Abstract
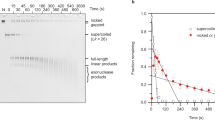
In interphase nuclei and metaphase chromosomes, eukaryotic chromosomal DNA is arranged in negatively supercoiled loops whose length averages several tens of kilobases (kb). Supercoiling indicates that the loops are tenaciously bound at both ends through the periodic attachment of DNA to a non-histone protein structural support, termed the nuclear matrix, skeleton or cage in interphase nuclei and the scaffold in metaphase chromosomes1–16. The looped organization has been envisaged to be important in DNA replication2,11,15,17–25 and transcription26–28. We report here a close relationship between average loop length and replicon size in different animal and plant species. Autoradiographic experiments support the hypothesis that DNA synthesis occurs at fixed sites on the nuclear matrix15,20. We also describe a modification of the looped organization occurring during early embryonic development in Xenopus laevis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Piñon, R. & Salts, Y. Proc. natn. Acad. Sci. U.S.A. 74, 2850–2854 (1977).
McCready, S. J., Godwin, J., Mason, D. W., Brazell, I. A. & Cook, P. R. J. Cell Sci. 46, 365–386 (1980).
Benyajati, C. & Worcel, A. Cell 9, 393–407 (1976).
Hartwig, M. Acta biol. med. germ. 37, 421–432 (1978).
Marsden, M. P. F. & Laemmli, U. K. Cell 17, 849–858 (1979).
Ide, T. Nakane, M., Anzai, K. & Andoh, T. Nature 258, 445–447 (1975).
Igo-Kemenes, T. & Zachau, H. G. Cold Spring Harb. Symp. quant. Biol. 42, 109–118 (1977).
Okada, T. A. & Comings, D. E. Chromosoma 72, 1–14 (1979).
Mattern, M. R. & Painter, R. B. Biochim. biophys. Acta 563, 293–305 (1979).
Mattern, M. R. & Painter, R. B. Biochim. biophys. Acta 563, 306–312 (1979).
Vogelstein, B., Pardoll, D. M. & Coffey, D. S. Cell 22, 79–85 (1980).
Laemmli, U. K. et al. Cold Spring Harb. Symp. quant. Biol. 42, 351–360 (1977).
McCready, S. J. Cox, B. S. & McLaughlin, C. S. Expl Cell Res. 108, 473–478 (1977).
Cook, P. R. & Brazell, I. A. Nucleic Adids Res. 8, 2895–2906 (1980).
Shaper, J. H. et al. Adv. Enzyme Regulation 17, 213–248 (1979).
Comings, D. E. in The Cell Nucleus Vol. 4 (ed. Bush, H.) 345–371 (Academic, New York, 1978).
Wilkins, A. S. J. theor. Biol. 89, 715–717 (1981).
Berezney, R. Expl Cell Res. 123, 411–414 (1979).
Berezney, R. & Buchholtz, L. A. Expl Cell Res. 132, 1–13 (1981).
Pardoll, D. M., Vogelstein, B. & Coffey, D. S. Cell 19, 527–536 (1980).
Berezney, R. & Coffey, D. S. Science 189, 291–293 (1975).
Dijkwel, P. A., Mullenders, L. H. F. & Wanka, F. Nucleic Acids Res. 6, 219–230 (1979).
Hunt, B. F. & Vogelstein, B. Nucleic Acids Res. 9, 349–363 (1981).
Nelkin, B. D., Pardoll, D. M. & Vogelstein, B. Nucleic Acids Res. 8, 5623–5633 (1980).
Buckler-White, A. J., Humphrey, G. W. & Pigiet, V. Cell 22, 37–46 (1980).
Jackson, D. A., McCready, S. J. & Cook, P. R. Nature 292, 552–555 (1981).
Smith, G. R. Cell 24, 599–600 (1981).
Robinson, S. I., Nelkin, B. D. & Volgelstein, B. Cell 28, 99–106 (1982).
Edenberg, H. J. & Huberman, J. A. A. Rev. Genet. 9, 245–284 (1975).
Dingman, C. W. J. theor. Biol. 38, 187–195 (1974).
Callan, H. G. Phil. Trans. R. Soc. B181, 19–41 (1972).
Blumenthal, A. B., Kriegstein, H. J. & Hogness, D. S. Cold Spring Harb. Symp. quant. Biol. 38, 205–224 (1974).
Huberman, J. A. & Riggs, J. D. J. molec. Biol. 39, 327–341 (1968).
Van't Hof, J. Expl Cell Res. 93, 95–104 (1975).
McFarlane, P. W. & Callan, H. G. J. Cell Sci. 13, 821–839 (1973).
Hand, R. & Tamm, I. J. molec. Biol. 82, 175–183 (1974).
Nieuwkoop, P. D. & Faber, J. Normal Tables of Xenopus laevis (North Holland, Amsterdam, 1967).
Funderud, S., Andreassen, R. & Haugli, F. Nucleic Acids Res. 6, 1417–1431 (1979).
Rasch, E. M., Barr, H. J. & Rasch, R. W. Chromosoma 33, 1–18 (1971).
Bachman, K. Chromosoma 37, 85–93 (1972).
Buongiorno-Nardelli, M., Amaldi, F. & Lava-Sanchez, P. Nature new Biol. 238, 134–137 (1972).
Van't Hof, J. & Sparrow, A. H. Proc. natn. Acad. Sci. U.S.A. 49, 897–902 (1963).
Maul, G. G. & Deaven, L. J. Cell Biol. 73, 748–760 (1977).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Buongiorno-Nardelli, M., Micheli, G., Carrĩ, M. et al. A relationship between replicon size and supercoiled loop domains in the eukaryotic genome. Nature 298, 100–102 (1982). https://doi.org/10.1038/298100a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/298100a0
This article is cited by
-
Transcription-coupled structural dynamics of topologically associating domains regulate replication origin efficiency
Genome Biology (2021)
-
Replication timing of large Sorex granarius (Soricidae, Eulipotyphla) telomeres
Protoplasma (2018)
-
Limited nucleotide pools restrict Epstein–Barr virus-mediated B-cell immortalization
Oncogenesis (2017)
-
Transformation-induced changes in the DNA-nuclear matrix interface, revealed by high-throughput analysis of DNA halos
Scientific Reports (2017)
-
4D Visualization of replication foci in mammalian cells corresponding to individual replicons
Nature Communications (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.