Abstract
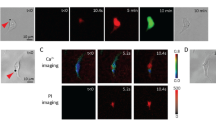
The transmission of signals between cells of the early preimplantation embryo may be important in the recognition of relative cell position, thereby influencing the direction of cell differentiation1–5. Gap junctional (water-permeable) channels may provide one route for such signal transmission6. A previous report7, in which intact, zona-free mouse embryos were impaled by microelectrodes, demonstrated that gap junction-mediated dye transfer and electrical continuity were absent between blastomeres of two- and four-cell embryos but appeared during the eight-cell stage. It is at the eight-cell stage that compaction occurs, during which cells polarize3, flatten on each other3,8 and show the first morphological evidence of intercellular junctions9,10. We report here the use of a simple, non-iontophoretic method to detect the passage of a low-molecular-weight dye between aggregated blastomeres of the early preimplantation mouse embryo. Cells are incubated in the nonfluorescent, non-polar reagent, 6-carboxyfluorescein diacetate (CFDA)11,12, which enters the cell freely, where it becomes entrapped following enzymatic conversion13 to the hydrophilic fluorophore 6-carboxyfluorescein (CF; molecular weight 370). The labelled cells may then be aggregated with unlabelled cells and the formation of communicating channels between cells is revealed by the passage of dye from labelled to unlabelled blastomeres. CFDA provides better results than previously used fluorescein esters14,15, which hydrolyse to a much less hydrophilic fluorophore that leaks from the cell relatively quickly11,15. Using CFDA we have confirmed that the capacity to form channels is lacking at all stages from the mature oocyte to the four-cell embryo but appears during the first 6 h of the eight-cell stage, preceding the fully compacted state.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Tarkowski, A. K. & Wroblewska, J. J. Embryol. exp. Morph. 18, 155–180 (1967).
Hillman, N., Sherman, M. I. & Graham, C. F. J. Embryol. exp. Morph. 28, 263–278 (1972).
Ziomek, C. A. & Johnson, M. H. Cell 21, 935–942 (1980).
Gardner, R. L. J. Embryol. exp. Morph. 28, 279–312 (1972).
Rossant, J. J. Embryol. exp. Morph. 33, 991–1001 (1975).
Lo, C. W. in Development in Mammals Vol. 4 (ed. Johnson, M. H.) 39–80 (Elsevier, Amsterdam, 1980).
Lo, C. W. & Gilula, N. B. Cell 18, 399–409 (1979).
Lehtonen, E. J. Embryol. exp. Morph. 58, 231–249 (1980).
Ducibella, T., Albertini, D. F., Anderson, E. & Biggers, J. D. Devl Biol. 45, 231–250 (1975).
Magnuson, T., Jacobson, J. B. & Stackpole, C. W. Devl Biol. 67, 214–224 (1978).
Bruning, J. W., Kardol, M. J. & Arentzen, R. J. immun. Meth. 33, 33–44 (1980).
Van der Poel, J. J., Kardol, M. H., Goulmy, E., Blokland, E. & Bruning, J. W. Immun. Lett. 2, 187–193 (1981).
Rotmann, B. & Papermaster, B. W. Proc. natn. Acad. Sci. U.S.A. 55, 134–141 (1966).
Sellin, D. et al. Eur. J. Immun. 4, 189–193 (1974).
Loewenstein, W. R. Biochim. biophys. Acta 560, 1–65 (1979).
Whittingham, D. B. & Wales, R. G. Aust. J. biol. Sci. 22, 1065–1068 (1969).
Gilula, N. B., Epstein, M. L. & Beers, W. H. J. Cell Biol. 78, 58–75 (1978).
Dahl, G., Azarnia, R. & Werner, R. Nature 289, 683–685 (1981).
Johnson, M. H. & Ziomek, C. A. J. Cell Biol. 91, 303–308 (1981).
Calarco, P. G. & Epstein, C. J. Devl Biol. 32, 208–213 (1973).
Ducibella, T. & Anderson, E. Devl Biol. 47, 45–58 (1975).
Reeve, W. J. D. & Ziomek, C. A. J. Embryol. exp. Morph. 62, 339–350 (1981).
Pratt, H. P. M. in Development in Mammals Vol. 3 (ed. Johnson, M. H.) 83–129 (Elsevier, Amsterdam, 1978).
Sheridan, J. D., Finbow, M. E. & Pitts, J. D. Expl Cell Res. 123, 111–117 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Goodall, H., Johnson, M. Use of carboxyfluorescein diacetate to study formation of permeable channels between mouse blastomeres. Nature 295, 524–526 (1982). https://doi.org/10.1038/295524a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/295524a0
This article is cited by
-
Uptake and compartmentalisation of fluorescent probes byPisolithus tinctorius hyphae: evidence for an anion transport mechanism at the tonoplast but not for fluid-phase endocytosis
Protoplasma (1997)
-
The cellular pathway of sucrose transport in developing cotyledons ofVicia faba L. andPhaseolus vulgaris L.: a physiological assessment
Planta (1995)
-
A simple fluorometric assay for quantifying the adhesion of tumour cells to endothelial monolayers
Clinical & Experimental Metastasis (1995)
-
Intercellular communication between cultured granulosa cells of the marmoset (Callithrix jacchus)
Cell & Tissue Research (1992)
-
Cell-to-cell communication competence in simian virus 40-transfected rat ovarian cells is reduced following tumor selection
In Vitro Cellular & Developmental Biology - Animal (1992)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.