Abstract
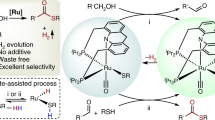
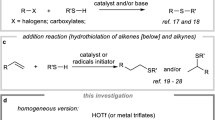
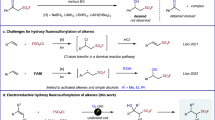
THE homolytic decarbonylation of aliphatic aldehydes regularly occurs at elevated temperatures in the vapour phase1. We have studied this reaction with a number of aliphatic aldehydes in the liquid phase at 90°–120° C., using the Me2C—CO2Me free radical (produced by the thermal decomposition of dimethyl-2 : 2′-azo-isobutyrate) as the initiating catalyst, and have found that only about 5 per cent of these radicals are effective as dehydrogenators (1). 
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
See Steacie, E. W. R., “Atomic and Free Radical and Reactions” (1946).
J. Amer. Chem. Soc., 69, 2917 (1947).
J. Org. Chem., 14, 248 (1949).
See preceding communication.
Waters, “The Chemistry of Free Radicals”, 257, 283 (1946).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
HARRIS, E., WATERS, W. Thiol Catalysis of the Homolytic Decomposition of Aldehydes. Nature 170, 212–213 (1952). https://doi.org/10.1038/170212a0
Issue Date:
DOI: https://doi.org/10.1038/170212a0
This article is cited by
-
Mitochondrial chemiluminescence elicited by acetaldehyde
Journal of Bioenergetics and Biomembranes (1982)
-
Comparison of some lipoxidases and their mechanism of action
Journal of the American Oil Chemists' Society (1957)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.