Abstract
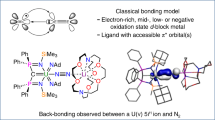
WE have reported1 the reduction of dinitrogen, in cis-[M(N2)2-(PMe2Ph)4] (A, where M = Mo or W), to ammonia, on treatment with sulphuric acid in methanol at 20 °C. The reaction of the molybdenum complex is complete in 3–4 min but the yield (≤ 0.8 mol per Mo atom) is only about half that from the tungsten complex (≤ 1.8 mol per W atom), which requires about 0.5 h for complete reaction. We now find that ammonia is produced almost equally well, (1.7 mol per W atom), but much more slowly, from a methanol solution of A, (with M = W) either at the boil (3–4 h) or on irradiation for 30 h at 20 °C (2 × 150-W tungsten filament lamps); a boiling ethanol solution yields only 0.4 mol NH3 per W atom. In contrast, A, (with M = Mo) gives very little ammonia (only 0.08 mol per Mo at best) in any of the above conditions; instead it evolves dinitrogen and dihydrogen.
Similar content being viewed by others
Article PDF
References
Chatt, J., Pearman, A. J., and Richards, R. L., Nature, 253, 39 (1975).
Chatt, J., Pearman, A. J., and Richards, R. L., I. Organomental. Chem., 101, C45 (1975).
Shilov, A. E., Russ. Chem. Rev., 43, 5 (1974).
Manriquez, J. M., and Bercaw, J. E., J. Am. Chem. Soc., 96, 6229 (1974).
Brulet, C. R., and van Tamelen, E. E., J. Am. Chem. Soc., 97, 911 (1975).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
CHATT, J., PEARMAN, A. & RICHARDS, R. Relevance of oxygen ligands to reduction of ligating dinitrogen. Nature 259, 204 (1976). https://doi.org/10.1038/259204a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/259204a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.