Abstract
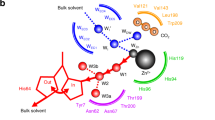
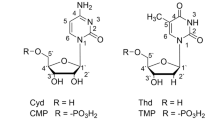
ACETAZOLAMIDE (2-acetylamino-1,3,4-thiadiazole-5-sulphonamide, ‘Diamox’) is the most potent known inhibitor of the zinc enzyme carbonic anhydrase. This communication reports the direct demonstration that binding of acetazolamide to human carbonic anhydrase requires the presence of a metal ion at the active site and that binding depends on the species of divalent metal ion present. Zinc (II) and cobalt (II) ions are the only ions which induce the formation of very stable acetazolamide carbonic anhydrase complexes and are also the ions which most effectively catalyse the hydration of carbon dioxide and the hydrolysis of p-nitrophenyl acetate. Metal-binding monodentate ions, CN−, HS−, OCN−, and N3−, known as effective carbonic anhydrase inhibitors, compete for the acetazolamide binding site of the zinc enzyme.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Coleman, J. E., and Vallee, B. L., Biochemistry, 3, 1874 (1964).
Langham, W. H., Eversole, W. J., Hayes, F. N., and Trujillo, T. T., J. Lab. Clin. Med., 47, 819 (1956).
Rickli, E. E., Ghazanfar, S. A. S., Gibbons, B. H., and Edsall, J. T., J. Biol. Chem., 239, 1065 (1964).
Lindskog, S., J. Biol. Chem., 238, 945 (1963).
Pocker, Y., and Meany, J. E., Biochemistry, 4, 2535 (1965).
Coleman, J. E., Biochemistry, 4, 2644 (1965).
Lindskog, S., and Nyman, P. O., Biochim. Biophys. Acta, 85, 462 (1964).
Tilander, B., Strandberg, B., and Fridborg, K., J. Mol. Biol., 12, 740 (1965).
Wilbur, K. M., and Anderson, N. G., J. Biol. Chem., 176, 147 (1948).
Duff, T. A., and Coleman, J. E., Biochemistry, 5, 2009 (1966).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
COLEMAN, J. Metal Ion Dependent Binding of Sulphonamide to Carbonic Anhydrase. Nature 214, 193–194 (1967). https://doi.org/10.1038/214193a0
Issue Date:
DOI: https://doi.org/10.1038/214193a0
This article is cited by
-
Elucidating the role of metal ions in carbonic anhydrase catalysis
Nature Communications (2020)
-
A catalytically active [Mn]-hydrogenase incorporating a non-native metal cofactor
Nature Chemistry (2019)
-
Evolution and diversity of alpha-carbonic anhydrases in the mantle of the Mediterranean mussel (Mytilus galloprovincialis)
Scientific Reports (2019)
-
Emergence of metal selectivity and promiscuity in metalloenzymes
JBIC Journal of Biological Inorganic Chemistry (2019)
-
Structure and metal exchange in the cadmium carbonic anhydrase of marine diatoms
Nature (2008)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.