Abstract
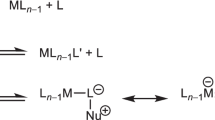
THE problem of assigning mechanisms to certain octahedral substitution reactions has been complicated by two principal ambiguities. If the reacting complex has acidic protons on the non-participating groups, second-order kinetics resulting from substitution by basic anions may indicate either an SN2 or an SN1CB mechanism, and, in general, it is difficult to distinguish the two. With non-basic substituting anions or systems without extractable protons, first order kinetics may indicate either an SN1 mechanism or substitution involving preliminary bimolecular solvent intervention. The compound cis-bis-pyridino-bis-o-phenanthroline ruthenium II perehlorate, cis-[Ru phen2 py2] (ClO4)2, has no extractable protons, and therefore should give a unique answer to the first ambiguity; and it was hoped that dry solvent acetone would not engage in preliminary bimolecular substitution.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BOSNICH, B. Bimolecular Substitution in Octahedral Transition Metal Ions, and the Concept of Amphiphilicity. Nature 196, 1196–1197 (1962). https://doi.org/10.1038/1961196a0
Issue Date:
DOI: https://doi.org/10.1038/1961196a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.