Abstract
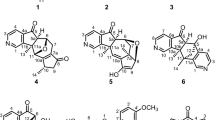
THE more significant experimental facts for basing the structure of α-fagarine, one of the alkaloids isolated from Fagara coco1, were the presence of one methylimino group, two methoxyls and one dioxymethylene group, and the formation of m-methoxybenzaldehyde by oxidation2. α-Fagarine behaves as a tertiary base, since it does not react with nitrous acid and also lacks rotatory power.
Similar content being viewed by others
Article PDF
References
Deulofeu, V., Labriola, R., and De Langhe, J., J. Amer. Chem. Soc. 64, 2326 (1942).
Deulofeu, V., Labriola, R., and Berinzaghi, B., J. Org. Chem., 12, 217 (1947).
Labriola, R., Ishii, M., and Mariani, I., An. Asoc. Quim. Arg., 33, 156 (1945).
Labriola, R., Dorronsoro, I., and Verruno, O., paper read before the Fourth South American Chemical Congress, Santiago, Chile, March 1948.
Slotta, K. H., and Haberland, G., Ber., 65, 127 (1932).
Ewing, G. W., and Steck, E. A., J. Amer. Chem. Soc., 68, 2181 (1946).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
DEULOFEU, V., LABRIOLA, R. & BERINZAGHI, B. Structure of α-Fagarine. Nature 162, 694 (1948). https://doi.org/10.1038/162694a0
Issue Date:
DOI: https://doi.org/10.1038/162694a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.