Summary:
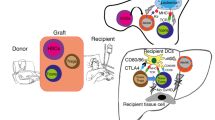
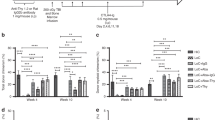
Donor lymphocyte infusion mediates most effective graft- versus-leukemia (GVL) effects following induction of host-versus-graft tolerance by transplantation of donor stem cells. This study was designed to maximize GVL effects across both major (MHC) and minor (mHgs) histocompatibility barriers in recipients inoculated with murine B-cell leukemia (BCL1), using specifically immune donor lymphocytes. GVL effects were induced with donor spleen cells from mice immunized across MHC or mHgs barriers with BCL/1 cells or normal BALB/c spleen cells. Our data suggest that spleen cells from donor mice immunized against murine B-cell leukemia of BALB/c origin, or to a lesser extent against normal host alloantigens, induce better therapeutic GVL effects with less great-versus-host disease (GVHD) across both mHgs and MHC. The cytokine profile of effector cells inducing predominantly GVL effects with reduced GVHD across MHC and mHg barriers consisted preferentially of upregulated IFN-gamma, IL-2, IL-10 and IL-12 in donors, implying a Th-1 to Th-2 cytokine shift. We hypothesize that immunotherapy with immune donor lymphocytes sensitized in vivo or in vitro with allogeneic tumor cells or normal host cells together with allogeneic BMT may provide an effective approach for amplifying GVL effects, while reducing procedure-related morbidity and mortality due to uncontrolled GVHD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Weiden PL, Fluornoy N, Sanders JE et al. Anti-leukemic effect of graft-versus-host disease contributes to improved survival after allogeneic marrow transplantation. Transplant Proc 1981; 13: 248–251.
Horowitz MM, Gale RP, Sondel PM et al. Graft-vs-leukemia reactions after bone marrow transplantation. Blood 1990; 75: 555–562.
Slavin S . Immunotherapy of cancer with alloreactive lymphocytes. N Engl J Med 2000; 343: 802–803.
Slavin S, Weiss L, Morecki S et al. Eradication of murine leukemia with histoincompatible marrow grafts in mice conditioned with total lymphoid irradiation (TLI). Cancer Immunol Immunother 1981; 11: 155–158.
Weiss L, Lubin I, Factorowich I et al. Effective graft vs leukemia effects independent of graft vs host disease after T-cell depleted allogeneic bone marrow transplantation in a murine model of B cell leukemia/lymphoma. Role of cell therapy and rIL-2. J Immunol 1994; 153: 2562–2567.
Truitt RL, Atasoylu AA . Impact of pretransplant conditioning and donor T cells on chimerism, graft-versus-host disease, graft-vs-leukemia reactivity, and tolerance after bone marrow transplantation. Blood 1991; 77: 2515–2523.
Slavin S, Strober S . Spontaneous murine B-cell leukaemia. Nature 1978; 272: 624–626.
Slavin S, Weiss L, Morecki S et al. Ultrastructural, cell membrane and cytogenetic characteristics of B-cell leukemia, a murine model of chronic lymphocytic leukemia. Cancer Res 1981; 41: 4162–4166.
Sander B, Hoiden I, Anderrson U et al. Similar frequencies and kinetics and cytokine producing cells in murine peripheral blood and spleen. Cytokine detection by immunoassay and intra-cellular immunostaining. J Immunol Methods 1993; 166: 201–214.
Abrams JS . Immunoenzymetric assay of mouse and human cytokines using NIP-labeled anti-cytokine antibodies. In: Caligan J, Knisbeck A, Margulis D, Shevach E, Strober W (eds). Current Protocols in Immunology. John Wiley and Sons: New York, 1995.
Cohen P, Vourka-Karussis U, Weiss L, Slavin S . Spontaneous and IL-2 induced anti-leukemic and anti-host effects against tumor- and host-specific alloantigens. J Immunol 1993; 151: 4803–4810.
Pugatsch T, Weiss L, Slavin S . Minimal residual disease in murine B cell leukemia (BCL1) Detected by PCR. Leuk Res 1993; 17: 999–1002.
Wilcoxon F . Individual comparisons by rank methods. Biometrics 1945; 1: 80–83.
Weiss L, Morecki S, Vitetta ES et al. Suppression and elimination of BCL1 leukemia by allogeneic bone marrow transplantation. J Immunol 1983; 130: 2452–2455.
Slavin S, Ackerstein A, Weiss L . Adoptive immunotherapy in conjunction with bone marrow transplantation – amplification of natural host defense mechanisms against cancer by recombinant IL-2. Nat Immun Cell Growth Regul 1988; 7: 180–184.
Morecki S, Pugatsch T, Levi S et al. Tumor-cell vaccination induces tumor dormancy in a murine model of B-cell leukemia/lymphoma (BCL1). Int J Cancer 1996; 65: 204–208.
Yefenof E, Picker LJ, Scheuermann RH et al. Cancer dormancy; isolation and characterization of dormant 1 cells. Proc Natl Acad Sci USA 1993; 90: 1829–1833.
Slavin S, Naparstek E, Nagler A et al. Allogeneic cell therapy with donor peripheral blood cells and recombinant human interleukin-2 to treat leukemia relapse post allogeneic bone marrow transplantation. Blood 1996; 87: 2195–2204.
Van Lochem E, De Grast B, Goulmy E . In vitro separation of host specific graft-versus-host disease and graft-versus-leukemia cytotoxic T cell activities. Bone Marrow Transplant 1992; 10: 181–183.
Zeis M, Uharek L, Glass B et al. Allogeneic MHC-mismatched activated natural killer cells administered after bone marrow transplantation provide a strong graft-versus-leukemia effect in mice. Br J Haematol 1997; 96: 757–761.
Slavin S, Ackerstein A, Kedar E, Weiss L . IL-2 activated cell-mediated immunotherapy: control of minimal residual disease in malignant disorders by allogeneic lymphocytes and IL2. Bone Marrow Transplant 1990; 6(Suppl 1): 86–90.
Ruggeri L, Capanni M, Casucci M et al. Role of natural killer cell alloreactivity in HLA-mismatched hematopoietic stem cell transplantation. Blood 1999; 94: 333–339.
Kolb HJ, Schattenberg A, Goldman JM et al. Graft-versus-leukemia effect of donor lymphocyte infusions in marrow grafted patients. Blood 1995; 86: 41–50.
Zeis M, Uharek L, Glass B et al. Eradication of residual disease by administration of leukemia-specific T cells after experimental allogeneic bone marrow transplantation. Exp Hematol 1998; 26: 1068–1073.
Morecki A, Puyesky Y et al. Induction of tumor immunity by intact irradiated leukemic B cells (BCL1) bearing a tumor-associated cell surface idiotype and the costimulatory B7 molecule. Cancer Immunol Immunother 1995; 41: 236–242.
Morecki S, Yacovlev E, Gelfand Y et al. Cell therapy with pre-immunized effector cells mismatched for minor histocompatible antigens, in the treatment of a murine mammary carcinoma. J Immunother 2001; 24: 114–121.
Blazar BR, Roopenian DC, Taylor PA et al. Lack of GVHD across classical, single minor histocompatibility (miH) locus barriers in mice. Transplantation 1996; 61: 619–624.
Rocha M, Umansky V, Lee KH et al. Differences between graft-versus-leukemia and graft-versus-host reactivity. Interaction of donor immune T cells with tumor and/or host cells. Blood 1997; 89: 2189–2202.
Blazar BR, Taylor PA, Panoskaltsis N et al. Opposing roles of CD28:B7 and CTLA-4:B7 pathways in regulating in vivo all responses in murine recipients of MHC disparate T cells. J Immunol 1999; 162: 6368–6377.
De La Selle V, Riche N, Dorothe G et al. CD8+ cytotoxic T cell repertoire implicated in graft-vs-leukemia effect in a murine bone marrow transplantation model. Bone Marrow Transplant 1999; 23: 951–958.
Taylor PA, Panoskatsis N, Mortari A et al. Analysis of the requirements for the induction of CD4+ T cell alloantigen hyporesponsiveness by ex vivo anti-CD40 ligand antibody. J Immunol 2000; 164: 612–622.
Weiss L, Weigensberg M, Morecki S . Characterization of effector cells of graft-vs-leukemia (GVL) following allogeneic bone marrow transplantation in mice inoculated with murine B-cell leukemia (BCL1). Cancer Immunol Immunother 1990; 31: 236–242.
Okubo T, Hagiwara E, Ohno S et al. Administration of an IL-12-encoding DNA plasmid prevents the development of chronic graft-versus-host disease (GVHD). J Immunol 1999; 162: 4013–4017.
Blazar BR, Taylor PA, Panoskaltsis N et al. Rapamycin inhibits the generation of graft-versus-host disease and graft-versus-leukemia-causing T cells by interfering with the production of Th1 or Th1 cytotoxic cytokines. J Immunol 1998; 160: 5355–5365.
Sefrioui H, Billiau AD, Overbergh L et al. Influence of the additional injection of host-type bone marrow on the immune tolerance of minor antigen-mismatched chimeras: possible involvement of double-negative (natural killer) T cells. Transplantation 1999; 68: 1560–1567.
Krenger W, Snyder KM, Byon JC et al. Polarized type 2 alloreactive CD4+ and CD8+ donor T cells fail to induce experimental acute graft-versus-host disease. J Immunol 1995; 155: 585–593.
Salgame P, Abrams JS, Clayberger C et al. Differing lymphokine profiles of functional subsets of human CD4 and CD8 T cell clones. Science 1991; 254: 279–282.
Seder RA, Boulay JL, Finkelman F et al. CD8+ T cells can be primed in vitro to produce IL-4. J Immunol 1992; 148: 1652–1656.
Fowler DH, Kurasawa K, Husebekk A et al. Cells of the Th2 cytokine phenotype prevent LPS-induced lethality during murine graft-versus-host reaction. Regulation of cytokines and CD8+ lymphoid engraftment. J Immunol 1994; 152: 1004–1013.
Slavin S, Nagler A, Naparstek E et al. Non-myeloablative stem cell transplantation and cell therapy as an alternative to conventional bone marrow transplantation with lethal cytoreduction for the treatment of malignant and non-malignant hematologic diseases. Blood 1998; 91: 756–763.
Slavin S, Naparstek E, Nagler A et al. Allogeneic cell therapy for relapsed leukemia following bone marrow transplantation with donor peripheral blood lymphocytes. Exp Hematol 1995; 23: 1553–1562.
Moscovitch M, Slavin S . Anti-tumor effects of allogeneic bone marrow transplantation in (NZB × NZW) F1 hybrids with spontaneous lymphosarcoma. J Immunol 1984; 132: 997–1000.
Morecki S, Moshel Y, Gelfand Y et al. Induction of graft vs tumor effect in a murine model of mammary adenocarcinoma. Int J Cancer 1997; 71: 59–63.
Childs R, Clave E, Contentin N et al. Engraftment kinetics after nonmyeloablative allogeneic peripheral blood stem cell transplantation: full donor T-cell chimerism precedes alloimmune responses. Blood 1999; 94: 3234–3241.
Acknowledgements
We wish to thank the Danny Cunniff Leukemia Research Laboratory, the Gabrielle Rich Leukemia Research Foundation, the Cancer Treatment Research Foundation, the Novotny Trust, the Szydlowsky Foundation, the Fig Tree Foundation, and Ronne & Donald Hess for their continuous support of our ongoing basic and clinical research.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ji, Y., Weiss, L., Zeira, M. et al. Allogeneic cell-mediated immunotherapy of leukemia with immune donor lymphocytes to upregulate antitumor effects and downregulate antihost responses. Bone Marrow Transplant 32, 495–504 (2003). https://doi.org/10.1038/sj.bmt.1704150
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704150