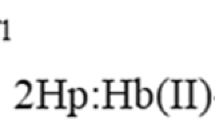Abstract
IT is generally considered that the hæm iron in hæmoglobin and myoglobin and their derivatives is bound to the apoprotein through histidine imidazole groups, although there is some uncertainty1 whether the stronger of the two postulated imidazoles (ascribed pK1 = 5.3 in ferrihæmoglobin and ferrimyoglobin2) has been so identified.
Similar content being viewed by others
Article PDF
References
Wyman, J., “Adv. Protein Chem.”, 4, 407 (1948).
Theorell, H., and Ehrenberg, A., Acta Chem. Scand., 5, 823 (1951).
O'Hagan, J. E., Ph.D. thesis, University of Queensland (1958).
Holden, H. F., Aust. J. Exp. Biol. and Med. Sci., 14, 291 (1936). Steinhardt, J., and Zaiser, E. M., “Adv. Protein Chem.”, 10, 151 (1955).
Lewis, U. J., J. Biol. Chem., 206, 109 (1954).
Cohn, E. J., Green, A. A., and Blanchard, M. H., J. Amer. Chem. Soc., 59, 509 (1937).
Theorell, H., and Paul, K. G., Ark. Kemi, Mineral. Geol., 18, A, No. 12 (1943).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
O'HAGAN, J. The Iron–Protein Link in Hæmoglobin and Myoglobin. Nature 183, 393 (1959). https://doi.org/10.1038/183393a0
Issue Date:
DOI: https://doi.org/10.1038/183393a0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.