Abstract
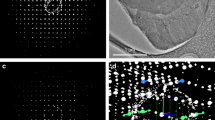
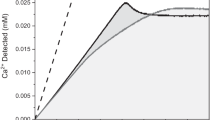
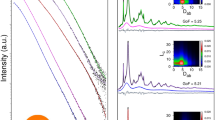
IT is commonly observed that a change in the size and shape of a crystal occurs when an impurity is added to the solution from which the crystal is grown. One such change, or habit modification, is that of crystals of gypsum (calcium sulphate), which in the presence of sodium citrate change from long needles to short stubby rods1. The mechanism of such habit modification of crystals is obscure, but the impurity is believed to be adsorbed on the surfaces of the growing crystal, and thus to cause an alteration in their rates of growth which results in the observed change in size and shape. Since the calcium and citrate ions associate in solution to form a complex, it was possible that other calcium compounds than gypsum adsorb the citrate ion, leading to habit modification of their crystals. In fact, the surface of the calcium phosphate crystals in bone has been supposed to be the site of the bone citrate2. It is therefore of interest to investigate the effect of citrate on the growth of calcium phosphate crystals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Lea, E. M., and Nurse, R. W., Discuss. Farad. Soc., 5, 345 (1949).
Hendricks, S. B., and Hill, W. L., Trans. Macy Conf. Metabolic Interrelations, 3, 173 (1951).
Waldman, J., Trans. Macy Conf. Metabolic Interrelations, 2, 203 (1950).
Brandengurger, E., and Schinz, H. R., Helv. Med. Acta, 12 (Supp. 16) (1945).
Moeller, H., and von Troemel, G., Z. anorg. Chim., 206, 227 (1932).
Dallemagne, M. J., J. Physiologie, 43, 425 (1951).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
PATTERSON, D. Effect of a Citrate Ion Impurity on the Size and Shape of Calcium Phosphate Crystals. Nature 173, 75–76 (1954). https://doi.org/10.1038/173075b0
Issue Date:
DOI: https://doi.org/10.1038/173075b0
This article is cited by
-
Precipitation of calcium phosphates from electrolyte solutions V. The influence of citrate ions
Calcified Tissue International (1979)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.