consents were obtained from each patient, and the medical ethical committee at this hospital approved this study.
Age, sex, voiding method and duration of rehabilitative treatment of the subjects after injury was evaluated, and the sensory and motor status and reflexes of the affected area were also clinically evaluated. A urodynamic study was performed with a Duet urodynamic machine (Dantec, Denmark), and the maximal bladder capacity, maximal detrusor pressure, compliance and involuntary detrusor contraction were measured during the filling phase. This study was done by an infusion of normal saline at the rate of 30 ml/min through a double lumen catheter, and a pressure sensor device placed at the rectum measured the abdominal pressure. Measuring the volume of the bladder until an abrupt rise in detrusor pressure during filling phase, and then dividing this volume by the detrusor pressure calculated compliance. We defined ADC as an involuntary contraction with an increase of more than 15 cmH2O in detrusor pressure, and the normal bladder capacity was defined when the bladder volume was more than 400 ml. Those whose compliance was higher than 15 ml/cmH2O were assigned to the normal compliance group, while those whose compliance was lower than 15 ml/cmH2O were assigned to the low compliance group. The time span to the hospitalization following injury, voiding method prior to admission, interval between the onset of injury and rehabilitative treatment, maximal bladder capacity, maximal detrusor pressure and presence or absence of ADC were compared between the two groups.
In the low compliance group or those with ADC, 5 mg of oxybutynin chloride in 20 ml of distilled water were again mixed with 20 ml of normal saline, and this mixture was intravesically instilled twice a day for 7 days for an interval of 4 weeks.12,13 In addition, 20 mg of oxybutynin and between 20 and 40 mg of propiverine were given orally. When the bladder volume reached more than 400 ml and when no incontinence was clinically observed following intermittent catheterization, 5 times per day, the medication was stopped. After 3 days of discontinuation of medication, follow-up urodynamic studies were repeatedly performed to evaluate any changes of neurogenic bladder by the same method.
For statistical analysis, SPSS statistical program for Windows was used, and independent t-test, paired t-test and Chi-square test were used. It was defined statistically significant when P<0.05.
Results
Comparison of clinical and urodynamic parameters between two groups
Among 50 patients, the compliance was within the normal range in 36 patients whereas 14 patients had decreased compliance (Table 1). No difference with statistical significance was observed between two groups in age and sex. In the low compliance group, the mean time from the onset of injury to hospitalization was 7.8 months, while the mean time interval from the onset of injury to rehabilitative treatment was 3.4 months in the normal compliance group, showing a longer time interval in the low compliance group (P<0.05). For the normal compliance group, the number of clean intermittent catheterizations as the voiding method was greater than that of the low compliance group (P<0.05).
In the low compliance group, the mean compliance and mean maximal bladder capacity were 8.3 ml/cmH2O and 266.1 ml, respectively, lower than those of the normal compliance group, and were statistically significant (P<0.01). On the other hand, the mean maximal detrusor pressure in the low compliance group was 58.1 cmH2O, higher than that in the normal compliance group (P<0.01) (Table 2).
ADC in the low compliance group
ADC during the filling phase was only observed in six of 14 patients in the low compliance group, but was not observed in the normal compliance group. In these six patients, the mean compliance (4.3 ml/cmH2O) and mean maximal bladder capacity (128.8 ml) were lower than those of the other patients without ADC (n=8), while the mean maximal detrusor pressure (70.8 cmH2O) was higher than that of those eight patients without ADC, and was statistically significant (P<0.05) (Table 3).
Effect of normalization of compliance in the low compliance group
All 14 patients in the low compliance group received both oral administration and intravesical instillation of oxybutynin and oral administration of propiverine while intermittent catheterization was simultaneously performed. The medication was stopped when the bladder capacity was clinically measured to be more than 400 ml with no evident incontinence, and an urodynamic study was performed 3 days after for the evaluation of changes in the bladder. The mean duration of therapy to follow-up was 54.2±12.7 days. The mean compliance and mean maximal bladder capacity increased from 8.3 ml/cmH2O and 266.1 ml at the beginning to 25.0 ml/cmH2O and 340.9 ml with statistical significance (P<0.05). Meanwhile, the mean maximal detrusor pressure decreased from 58.1 cmH2O to 30.8 cmH2O, also with statistical significance (P<0.05) (Table 4).
Effect of normalization of compliance and capacity of bladder in ADC
For those six patients with ADC detected on urodynamic study, follow-up showed that the ADC disappeared in four of them. In these cases, the compliance and maximal bladder capacity returned to the normal range. Although an increase in the compliance and maximal bladder capacity of the bladder was seen, they were not within the normal range, and especially, the compliance was minimally improved in the remaining two patients with persistent ADC (Table 5 and Figure 1).
Urodynamic study in 26-year-old female patient with cauda equina injury showed a significant increment in the maximal bladder capacity from 98 to 500 ml after 2 months of conservative therapy. (A) Prior to therapy, the detrusor pressure began to increase at 90 ml of infusion volume. The maximal detrusor pressure was 87 cmH2O while the compliance was 9.8 ml/cmH2O. The autonomous detrusor contraction (ADC) was observed in the filling phase. (B) The follow-up urodynamic study following the conservative treatment showed typical findings of areflexic neurogenic bladder. The maximal detrusor pressure decreased from 87 to 21 cmH2O and the compliance increased from 9.8 to 25.0 ml/cmH2O, respectively
Discussion
In patients with spinal cord injury, the goal of management of neurogenic bladder is the preservation of renal function and maintenance of urinary continence for a better social integration.1,14 Therefore, the low compliance neurogenic bladder is clinically receiving special attention because it causes deterioration of renal function as well as causing hydronephrosis.15,16 It is well known that patients with cauda equina injury involving the sacral cord containing the pelvic nucleus and the pudendal nucleus, typically show the high compliance areflexic neurogenic bladder, but the low compliance and/or small volume bladder is also clinically encountered in these patients. In the study done by Gunasekera et al,4 16 of 34 subjects showed low compliance, and low compliance bladder was observed in 28% of our patients in this study. When low compliance bladder was compared with the normal compliance group, the interval between the onset of injury and the initiation of rehabilitative treatment was significantly longer, and there were a higher number of voiding methods either by Crede's maneuver or by indwelling catheter prior to admission.
Previous studies proposed three possible mechanisms for the development of a low compliance bladder. First, they suggested that it was due to the changes of passive properties of the detrusor muscle. In this hypothesis, the proposed mechanism was prolonged infection and stimulation causing structural changes in the bladder wall such as fibrosis.17,18,19,20 The second possible mechanism is of myogenic origin,21 the denervated muscles were changed to the increased collagen fibers and hypertrophy of smooth muscles fibers.22,23 The third possible mechanism is of neurogenic origin. Levin et al24 stated that hypertrophy of the detrusor muscle could induce a hyperactivity of nerve and muscle cell, Neal et al25 and Norlen26 asserted that low compliance was induced by autonomous contraction of newly produced cholinergic nerve fibers due to prolonged injury of parasympathetic nerves.
In this study, several cases in the low compliance group were managed by indwelling catheterization without intermittent clamping during the early phase of injury. We thought that this might hinder the bladder to be properly distended, and further decrease of compliance of the bladder resulted. In addition, since the time interval between the onset of injury and the rehabilitative treatment was relatively longer and since voiding methods prior to admission were significantly different, changes in passive properties of the detrusor muscle is believed to be the main cause of low compliance.
In the patients with cauda equina injury, a more serious clinical problem is the hyperreflexic bladder due to ADC. In this study, six (42.9%) out of 14 patients with low compliance bladder had ADC, and these patients showed a significant decrease in the compliance and capacity of the bladder compared to those without ADC. The study done by Gunasekera et al,4 reported that among 34 patients with lower motor neuron injury, detrusor hyperreflexia was observed in 20 patients. Among these 20 patients, low compliance bladder was found in 16 and normal compliance in four. In this study, ADC was observed only in the low compliance bladder.
Although it is still uncertain what the exact mechanism of ADC in low compliance bladder due to lower motor neuron injury is, Shingleton and Bodner16 observed ADC in the isolated detrusor muscle specimen of cats with cauda equina injury and concluded that it was myogenic in origin. However, Norlen26 asserted that this contraction of the detrusor muscle resulted from the action of the sympathetic nervous system on the alpha-adrenergic receptor. On the other hand, Kinder and Mundy27 claimed that ADC developed by the peri-vesical nerves, and Langley and Anderson28 reported that the detrusor muscle contracted when the lumbar spinal cord was stimulated electrically. Moreover, Elbadawi and Schenk29 documented that there was a peripheral ganglion, called `urogenital short neuron system', on the wall of the bladder and urethra, and they claimed that complete demyelination of the bladder did not occur even after the nerve damage.
In this study, the resolution of ADC was noted in four of six patients in which the compliance and capacity of the bladder was normalized after 2 months of conservative treatment. Oxybutynin chloride has anti-muscarinic, antispasmodic and local anesthetic actions on smooth muscle,1,14 whereas propiverine hydrochloride is a calcium channel blocker that has antispasmodic and anticholinergic actions on smooth muscle.30,31 Therefore, it is difficult to determine whether the decrease in local cholinergic nerve activity or antispasmodic action is responsible for the normalization of compliance, resulting in the resolution of ADC. Nevertheless, our results indicate that since ADC is caused by the decrease in the compliance and capacity of the bladder, it is imperative to maintain the adequate capacity and compliance of the bladder for the management of neurogenic bladder in patients with cauda equina injury.
For the further investigation of the origin of ADC, we plan to determine whether there is any effect on ADC when only a pure anticholinergic or pure antispasmodic drug is used. Moreover, a conventional urodynamic study using warm normal saline in low compliance bladder, an ice saline provocative urodynamic study that can determine the activity of unmyelinated c-fiber study32,33 and a KCl provocative urodynamic study that can identify the detrusor instability due to chronic inflammation34,35 will be simultaneously performed for a more accurate evaluation.
In summary, our results suggested that in patients with cauda equina injury, the clinical history and physical examination, combined with a urodynamic study should be performed to evaluate the pattern of neurogenic bladder, such as the presence of a hyperreflexic or low compliance bladder. In addition, through the evaluation of change of the bladder after the normalization of the compliance and capacity of the bladder, we were able to assess the importance of the early initiation of rehabilitative treatment following injury. Finally, despite the fact that the conservative treatment was effective in normalizing compliance and resolving ADC in patients with atypical neurogenic bladder, long-term effect and exact mechanism have not been proven, and a further study is needed.
Conclusions
From 1998 to 2000 at Yonsei Rehabilitation Hospital, urodynamic studies were performed in 50 patients with traumatic cauda equina injury. In the low compliance group, combined medical treatment and intermittent catheterization was used to control incontinence. After conservative treatment, follow-up studies were performed to evaluate the change of bladder status and change of ADC, and the following results were obtained.
-
1
28% (14) of the 50 subjects belonged to the low compliance bladder, and these patients showed a longer time interval between the onset of injury to the initiation of rehabilitative treatment when compared to those of the normal compliance group with a statistically significant difference (P<0.05).
-
2
The low compliance group had the higher maximal detrusor pressure but a smaller maximal bladder capacity with a statistical significance (P<0.01).
-
3
ADC was only observed in six (42.8%) patients of the low compliance group, and after conservative treatment, ADC disappeared in four patients with normalized compliance and maximal bladder capacity.
Therefore, it is important to maintain the adequate compliance and capacity of the bladder in patients with cauda equina injury immediately following injury. Moreover, considering the fact that ADC disappeared after the normalization of the compliance and maximal bladder capacity, ADC is thought to arise from the activation of detrusor muscle induced by a decrease in compliance.
References
Cardenas DD, Mayo ME . Management of bladder dysfunction In: Braddom RL (ed) Physical Medicine and Rehabilitation 2nd edn WB Saunders: Philadelphia 2000 pp 561–577
Jamil F . Towards a catheter free status in neurogenic bladder dysfunction: a review of bladder management options in spinal cord injury Spinal Cord 2001 39: 355–361
Blaivas JG . The neurophysiology of micturition. A clinical study of 550 patients J Urol 1982 127: 958–963
Gunasekera WSL, Richardson AE, Seneviratne KN, Eversden ID . Detrusor hyperreflexia in neurogenic bladder disorders caused by localized partial lesions of the spinal cord and cauda equina Surg Neurol 1983 20: 63–66
Denny-Brown D, Robertson GE . The state of the bladder and its sphincters in complete transverse lesion of the spinal cord and cauda equina Brain 1933 56: 397–462
Kaplan SA, Chancellor MB, Blaivas JG . Bladder and sphincter behavior in patients with spinal cord lesion J Urol 1991 146: 113–117
Coolsaet BL . Detrusor pressure versus wall stress in relation to ureterovesical resistance Neurourol Urodynam 1982 1: 105–112
Ghoniem GM, Bloom DA, McGuire EJ . Bladder compliance in meningomyelocele children J Urol 1989 141: 1404–1406
Madersbacher S et al. Interrelationships of bladder compliance with age, detrusor instability, and obstruction in elderly men with lower urinary tract symptoms Neurol Urodynam 1999 18: 3–15
McGuire EJ, Woodside JR, Borden TA . Upper urinary tract deterioration in patients with myelodysplasia and detrusor hypertonia: a follow-up study J Urol 1983 129: 823–826
Gerridzen RG, Thijssen AM . Risk factors for upper tract deterioration in chronic spinal cord injury patients J Urol 1992 147: 416–418
Shin JC et al. Clinical effectiveness of intravesical oxybutynin instillation in spinal cord injured patients with hyperreflexic or hypertonic neurogenic bladder J Korean Acad Rehab Med 2000 24: 28–34
Madersbacher H, Jilig G . Control of detrusor hyperreflexia by the intravesical instillation of oxybutynin hydrochloride Paraplegia 1991 29: 84–90
Linsenmeyer TA, Stone JM . Neurogenic bladder and bowel dysfunction In: DeLisa JA (ed) Rehabilitation Medicine: Principles and Practices 3rd edn Lippincott-Ravens: Philadelphia 1998 pp 1073–1094
Hackler RH, Hall MK, Zampieri TA . Bladder hypocompliance in the SCI population J Urol 1989 141: 1390–1393
Shingleton B, Bodner D . The development of urologic complications in relationship to bladder pressure in spinal cord injured patients J Am Paraplegia Soc 1993 16: 14–17
Abrams P, Blaivas JG, Stanton SL, Anderson JT . Standardization of terminology of lower urinary tract function Neurourol Urodynamics 1988 7: 403–427
German K, Bedwani J, Davies J, Brading AF . An assessment of the contribution of visco-elastic factors in the aetiology of poor compliance in the human neuropathic bladder Br J Urol 1994 74: 744–748
Macneil HF, Bradling AF, Williams JH . Cause of low compliance in a guinea pig model of instability and low compliance Neurourol Urodynamics 1992 11: 47–52
McGuire EJ, Morrissey SG . The development of neurogenic vesical dysfunction after experimental spinal cord injury or sacral rhizotomy in non-human primates J Urol 1982 128: 1390–1393
Sherrington CS . Notes on the arrangement of some motor fibers in the lumbosacral plexus J Physiol (Lond) 1982 13: 621–672
Ghoniem GM . Effect of bilateral sacral decentralization on detrusor contractility and passive properties in dogs Neurourol Urodynam 1984 3: 23–30
Brading AF, Turner WH . The unstable bladder; toward a common mechanism Br J Urol 1994 73: 3–8
Levin RM et al. Studies on experimental bladder outlet obstruction in the cat: long term functional effects J Urol 1999 148: 939–943
Neal DE, Bogue PR, Williams RE . Histological appearances of the nerves of the bladder in patients with denervation of the bladder after excision of the rectum Brit J Urol 1982 54: 658–666
Norlen L . The autonomic bladder, a clinical and experimental study Scand J Urol Nephrol suppl 1976 36: 1–28
Kinder RB, Mundy AR . Pathophysiology of idiopathic detrusor instability and detrusor hyper-reflexia. An in vitro study of human detrusor muscle Br J Urol 1987 60: 509–515
Langley JN, Anderson HK . The innervation of pelvic and adjoining viscera J Physiol 1895 19: 71–131
Elbadawi A, Schenk EA . A new theory of the innervation of the bladder musculature. IV. Innervation of the vesicourethral junction and external urethral sphincter J Urol 1974 111: 613–615
Haruno A et al. Effects of propiverine hydrochloride and its metabolites on isolated guinea pig urinary bladder Folio Pharmacol Jpn 1989 94: 145–150
Muller C et al. Kinetics of propiverine assessed by radioreceptor assay in poor and extensive metabolites of debrisoquine Eur J Drug Metab Pharmacokinet 1993 18: 265–272
Ronzoni G, Menchinelli P, Manca A, De Giovanni L . The ice water test in the diagnosis and treatment of the neurogenic bladder Br J Urol 1997 79: 698–701
Shin JC et al. Ice water urodynamic study in neurogenic bladder of spinal cord injured patients J Korean Acad Rehab Med 2000 24: 439–445
Hohlbrugger G . Urinary potassium and the overactive bladder Br J Urol Int 1999 83: 22–28
Shin JC et al. The clinical usefulness of KCl provocative urodynamic study in spinal cord injury patients with neurogenic bladder J Korean Acad Rehab Med 2000 24: 908–916
Acknowledgements
This article was present at the 40th Annual Scientific Meeting of International Medical Society of Paraplegia.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shin, J., Park, Ci., Kim, H. et al. Significance of low compliance bladder in cauda equina injury. Spinal Cord 40, 650–655 (2002). https://doi.org/10.1038/sj.sc.3101380
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.sc.3101380
Keywords
This article is cited by
-
Urodynamic study findings and related influential factors in pediatric spastic cerebral palsy
Scientific Reports (2022)
-
The determination of sensation-dependent bladder emptying time in patients with complete spinal cord injury above T11
Spinal Cord (2008)
-
The Effects of Long-Term Spinal Cord Injury on Mechanical Properties of the Rat Urinary Bladder
Annals of Biomedical Engineering (2008)
-
Effect of the intravesical resiniferatoxin instillation evaluated by the ice provocative urodynamic study
Spinal Cord (2006)
-
Quantification of Bladder Smooth Muscle Orientation in Normal and Spinal Cord Injured Rats
Annals of Biomedical Engineering (2005)