Abstract
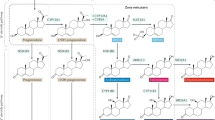
DURING the course of clinical investigations being carried out here the need arose for tritium-labelled testosterone of very high specific activity. Although 1,2-tritiated androgens are available1,2, their use was precluded in this instance by the extensive loss of radioactivity which would occur during the metabolism of the steroid3. Carrier-free (7-3H)-testosterone acetate was prepared by catalytic reduction in pure tritium of 17β-acotoxy-androstan-4,6-dien-3-one.
Similar content being viewed by others
Article PDF
References
Chang, E., and Kenny, N., Steroids, 1, 325 (1963).
Brodie, H. J., Hayano, M., and Gut, M., J. Amer. Chem. Soc., 84, 3766 (1962).
Morato, T., Raab, K., Brodie, H. J., Hayano, M., and Dorfman, R. I., J. Amer. Chem. Soc., 84, 3764 (1962).
Schaub, R. E., and Weiss, M. J., J. Org. Chem., 26, 3915 (1961).
Djerassi, C., Rosenkranz, G., Romo, J., Kaufmann, S., and Pataki, S., J. Amer. Chem. Soc., 72, 4534 (1950).
Caspi, E., Cullen, E., and Grover, P. K., J. Chem. Soc., 212 (1963). Ringold, H. J., and Turner, A., Chem. and Ind., 211 (1962).
Shepherd, D. A., Donia, R. A., Campbell, J. A., Johnson, B. A., Holysz, R. P., Slomp, G., Stafford, J. E., Pederson, R. L., and Ott, A. G., J. Amer. Chem. Soc., 77, 1212 (1955).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
COOMBS, M., RODERICK, H. Carrier-free (7-3H)-Testosterone. Nature 203, 523 (1964). https://doi.org/10.1038/203523a0
Issue Date:
DOI: https://doi.org/10.1038/203523a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.