Abstract
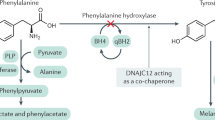
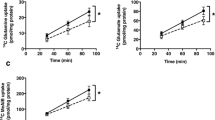
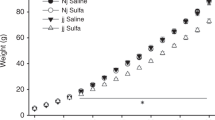
THE mammalian placenta can concentrate amino acids and release them into the foetal circulation, where they appear at levels higher than on the maternal side1. This mechanism provides a larger pool for foetal protein synthesis, but normal placental function can become a handicap in some situations. In the presence of an excess of amino acids the placenta exacerbates the maternal imbalance and produces a prolonged hyperaminoacidaemia in the foetus2. This in turn may affect the transplacental transport of other amino acids. Such can be the case in ‘material phenylketonuria (PKU)’, which often results in offspring with brain damage and mental retardation3. Using an animal model of ‘maternal PKU’ we have studied alterations in tissue levels and placental transport of L-tyrosine (Tyr), L-tryptophane (Trp) and certain foetal brain enzymes. We have found a reduced foetal tissue concentration of Trp and impaired foetal brain uptake of Tyr and Trp in the foetuses of hyper-phenylalaninaemic dams, as well as reduced brain pyruvate kinase activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Ghadimi, H., and Pecora, P., Pediatrics, 33, 500–506 (1964).
Wapnir, R. A., and Dierks-Ventling, C., Biol. Neonate, 17, 373–380 (1971).
MacCready, R. A., and Levy, H. L., Am. J. Obstet. Gynec., 113, 121–128 (1972).
Rosso, P. Science, 187, 648–650 (1975).
Weber, G., Proc. natn. Acad. Sci. U.S.A., 63, 1365–1369 (1969).
Adlard, B. P. F., and Dobbing, J., Brain Res., 28, 97–107 (1971).
Wong, P. W. K., O'Flynn, M. E., and Inouye, T., Clin. chim. Acta, 10, 1098–1104 (1964).
Wapnir, R. A., and Stevenson, J. H., Clin. chim. Acta, 26, 203–206 (1969).
Miller, A. L., Hawkins, R. A., and Veech, R. L., Science, 179, 904–906 (1973).
Gimenez, C., Valdivieso, F., and Mayor, F., Biochem. Med., 11, 81–86 (1974).
Cohen, L. L., and Huang, K. C., Am. J. Physiol., 206, 647–652 (1964).
Wooley, D. W., and van der Hoeven, T., Science, 144, 883–884 (1964).
Hamon, and Glowinski, J., Life Sci., 15, 1533–1548 (1974).
Diez, J. A., Sze, P. Y., and Ginsburg, B. E., Brain Res., 104, 396–400 (1976).
Peters, D. A. V., Biochem. Pharmac., 21, 1051–1053 (1972).
Wapnir, R. A., and Lifshitz, F., Clin. chim. Acta, 54, 349–356 (1974).
Fernstrom, J. D., and Wurtman, R. J., Science, 173, 149–152 (1971).
Angeli, E., Denman, A. R., Harris, R. F., Kirman, B. H., and Stern, J., Dev. Med. Child Neurol., 16, 800–807 (1974).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
WAPNIR, R., MOAK, S. & LIFSHITZ, F. Decreased foetal amino acid uptake, brain pyruvate kinase and intrauterine damage in maternal PKU. Nature 265, 647–648 (1977). https://doi.org/10.1038/265647a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/265647a0
This article is cited by
-
Effects of phenylalanine loading on protein synthesis in the fetal heart and brain of rat: an experimental approach to maternal phenylketonuria
Journal of Inherited Metabolic Disease (1986)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.